Abstract
In the Amazon region of northern Brazil, Panicum maximum cultivars Mombaça, Tanzânia, and Massai cause severe colic and death in horses and mules. The disease occurs in the rainy season, when sprouting pastures are grazed by equidae. In the 8 separate disease outbreaks studied, a total of 52 out of 153 equidae were affected, including 19 that died (10 mules and 9 horses). Clinical signs were colic and abdominal dilatation, with a clinical manifestation period of 12 hr to 4 days. Serum activities of gamma-glutamyl transferase and aspartate aminotransferase were within reference intervals; however, serum urea nitrogen and creatinine concentrations were occasionally elevated. The primary gross and histologic lesions were observed in the digestive system. The stomach, small intestine, and large intestine had severe hemorrhages and occasional mucosal erosions and ulcerations. Ulceration and hemorrhage of the urinary bladder were rarely observed. Histologic examination revealed diffuse lymphoplasmacytic gastritis and enteritis with severe congestion, hemorrhage, and occasional epithelial necrosis and ulceration. Lymphocellular necrosis was occasionally observed within gut-associated lymphoid tissue. Tubular nephrosis occurred in some animals. Degeneration and occasional necrosis of bile duct epithelial cells and degeneration of hepatocytes were observed in the liver. Toxic pastures were negative for diosgenin- and yamogenin-based saponins, and oxalate concentrations were within reference intervals for the species. The toxin or toxins causing disease and the reason for the toxicity of the plant in the northern region are unknown.
In the last 15 years, an acute disease causing colic and bloat in horses and mules has been frequently reported by veterinary practitioners and government agencies in the states of Pará, Maranhão, Tocantins, Acre, and Rondonia in the Amazon region of Brazil during the rainy season (November-May). Pastures with affected animals have contained Panicum maximum cultivars Mombaça, Tanzânia, and Massai that were developed by EMBRAPA (Brazilian Enterprise for Research on Farming and Cattle Raising). These cultivars originated from 156 accessions of P. maximum Jacq. introduced into Brazil from Africa by an agreement with the Research Institute for Development. The selection of cultivars started in 1984, and the commercialization of the different cultivars began in 1990 for cv. Tanzânia, in 1993 for cv. Mombaça, and in 2001 for cv. Massai. 1,8 These cultivars were introduced in northern Brazil, where they have been used as a rotational grazing feed source for cattle and equidae. Nearly 10 million hectares in Brazil have been planted with P. maximum varieties, which represents approximately 20% of the total area occupied by planted pastures. 9 An analysis 1 of the nutritive value of the leaves of different cultivars of P. maximum yielded 6–12% crude protein, 40.3–48.5% acid detergent fiber, 28.9–37% cellulose, 5.3–9.8% lignin, and 2.1–6.9% silica. The objectives of the current paper are to report the epidemiology, clinical signs, and pathology of a disease of equidae associated with grazing lush sprouting pastures of P. maximum.
Location, month, and year of occurrence; cultivar of Panicum maximum in the pasture; and morbidity and mortality rates in 8 disease outbreaks associated with Panicum spp. ingestion.*
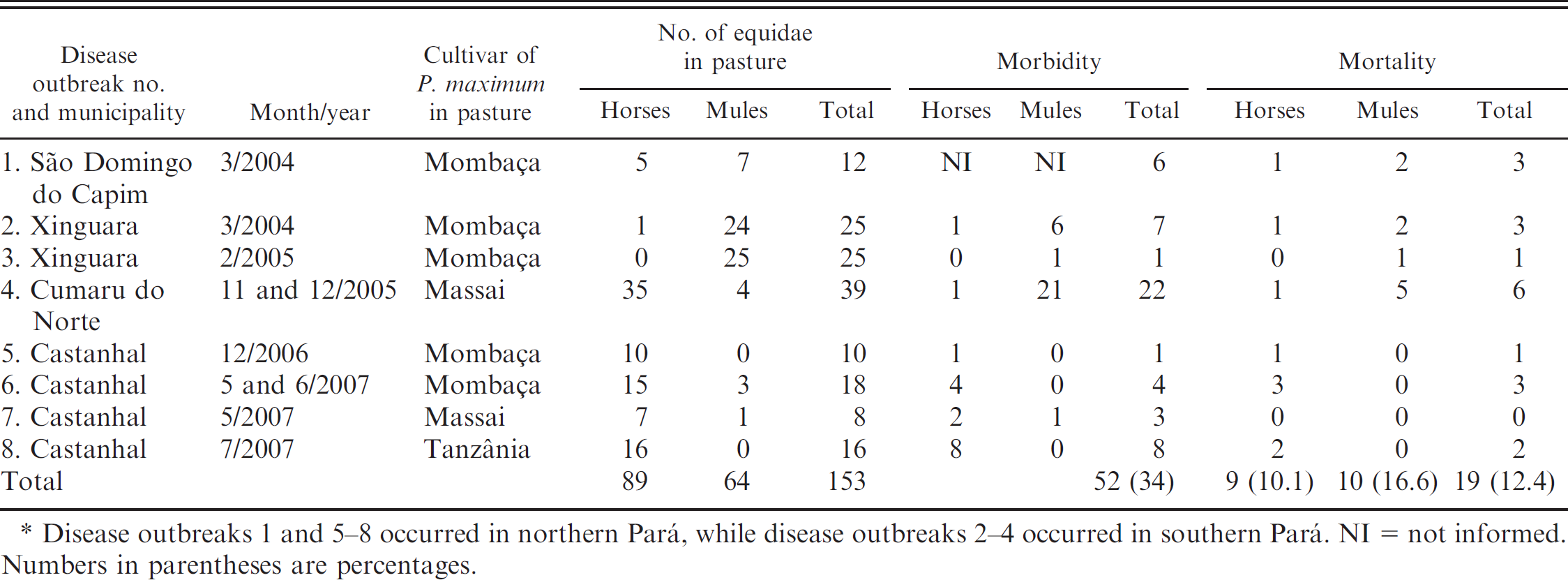
