Abstract
Epiploic foramen entrapment (EFE) is a common cause of small intestinal colic in horses and may lead to intestinal strangulation. Strangulating intestinal obstruction impairs the gastrointestinal outflow and can lead to secondary gastric rupture and endotoxemia. Clostridioides difficile can cause enterotyphlocolitis with colic in horses of all ages, and the process is commonly referred to as C. difficile–associated disease (CDAD). Here we report the results of the postmortem examination of a 7-y-old Thoroughbred racehorse with concurrent CDAD, EFE, and gastric rupture that was euthanized following a history of colic over several days. A segment of distal jejunum and proximal ileum had passed through the epiploic foramen, and the intestinal wall was thickened and dark-red. The remaining small intestinal loops were distended and filled with blood-tinged contents. Peritonitis had resulted from escape of gastric contents into the abdominal cavity through a tear in the major curvature of the stomach. Histologically, the incarcerated segment had acute transmural hemorrhage with congestion and mucosal necrosis; neutrophilic infiltrates with fibrin thrombi were in the mucosa of the non-incarcerated small intestinal segments. C. difficile toxins were detected in the small intestinal contents, and C. difficile was isolated from the small intestine, colon, and cecum.
The epiploic foramen (EF; or Winslow foramen in human medicine) is an opening in the dorsal abdomen that connects the greater peritoneal sac and the omental vestibule, and is delimited by the caudate lobe of the liver, the portal vein, and the pancreas. 24 In horses, the EF is a 4–6-cm slit-like aperture. If for any reason the EF becomes dilated, the intestines, most often the distal jejunum and ileum, may pass through and become entrapped, which frequently results in intestinal strangulation and infarction.5,19,21 EF entrapment (EFE) is a common cause of small intestinal colic in horses, and ~5% of the animals that undergo colic surgery have this condition. 22 Despite the possibility of surgical correction, mortality associated with EFE is high because the strangulating lesion is life-threatening, and postsurgical recurrence can occur.4,23 In addition, there is total or partial intestinal obstruction, with reduction of gastrointestinal outflow and increased intraluminal pressure on proximal segments of the alimentary canal, which may suffer other complications, including secondary gastric rupture.18,21
Clostridioides difficile (formerly Clostridium difficile) is a gram-positive, anaerobic, spore-forming bacillus, which may be a normal component of the intestinal microbiota, but can cause enterotyphlocolitis in horses of all ages mainly via the production of 2 toxins, toxin A (TcdA) and toxin B (TcdB). 9 C. difficile–associated disease (CDAD) in adult horses usually has a more protracted clinical course than in foals, and clinical signs include abdominal pain, fever, and diarrhea.9,20 Antibiotic administration and hospitalization may predispose horses to develop CDAD, but other, non-defined risk factors could play a role.8,25 On autopsy, there may be watery and hemorrhagic-to-green intestinal contents and pseudomembrane formation; adult horses usually have a more aboral distribution of the lesions, with the cecum and colon most often affected, although lesions in the small intestine can also occur. 8 Histologically, there is necrosis and ulceration of the mucosal epithelium, with variable neutrophilic infiltrates in the lamina propria, fibrin thrombi, and rare volcano ulcers. Diagnosis is confirmed by the detection of TcdA and TcdB in intestinal contents by ELISA.8,20
Here we describe the results of the postmortem examination of a horse that had concurrent CDAD, EFE, and gastric rupture. We discuss the possible sequence of events and pathogenesis, and review some of the available literature about these conditions in horses.
A 7-y-old, Thoroughbred racehorse gelding was submitted for autopsy and diagnostic workup to the San Bernardino branch of the California Animal Health and Food Safety laboratory system (CAHFS; University of California–Davis). The horse had a history of colic over several days and remained in the barn with minimal veterinary care. Despite treatment, including acepromazine, flunixin, phenylbutazone, butorphanol, and mineral oil via nasogastric tube, the horse continued to decline and was euthanized with a pentobarbital overdose.
