Abstract
A disease known as water belly (barriga d'água), characterized by chronic progressive ascites, affects sheep in the semiarid region of northeastern Brazil. The objectives of this investigation were to study the disease and to determine its cause. Only sheep grazing for long periods in pastures where Tephrosia cinerea represents 80% to 100% of the available forage are affected. Most animals die after a clinical manifestation period of some weeks or months, but others recover when they are withdrawn from the pastures or after the first rains. At necropsy, large amounts of liquid were found in the abdominal cavity, and the liver was hard, with an irregular surface. On histology examination, the main liver lesion was chronic periportal and subcapsular fibrosis with bridging. The disease was produced experimentally in 1 sheep by the administration of large amounts of T. cinerea for 232 days. Another sheep, recovered from the spontaneous disease, had clinical signs after the ingestion of large amounts of the plant for 40 days. Seeds and leaves of the plant were examined by gas chromatography-mass spectrometry for the presence of pyrrolizidine alkaloids with negative results. It is concluded that the disease is caused by the ingestion of T. cinerea.
Tephrosia cinerea Pers. (Fig. 1) belongs to the family Leguminosae, subfamily Faboideae. It is an undershrub of variable aspect (prostrate, decumbent, or erect), up to 60 cm tall, and ramose; the stem, branches, and leaves usually have gray hairs; the leaves are odd-pinnate, with 7–13 linear, oblong leaflets; flowers are violet to slightly purple, 12- to 15-mm long, and disposed in fascicles; pods are straight to slightly curved, 3.5- to 5.0-cm long and 4- to 5-mm wide, covered with short hairs, and containing 5–9 seeds. 1,4 The plant is found in all Brazilian states in dry and sandy soils. Because of its resistance to drought, it is a particularly common weed in the Brazilian semiarid region where it stays green for most of the year. It is a troublesome weed and becomes particularly widespread where the soil has been denuded of grass as a result of bad farming practices, such as continuous cultivation and/or overgrazing. It is relatively unpalatable, but sheep are forced to graze the plant during dry periods when it becomes the only available forage. Farmers from the states of Rio Grande do Norte and Ceará, northeastern Brazil, in areas severely invaded by T. cinerea, claim that the ingestion of this plant causes a disease of sheep known as “barriga d'água” (water belly). The objectives of this investigation were to study the disease and to determine its cause.
Farms where the disease occurs were visited to examine affected animals and to inspect the pastures. The disease occurs in the semiarid area of northeastern Brazil in the municipalities of Acari, Cruzeta, Jardim do Seridó, Ouro Branco, Parelhas, and São José do Seridó in the state of Rio Grande do Norte, and in the municipality of Quixeramobin in the state of Ceará. Large amounts of T. cinerea representing 80% to 100% of the vegetation were observed in all paddocks where the disease occurred. The paddocks had been used continually for many years to cultivate beans, corn, or cassava, and the invasion of the paddocks by the plant increased continuously. Farmers reported that goats and cattle were not affected, and they did not ingest the plant. The disease was never observed on farms where the plant did not exist.

Tephrosia cinerea. Municipality of Caicó, Rio Grande do Norte. Insert. T. cinerea flower, leaves, and pods.
During visits to the farms, it was observed that, after some months of ingestion, sheep present with a disease characterized by chronic progressive ascites, called “barriga d'água” (water belly) by the farmers. When the dry season is advancing, new cases of the disease are observed affecting haired sheep of both sexes, different breeds, and all ages, but generally older than 1 year. Morbidity varies from 10% to 70%, and the case fatality rate is 50% to 100%. The amount of liquid in the abdominal cavity increases (Fig. 2), and anorexia, depression, dyspnea, and intolerance to exercise are observed. If the flock is moved to other areas without the plant at the start of clinical signs, then most affected sheep recover and new cases are not observed.
Two crossbreed 2-year-old sheep with clinical signs of the disease were euthanized and necropsied. Sheep no. 1 was a male and sheep no. 2 a female. Samples of different tissues of the thoracic and abdominal cavities, and the central nervous system were fixed in 10% neutral formalin, embedded in paraffin, sectioned at 6 μm, and stained by hematoxylin and eosin. Sections of the liver were stained by Masson trichromic for fibrous tissue, and sections of liver, kidney, and spleen were stained by Congo red for amyloid.
At necropsy, sheep no. 1 had severe ascites, with more than 15 liters of clear liquid in the abdominal cavity. Hydrothorax and hydropericardium were also observed. Sheep no. 2 also had ascites but with less liquid than sheep no. 1. The liver of both sheep had a whitish irregular nodular surface (Fig. 3A) with the presence of small nodules, up to 1 mm. The cut surface of the liver had a hard consistency and had whitish areas mixed with normal colored areas. The gallbladder was slightly enlarged, with discrete edema in the wall and dilated lymphatic vessels in the surface.
On histology, lesions in sheep nos. 1 and 2 were chronic periportal and subcapsular fibrosis with bridging (Figs. 3B, 3C). Moderate proliferation of bile-duct cells was observed in the portal triads, rarely extending to other regions of the lobe. In the areas of fibrosis and in the wall of the gall bladder, lymphatic vessels, and veins were markedly dilated. Mild infiltration of lymphocytes, plasma cells, and undifferentiated mononuclear cells was observed in the periportal areas. Most hepatocytes were enlarged, with fine vacuolation of the cytoplasm. This lesion was more severe in the subcapsular hepatocytes (Fig. 3C). Some hepatocytes of the periportal and subcapsular areas had individual necrosis, with marked eosinophilic cytoplasm and pyknotic nuclei. Mild bile stasis was also observed. The Glisson capsule was thickened by proliferation of connective tissue, with projections of connective tissue into the liver parenchyma (Fig. 3C). Mild, multifocal, random accumulations of neutrophils were also observed in sheep no. 2.