Disease outbreaks 1 and 5–8 occurred in northern Pará, while disease outbreaks 2–4 occurred in southern Pará. NI = not informed. Numbers in parentheses are percentages.
Eight outbreaks of disease were studied, including 5 outbreaks in northern Pará and 3 outbreaks in southern Pará. The municipalities in which the disease outbreaks occurred, month and year of occurrence, cultivar of P. maximum in the pasture, and morbidity and mortality rates in these outbreaks are shown in Table 1. Outbreak 1 occurred in Sao Domingo do Capim. The animals were in 2 paddocks, one with P. maximum cv. Mombaça (Fig. 1), planted 3 years prior to the outbreak, and another with P. maximum cv. Mombaça mixed with Brachiaria brizanta and small amounts of P. maximum cv. Tanzânia, planted 1 year before the outbreak. Disease outbreaks 2 and 3 occurred at the same farm in Xinguara in 2 paddocks containing P. maximum cv. Mombaça planted in 1998. These outbreaks occurred in March 2004 and February 2005. The rainy period in this region extends from October through March, with an increased intensity of rain showers from January to March. Outbreak 4 occurred in November and December of 2005 in a farm in Cumaru do Norte. In this area, the rainy season starts in September. This farm was divided into 11 units, and each unit contained a herd of 20–25 adult mules and 1 or 2 adult horses in 40-ha paddocks. In 3 of the paddocks, animals grazed on new growth of P. maximum cv. Massai that had been planted 2 years earlier. The other herds were in pastures of Brachiaria radicans and Brachiaria humidicola. The herds of 2 units were affected after being introduced into 2 paddocks with P. maximum pastures. Disease outbreaks 5 and 6 occurred in Castanhal in two 10-ha paddocks containing P. maximum cv. Mombaça planted 3 years earlier. In outbreak 5, horses had been grazing in the paddocks for 5 months. In November, the pastures were vacated for 30 days after previously being grazed by cattle. In December, a herd of horses was introduced into the pasture as the grass was initiating growth. Eight days after introduction into the paddock, 6 horses were sick and 1 horse died. In outbreak 6, a herd of 9 horses and 3 mules from another farm were introduced into 2 paddocks with P. maximum pastures. These animals remained in the pastures for 60 days without problems. Because both pastures were mature, cattle were introduced into one pasture and the horse and mule herd stayed in the other pasture. After the pasture was grazed by cattle, it was vacated for 30 days. The farmer then introduced 12 recently purchased horses and mules along with 10 resident horses from the farm into the pasture. In June, following 35 days of grazing, 2 horses from the farm became ill and died. Outbreak 7 occurred in Castahnal in a 2-ha pasture of P. maximum cv. Massai planted in May 2006. Seven horses and 1 mule were introduced into the paddock after the start of the rainy season. Two of the horses and the mule were affected. Outbreak 8 also occurred in Castanhal, where adult horses in the stable were fed with freshly cut P. maximum cv. Tanzânia from a pasture that had been planted 90 days earlier. On the same farm in July 2007, 3 horses became ill and 2 horses died while being maintained in a pasture of P. maximum cv. Tanzânia planted 8 months previously. In all farms, livestock illness occurred in almost-pure pastures of P. maximum, with few contaminating weeds or other grasses.