On gross examination, the carcass was in fair nutritional condition. There was peritonitis with 3 L of dark-red, dense, and turbid fluid with fragments of vegetal material and fibrin in the abdominal cavity. There was a ~20-cm tear through the gastric major curvature, involving the serosal and muscular layers, with hemorrhage in the borders, and a 5-cm, full-thickness wall perforation close to the proximal margin of the tear (Fig. 1A). The duodenum and most of the jejunum were distended with watery, slightly blood-tinged contents, and had light-red mucosal surfaces. A 40- to 50-cm loop of distal jejunum and proximal ileum had passed through a dilated EF from the left to the right side of the abdomen (Fig. 1B); an area of constriction at the point of entrapment, and the entrapped intestinal segment, had a partial, 90°–180° volvulus with the axis of the twist close to the EF. The wall of this entrapped and partially twisted segment was thickened and had congested serosal and mesenteric vessels, dark-red mucosal surface, and partially clotted blood in the lumen. The large colon and cecum had scant, pasty contents, and the small colon and rectum contained scant fecal balls. Extra-gastrointestinal gross lesions included pulmonary edema with pleural hemorrhages and petechiae in the epicardium. In addition, there was mild-to-moderate degenerative joint disease in several joints compatible with a racehorse in active training and racing.
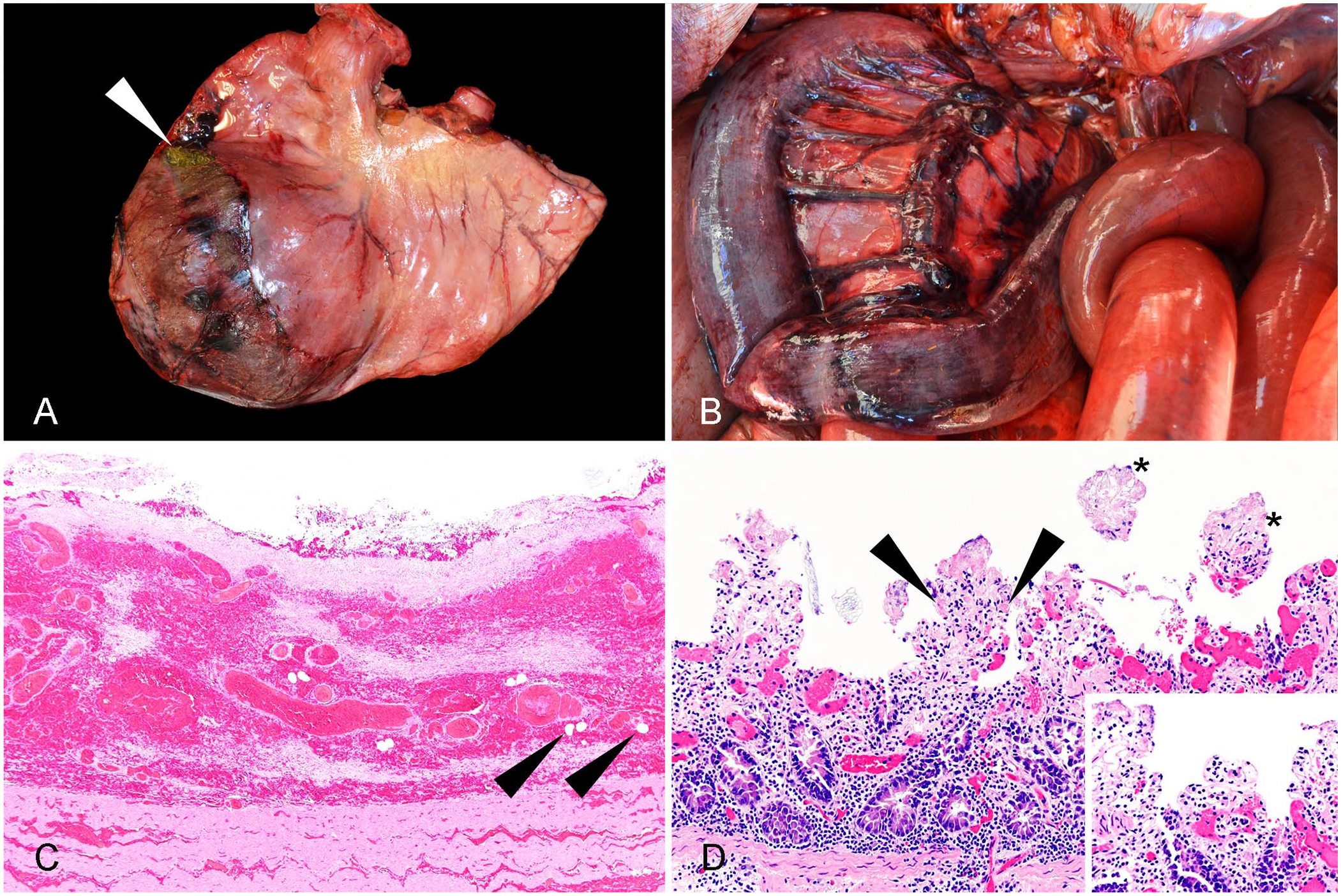
Gross and microscopic findings in a Thoroughbred racehorse with epiploic foramen entrapment (EFE), Clostridioides difficile–associated disease (CDAD), and gastric rupture.