Four municipalities in the southern region of Pará, where the disease had occurred previously, were visited in December 2007 to determine the type of pastures, frequency of disease, and clinical signs. In this region, illness had been reported since 2002. According to one farmer, horses were affected more frequently than mules. In recent years, the disease had become less severe, and most affected animals recovered. Farmers mentioned that disease had occurred in pastures of P. maximum during the rainy season in many farms throughout the region. In pastures with other grass species, outbreaks of disease were not reported. In addition, there have been no reports of cattle being affected by this disease after introduction into pastures containing P. maximum.
Manifestation of clinical signs varied from 12 hr to 4 days. Signs of colic were moderate to severe and included flank watching, repeated lying down and rising, and rolling (Fig. 2). The abdomen was usually dilated bilaterally, but right-sided abdominal dilatation was present in 2 animals (Fig. 2B, 2C). Affected animals were depressed, and the mucous membranes appeared congested or cyanotic. With increasing dehydration, the oral mucosae appeared dry, with occasional purple discoloration. Perfusion time varied from 2 to 5 sec, cardiac rate varied from 46 to 120 beats/min, and respiratory rates varied from 16 to 48 respirations/min. Nasogastric reflux occurred in 4 animals, and the reflux appeared greenish and putrid in 3 of these animals and red-tinged in the remaining animal. Rectal examination was difficult in most ill animals because of the extent of the bowel distention. In one animal, the small intestine was dilated and contained a large amount of liquid; gas was present in the cecum; the contents of the colon were dry. On abdominal auscultation, enteric sounds varied with the degree of clinical illness. When animals had severe colic, gut sounds were absent. When colic was less severe, peristaltic movements returned. Normal bowel sounds were present in animals with mild colic. One affected animal had nervous signs, including apathy, severe ataxia, decreased reflexes of the upper lip and palate, decreased sensitivity of the nasal mucosa and skin, and decreased tone of the tail tonus and anal reflex. This animal also had edema of the prepuce, penile extrusion, urine leakage, and a distended urinary bladder. The animal's breath had an ammonia odor, and an ulcer was observed on the upper lip.

Panicum maximum cv. Mombaça.
In disease outbreaks in the municipality of Castanhal, affected animals were treated with 1.1 mg/kg of flunixin-meglumine, nasogastric intubation to promote gastric emptying, and intravenous administration of approximately 12 liters of lactated Ringer's solution mixed with 200 ml of 16.6% calcium gluconate and 200 ml of 5% sorbitol. Animals without reflux during nasogastric intubation were rehydrated orally. Of the 19 horses that were treated, 14 recovered and 5 died.

Clinical signs of colic in 3 horses and 2 mules.
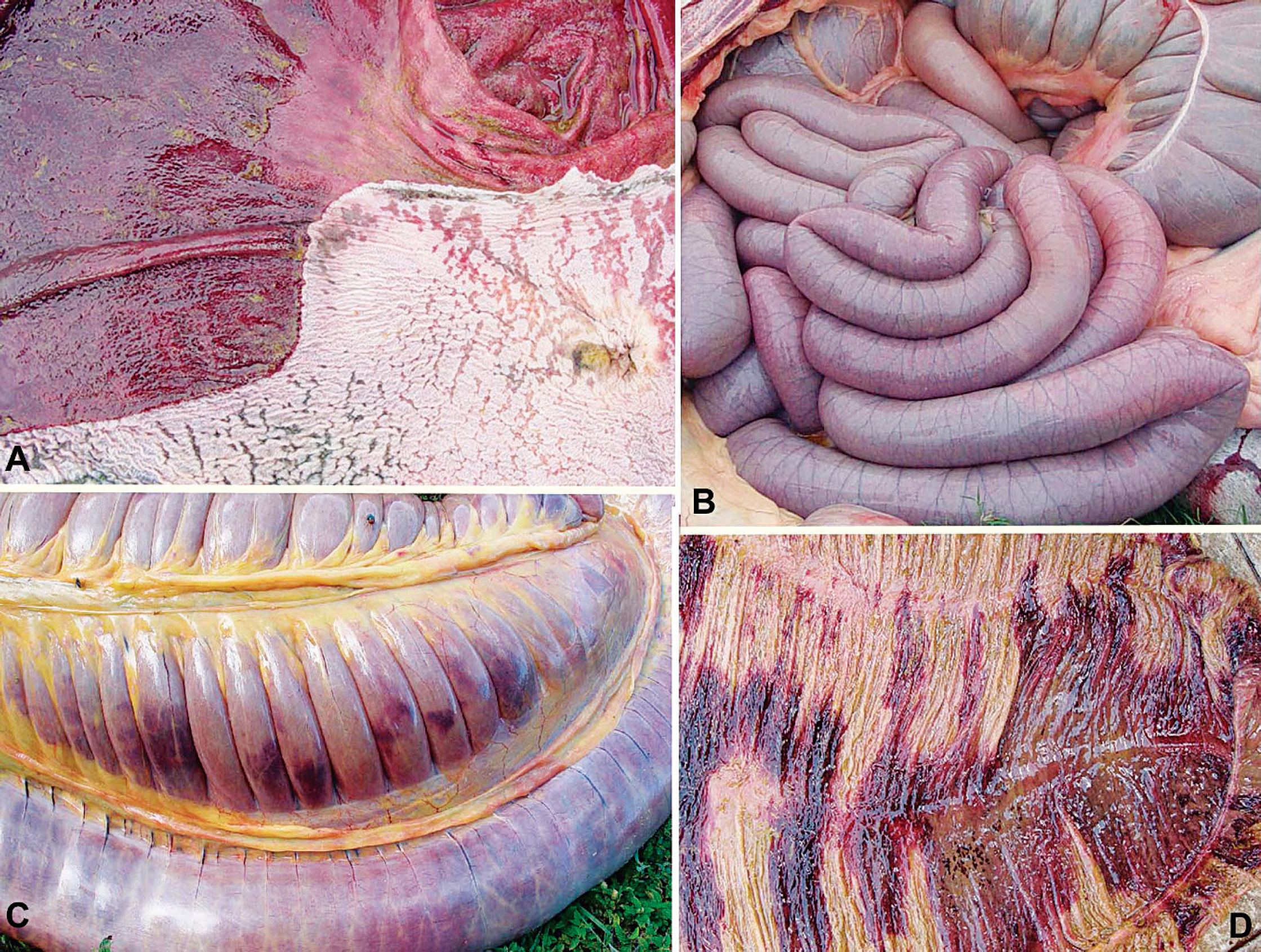
Gross pathology in equidae with colic.
Serum urea nitrogen and creatinine concentrations were within reference intervals or slightly increased. In disease outbreak 4, 16 animals with clinical signs of colic had serum urea nitrogen concentrations of 90.69 ± 51.21 mg/dl and creatinine concentrations of 3.08 ± 2.81 mg/kg. Nineteen animals without clinical signs had serum urea nitrogen concentrations of 51.49 ± 12.77 mg/dl and creatinine concentrations of 2.76 ± 0.69 mg/dl. Serum activities of aspartate aminotransferase (AST) and gamma-glutamyl transferase (GGT) were within reference intervals. Hematologic values were within reference intervals except for an increased packed-cell volume (36.1 ± 6.9%) due to dehydration.