Histologic examination was performed on lung, spleen, kidneys, lymph nodes, small intestine (including sections of duodenum and jejunum that were not involved in the entrapment, and sections of jejunum and ileum that were involved in the entrapment), colon, cecum, stomach, esophagus, liver, heart, adrenal gland, and trachea that had been fixed by immersion in 10% neutral-buffered formalin for 24 h, processed routinely to produce 4-μm sections, and stained with H&E; selected sections of small intestine were stained with Gram stain.
Ancillary tests were performed following CAHFS standard operating procedures as follows: equine enterotyphlocolitis bacteriology panel, which included bacterial aerobic and anaerobic cultures, Salmonella spp. enrichment cultures, C. difficile enrichment cultures, C. perfringens toxins (alpha, beta, epsilon) ELISAs, and C. difficile toxin (TcdA/TcdB) ELISAs on small intestine, colon, and cecum (cultures) and their contents (ELISAs); bacterial aerobic cultures from the abdominal cavity and lung; fecal flotation for parasitologic examination; and heavy metals (including lead, manganese, iron, mercury, arsenic, molybdenum, zinc, copper, cadmium) and selenium concentrations screen from liver.
Histologically, the sections of jejunum and ileum involved in the entrapment had severe transmural congestion and dissecting hemorrhage, a pseudomembrane attached to the necrotic mucosa, rare fibrin thrombi and fibrinoid necrosis in the mucosal and submucosal vessels, occasional gas bubbles in the submucosa, and fibrin and debris on the serosal surfaces (Fig. 1C). Within sections of jejunum not involved in the entrapment, there was marked mucosal congestion, rare fibrin thrombi in small capillaries of the superficial lamina propria, small aggregates of neutrophils, and sloughed epithelial cells with rare gram-positive and -negative coccobacilli in the lumen (Fig. 1D). Sections of the stomach at the level of the rupture had transmural hemorrhage that dissected the mucosal and muscular layers, and fibrin with vegetal material attached to the serosa. Lung sections were markedly congested and had alveolar edema and subpleural hemorrhage. Congestion of lymph nodes, kidneys, and liver was marked and diffuse.
C. difficile TcdA/TcdB were detected in small intestinal contents, which confirms a diagnosis of CDAD,8,20 and C. difficile was isolated from small intestine, colon, and cecum (Table 1). Isolation of C. difficile is not confirmatory, given that a small percentage of healthy horses may carry the organism6,9; however, in our case, bacterial isolation in 3 different intestinal segments further supports a diagnosis of CDAD given that the toxins were also detected. Paeniclostridium sordellii (formerly Clostridium sordellii) is another toxin-producing, anaerobic, gram-positive rod of the Clostridiaceae family that has been associated with enterocolitis and hemorrhagic enteritis in horses and can rarely be isolated from the intestine of healthy individuals.12,15,17 In our case, P. sordellii was isolated consistently from the 3 different intestinal segments, and a degree of involvement in tissue damage cannot be excluded. Nevertheless, given that a solid diagnosis of another known cause of enterocolitis in horses (CDAD) was made, the role of P. sordellii was probably secondary; exclusion of other agents has been proposed among the diagnostic criteria for P. sordellii–associated enterocolitis in horses.15,20 In addition, large numbers of Escherichia coli and mixed flora were isolated from the abdominal cavity and were considered possible contributors to peritonitis further to release of gastric content to the abdominal cavity. As well, no bacteria were isolated from the lung, no parasite eggs or oocysts were detected by fecal flotation, and the hepatic heavy metal and selenium concentrations were within acceptable or nondiagnostic ranges for horses.
Equine enterotyphlocolitis bacteriology panel in small and large intestine of a Thoroughbred racehorse with Clostridioides difficile–associated disease, epiploic foramen entrapment, and gastric rupture.

A. rossii (Actinobacillus rossii); C. difficile (Clostridioides difficile); E. cloacae (Enterobacter cloacae); E. coli (Escherichia coli); P. sordellii (Paeniclostridium sordellii); CPA = Clostridium perfringens alpha-toxin; CPB = C. perfringens beta-toxin; D = detected; ETX = C. perfringens epsilon-toxin; Lg = isolated or detected in large numbers; Mod = isolated or detected in moderate numbers; ND = not detected; Sm = isolated or detected in small numbers; TcdA = C. difficile toxin A; TcdB = C. difficile toxin B; + = positive; − = negative.