Twelve animals that died spontaneously were necropsied, including 1 animal each from disease outbreaks 1, 3, and 5; 2 animals from disease outbreaks 2, 6, and 8; and 3 animals from disease outbreak 4. All animals had marked gastric dilatation, and the stomach contents were green, liquid, and fetid, with the exception of 2 animals that had a semi-solid gastric content. In 11 animals, the glandular stomach was diffusely dark red (Fig. 2A) and edematous. In 6 animals, circular or oval darker depressions ranging from approximately 1 mm to 1 cm in diameter were distributed within the mucosa. Two animals had ulcers measuring approximately 1 cm in diameter near the margo plicatus. The nonglandular stomach was slightly reddened in 3 animals, while 6 animals had multifocal, sometimes coalescent erosions of the mucosa (Fig. 2A). In 3 animals, diffuse erosions were observed near the margo plicatus. The small intestine was distended by green liquid content (Fig. 2B), and the mucosa appeared red. One animal had round, prominent, 1-cm-diameter hemorrhages. Congestion or hemorrhage was observed on the serosal surface of the cecum and colon (Fig. 3C). The cecum of 3 animals had a red, hemorrhagic mucosa and contained semi-solid material with mild to moderate amounts of gas (Fig. 3D), while cecal erosions were present in another animal. The colonic contents were semi-solid to dry in 3 animals, and transverse, linear, pink to red areas were observed throughout the mucosa. In these areas, the gut contents were hemorrhagic. The small colon had dry contents, with the presence of blood striae. The esophagus of one animal had longitudinal erosions of the mucosa, mainly in the thoracic and abdominal portions. The liver had an increased lobular pattern and sometimes appeared yellow or enlarged. In 2 animals, the mucosa of the urinary bladder was diffusely red. The urinary bladder of one animal also had small hemorrhages.
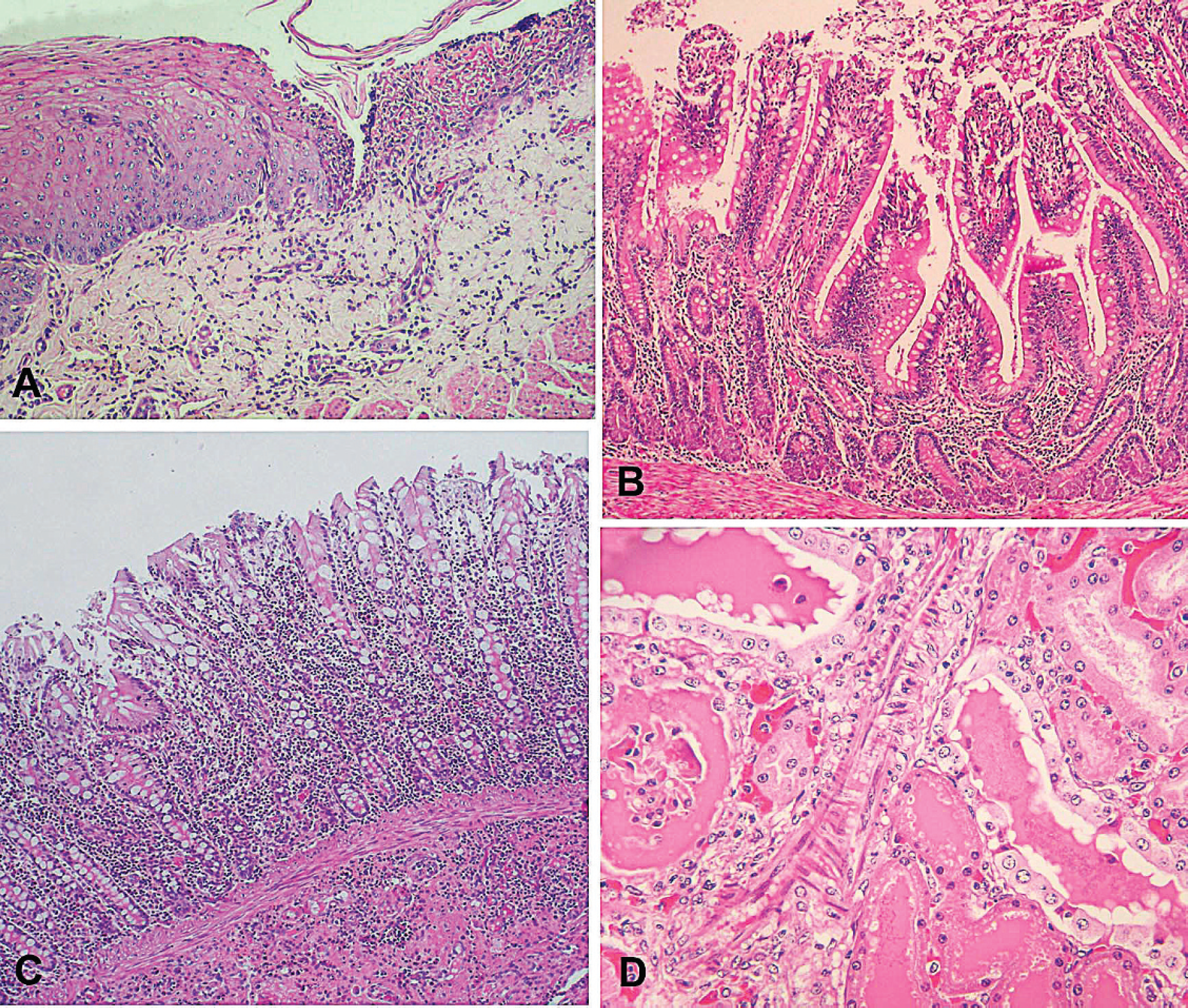
Microscopic pathology in equidae with colic.
Samples of the digestive system and other organs of the abdominal and thoracic cavities and central nervous system of 9 horses were fixed in 10% buffered formalin, trimmed, processed routinely, embedded in paraffin, sectioned at 4–6 μm, and stained with hematoxylin and eosin. In these horses, the primary histologic lesions were in the intestine. Both the large and small intestines had severe, diffuse enteritis, with infiltration of the lamina propria by mononuclear cells and numerous