In small intestine, cultures were taken from the mucosa of the entrapped segment, whereas the content of the non-entrapped segments was used for the ELISAs.
Gastric rupture is almost invariably fatal because, as occurred in our case, the contents are released to the abdominal cavity, causing septic peritonitis, endotoxemia, and the systemic inflammatory response syndrome. 7 Secondary gastric rupture is a common complication of equine colic associated with strangulating and non-strangulating intestinal obstruction, which can reduce outflow in the stomach, thus fostering distension and eventual rupture.7,21 Cases of small intestinal herniation or strangulation, which may include EFE, were reported among the lesions associated with gastric rupture in 2 of the first large case series about this condition in horses.13,18 Non-strangulating causes can include enteritis and colitis, such as CDAD, which may also promote ileus and reduce gastrointestinal outflow.11,18 Therefore, in our case, it is likely that gastric rupture was the latest event and occurred secondarily to EFE or to a combination of EFE and CDAD.
CDAD could have occurred before or during the course of EFE. Enteritis and diarrhea can increase gastrointestinal motility, and dilation of the EF for any reason could lead to herniation. 21 CDAD in adult horses has a longer clinical course than in foals. 20 In our case, the neutrophilic aggregates and fibrin thrombi detected in the lamina propria of the segments of small intestine not involved in the herniation were likely related to CDAD. 8 The microscopic lesions observed in the wall of the incarcerated segment seemed very acute, with a predominance of hemorrhage and necrosis without remarkable neutrophilic tissue infiltrates, hemosiderosis, or fibrosis. Considering the stereotypical response of the equine gut to hypoxia,14,21 these changes would suggest that ischemia occurred for no more than 24–48 h, and therefore support the view that EFE was a consequence of CDAD in our case. On the other hand, proliferation of anaerobes, including clostridia, tends to occur in the lumen of the languishing ischemic segments involved in a strangulating intestinal displacement, and toxin production can supersede and thus complicate the local and systemic effects of ischemia. 21 In fact, a 2019 study demonstrated that horses with colic shed significantly more C. difficile than did healthy individuals. 16 Altogether, these arguments suggest that CDAD was a consequence of the alteration of the intestinal microbiota by EFE, which would have occurred first, something also supported by the fact that no significant disease was detected in the large intestine of this horse.
In horses, we found no reports of intestinal displacements associated with CDAD in the scientific literature. Of 21 horses with CDAD, a hospitalized Warmblood had had surgical correction of EFE 8 ; however, CDAD occurred after the surgery, and was likely related to gentamicin treatment and/or hospitalization, given that these are considered risk factors for CDAD. In the same study, 8 a Thoroughbred horse with pelvic flexure impaction developed in-hospital fever and diarrhea shortly after admission, and died with CDAD. The racehorse of our report had a history of colic of several days duration and received various treatments. Antimicrobial therapy was not reported among those treatments, and the animal was not hospitalized. Nevertheless, the horse remained in the barn in a convalescent state under medical treatment and likely stress, which are considered among other less well-defined risk factors for CDAD by some authors. 20 In human medicine, transverse colon volvulus associated with CDAD was reported in an adult patient, and the authors suggested that the volvulus occurred as an event secondary to the pseudomembranous colitis caused by C. difficile. 26
EFE does not have a well-defined cause and was classically considered a relatively random event in equine populations.10,19 More recently, epidemiologic studies have identified some risk factors for EFE development, including, among others, crib-biting or windsucking behavior, being a Thoroughbred or a Thoroughbred-cross, greater height, colic episodes in the 12 mo prior to EFE, and prolonged periods of stall rest within 28 d before the entrapment.1–3,5 The racehorse in our report was a Thoroughbred, and the colic episode occurred in March over the course of a few days. There was no additional information available regarding the presence or absence of other risk factors. In this regard, the lack of specific timeframe for the clinical course and treatments applied in the days preceding euthanasia is a limitation of our report. A more thorough description of the clinical signs could have helped to establish whether CDAD occurred before or during the course of EFE.
In equine medicine, strangulating intestinal displacements are life-threating conditions that can cause death. Therefore, it is possible that veterinary pathologists do not request additional bacteriologic testing when one of these conditions is encountered on autopsy, because it may be considered enough to explain death. This could lead to underreporting associations such as the one in our case; a postmortem diagnosis of CDAD has not been reported previously in association with EFE and secondary gastric rupture, to our knowledge.
Footnotes
Acknowledgements
CAHFS autopsy and histology technicians, bacteriologists, and chemical analysts that performed some of the diagnostic tests of this case are deeply acknowledged.
Declarations of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