plasma cells (Fig. 4B, 4C). In most histologic sections, similar infiltrates also were observed in the submucosa (Fig. 4C). In 4 animals, eosinophils were also present, generally in association with helminths. Severe congestion with mucosal epithelial cell necrosis and sloughing were observed along the villi (Fig. 4B). Occasionally, villi were flattened and fused. In the large intestine, necrosis and sloughing of epithelia cells were occasionally observed along the surface of the intestinal glands. One animal had lymphocellular necrosis within the gut-associated lymphoid tissue of the large intestine.
The glandular epithelium of the stomach had multifocal to diffuse necrosis of epithelial cells, congestion, and hemorrhages. Mild to moderate infiltration of mononuclear cells was observed in the lamina propria. In 2 animals, severe infiltration by neutrophils was observed in the areas of epithelial necrosis. Necrosis of the gut-associated lymphoid tissue was also observed in 2 animals. One animal had no lesions of the glandular stomach. The nonglandular stomach of 4 animals had severe ballooning degeneration of the epithelial cells, with vesicle or pustule formation and multifocal erosions or ulcers (Fig. 4A). The submucosa was infiltrated by mononuclear cells and, in the ulcerated areas, by neutrophils. Congestion and hemorrhages of the submucosa were observed in tissue sections from some animals. In the nonglandular stomach of one animal, only mild ballooning degeneration of the squamous epithelium was observed. One animal had multifocal necrosis and ulceration of the epithelium of the esophagus.
Nephrosis characterized by vacuolation and necrosis of the renal tubular epithelium with numerous intratubular hyaline casts was observed in the kidneys of 5 animals (Fig. 4C). Other renal tubules were dilated and lined by flattened epithelium. Rare oxalate crystals were observed in the tubular lumina of one animal. Mild to moderate congestion was observed in the cortex and medulla.
In the liver, some bile ducts had mild vacuolation and occasional necrosis of epithelial cells. Other microscopic lesions included mild to moderate proliferation of bile duct epithelial cells, mononuclear infiltrates in portal areas, and mild bile retention and fine vacuolation of hepatocytes. In one animal the liver had scattered necrotic hepatocytes and multifocal abscesses. In another animal, the portal spaces and occasionally the hepatic parenchyma were infiltrated by neutrophils.
In one animal the urinary bladder had epithelial cell necrosis with ulceration of the mucosa and severe congestion and hemorrhages of the submucosa. Microscopic lesions were not present in the urinary bladder of the other animals. The mesenteric lymph nodes had congestion hemorrhages and edema. No major lesions were observed in the brain, spleen, heart, adrenal glands, thyroid glands, and pancreas.
In disease outbreaks 4–8, samples of pasture were collected to count Pithomyces chartarum spores 4 and to determine the presence of diosgenin- and yamogenin-based saponins by gas chromatography-mass spectrometry 7,15 In disease outbreaks 4–6, pasture samples were collected and analyzed for oxalate concentrations. 11 Spore counts of P. chartarum were negative or revealed <5,000 spores/g of sample. The 5 samples analyzed for diosgenin- and yanogenin-based saponins were negative. Samples of P. maximum cv. Massai collected in disease outbreak 4 had 1.73% total oxalates, samples of P. maximum cv. Mombaça collected in disease outbreak 5 contained 1.36% total oxalates, and samples of P. maximum cv. Mombaça from disease outbreak 6 contained 2.03% total oxalates. The other 13 samples collected in pastures in which disease outbreaks did not occur contained 0.28–1.39% total oxalates.
Epidemiological data demonstrated that the presence of disease was associated with pastures containing P. maximum (cv. Mombaça, Massai, and Tanzânia). Disease outbreaks were not observed in other pastures on the same farms; however, clinical disease appeared only after the introduction of P. maximum cultivars in the region. The possibility that the disease was caused by other grass or weed species was definitively excluded in disease outbreak 8, in which affected adult horses were fed in the stable with P. maximum cv. Tanzânia harvested from the field. Further, in the last few years the disease has become well known in the states of Pará, Maranhão, Tocantins, Acre, and Rondonia, always in association with nearly pure pastures of P. maximum containing only a few weeds or other contaminating grasses.
Some species of Panicum, including Panicum coloratum 3 and Panicum virgatum, 14 cause photosensitization in horses as a result of the presence of steroidal lithogenic saponins. However, it is suggested 14 that photosensitization in horses associated with P. virgatum may be caused by another plant toxin. The main lesions in hepatogenous photosensitization caused by Panicum spp. are scattered necrotic hepatocytes and birefringent crystals in bile ducts, hepatocytes, and Kupffer cells. 2 Nephrosis with presence of crystals in the renal tubules, necrosis of adrenal glands, and necrosis of cardiac muscles are also reported. 2 In the cases of colic caused by the ingestion of P. maximum, the absence of photosensitization, absence of crystals in the liver, presence of severe lesions in the digestive tract, and negative test results for diosgenin- and yamogenin-based saponins in samples of Panicum spp. indicate that the disease is not caused by these substances.
High oxalate concentrations are frequently noted in tropical grasses. Oxalate values of 2.21% are reported 12 in P. maximum cv. Colonião. Panicum maximum cv. Tanzânia contained 1.4% oxalates (ranging from 0.7% to 2.2% oxalates), and P. maximum cv. Massai contained 1% oxalates (ranging from 0.6% to 1.4% oxalates; Ribeiro CB, Medeiros SR, Macedo MCM, et al.: 2006, Teores de oxalato nas principais forrageiras utilizadas no Brasil e sua implicação na toxidez em pastejo [Oxalate concentrations in grasses used in Brazil and their implication in toxicity]. 2nd Jornada Científica da Embrapa Gado de Corte, October 18–20, Campo Grande, Mato Grosso do Sul, Brazil. Available at http://www.cnpgc.embrapa.br/eventos/2006/jornada2/TrabalhosProntos_Final_PDF/MS/046ribeirocb.pdf. Accessed on June 8, 2009. In Portuguese). Oxalate concentrations >2% are also reported in Brachiariaspp., Setaria spp., Digitaria decumbens, and Stylosanthes spp. (Ribeiro CB et al.: 2006, [Oxalate concentrations in grasses]). 12 Most of those oxalate concentrations are similar, or even higher, than concentrations in pastures containing Panicum spp. that cause the development of colic in equidae (1.36–2.03% oxalates). However, the results of the present study demonstrate that pastures containing Panicum spp. in which the disease did not occur had lower concentrations of oxalates (0.28–1.39%) than did pastures associated with the development of colic. This observation is probably explained by the fact that more mature pastures contained lower oxalate concentrations than did pastures with newly sprouting plants (Ribeiro CB et al.: 2006, [Oxalate concentrations in grasses]). This indicates that oxalates are not the cause of colic in equidae in P. maximum pastures in northern Brazil. Oxalate concentrations of >0.5% can cause secondary nutritional hyperparathyroidism in horses. 10 This disease is frequently observed in Brazilian tropical pastures but was not observed in the pastures of the farms visited during the present study. Furthermore, oxalate concentrations of 4–8% are considered toxic for cattle. 6
Colic caused by different P. maximum cultivars is probably due to other toxins that are produced by the plant in special environmental conditions found in the Amazonia or due to the presence of another toxigenic agent in the plant or in the environment. This toxin (or toxins) acts on the digestive system, causing severe gastroenteritis with mucosal ulceration. Nephrosis is also an important effect of the disease, as was observed in the histologic sections of kidney from 5 affected animals and increased serum urea nitrogen and creatinine concentrations in some animals. The gastrointestinal and esophageal ulcerations observed grossly could have resulted from uremia. In one animal, severe urinary bladder lesions were probably associated with the elimination of a toxin by the kidney. The GGT and AST activities were generally within reference intervals, and the mild hepatic changes observed microscopically could be associated with bacterial invasion due to gastroenteric lesions.
Panicum maximum toxicosis in horses is similar in some aspects to cantharidin toxicosis. In cantharidin toxicosis, the toxin is present in blister beetles (Epicauta spp.) found in hay or pasture grasses, especially alfalfa. Ingestion of the blister beetles and absorption of the toxin cause colic, gastroenteritis, frequent urination, cystitis, profuse salivation, renal dysfunction, and death. 13 Nevertheless, Epicauta spp. were not observed during inspection of the pastures.
One important aspect of the disease outbreaks reported in the present study is the fact that they occurred only during the rainy period, mainly in new growth pastures. This feature was clearly demonstrated in disease outbreaks 5 and 6, in which clinical signs appeared in animals placed on pastures with new growth of grass that had been rested for 30 days after being grazed by cattle. Another important fact in the epidemiology of the disease is that cattle and other ruminants (buffalo, sheep, and goats) found in the region were not affected. In addition, the disease described herein has not been reported in other Brazilian regions in which P. maximum is a common pasture plant for horses, cattle, and sheep. The identification of the toxin-causing disease will be important to determining appropriate methods of prevention and control.
Another possibility is that colic caused by P. maximum is due to a high content of carbohydrates that are rapidly hydrolyzed and fermented during the growing stage of the plant. Such carbohydrates could be fermented by microorganisms within the horse's digestive tract, resulting in rapid gas buildup and altered bowel motility. 5 This explains why colic is relatively common when horses are suddenly given unrestricted access to lush spring pastures, especially when the pastures contain a significant amount of clover (King C: 2000, Preventing spring colic in horses. Flying Changes. Available at http://www.flyingchanges.com/htmls/2000/mar00colic.html. Accessed on June 8, 2009). The carbohydrate load of green grass can also change the bacterial balance in the intestines, leading to the release of lactic acid and endotoxins, 5 which could be responsible for the lesions observed in the intestine and other organs.
As a result of the lack of detailed information about the cause of the disease reported herein, the only way to prevent its occurrence is to avoid allowing equidae to graze pastures with new growth of P. maximum. Unfortunately, other common tropical grasses, such as different species of Brachiaria, are not recommended as grazing plants for equidae because they are not palatable (B. decumbens and B. brizantha) and are occasionally toxic (all species) because of their high saponin and oxalate content. The main alternative is the use of Cynodon spp. in pastures designated for grazing; another option is only using pastures containing mature P. maximum. If colic occurs in equidae kept in pastures with Panicum spp., the animals should be removed from the pastures immediately at the first signs of colic.
Acknowledgements. This project was financially support by the National Council of Scientific and Technological Development/MCT, program of the Institutos do Milěnio, grant 420012/2005-2.