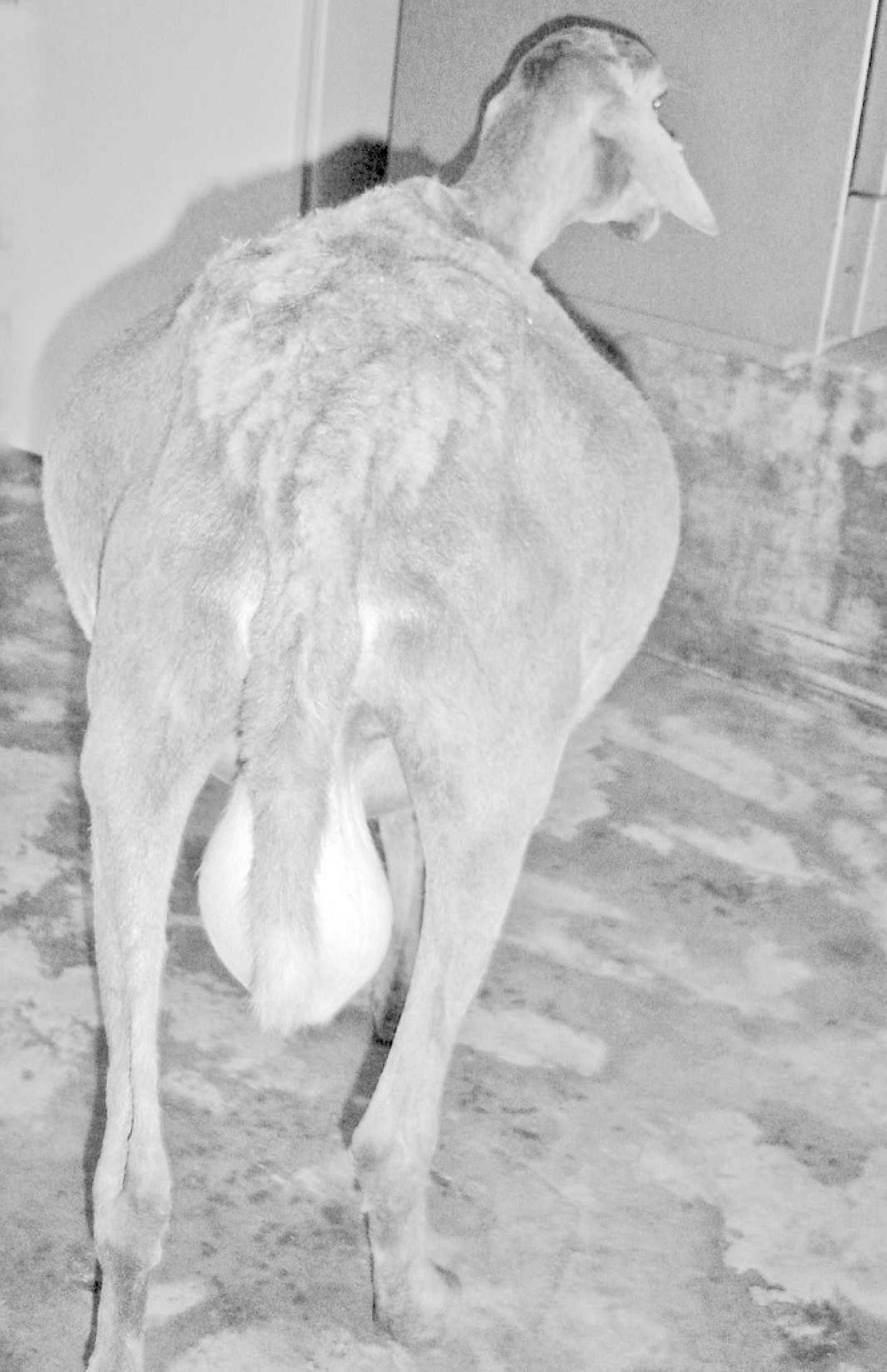
Sheep intoxicated by T. cinerea with abdominal enlargement because of severe ascites.
The disease was reproduced in 2 experimental sheep. Sheep no. 3, a 1-year old, Santa Inês crossbred ewe, weighing 20 kg, was from the Federal University of Campina Grande experimental farm where T. cinerea does not exist. Because it did not ingest the plant spontaneously, ground seeds of the plant were initially added to commercial ground sheep ration, in a ratio of 1 to 4, and were given in an amount equivalent to 0.5% body weight. After 1 week, the amount of ration that contained 20% of seeds was increased to 1% body weight. During the experimental period, the plant was administered in different concentrations and in different forms (Table 1). The animal also received Cynodon dactylon (Tifton grass) hay ad libitum, except for the last 90 days when this hay was substituted by a hay of T. cinerea that contained leaves, stems, seeds, and flowers of the plant. At the end of the experiment, the sheep had ingested the plant for 232 days. It ingested an amount equivalent to 47.7% of live weight of seeds and 63% of live weight of pods with seeds. It also received hay of T. cinerea during 90 days ad libitum. With the ingestion of hay between 0.5% and 1% body weight, the consumption of T. cinerea hay was estimated to be 45% to 90% of the live weight.
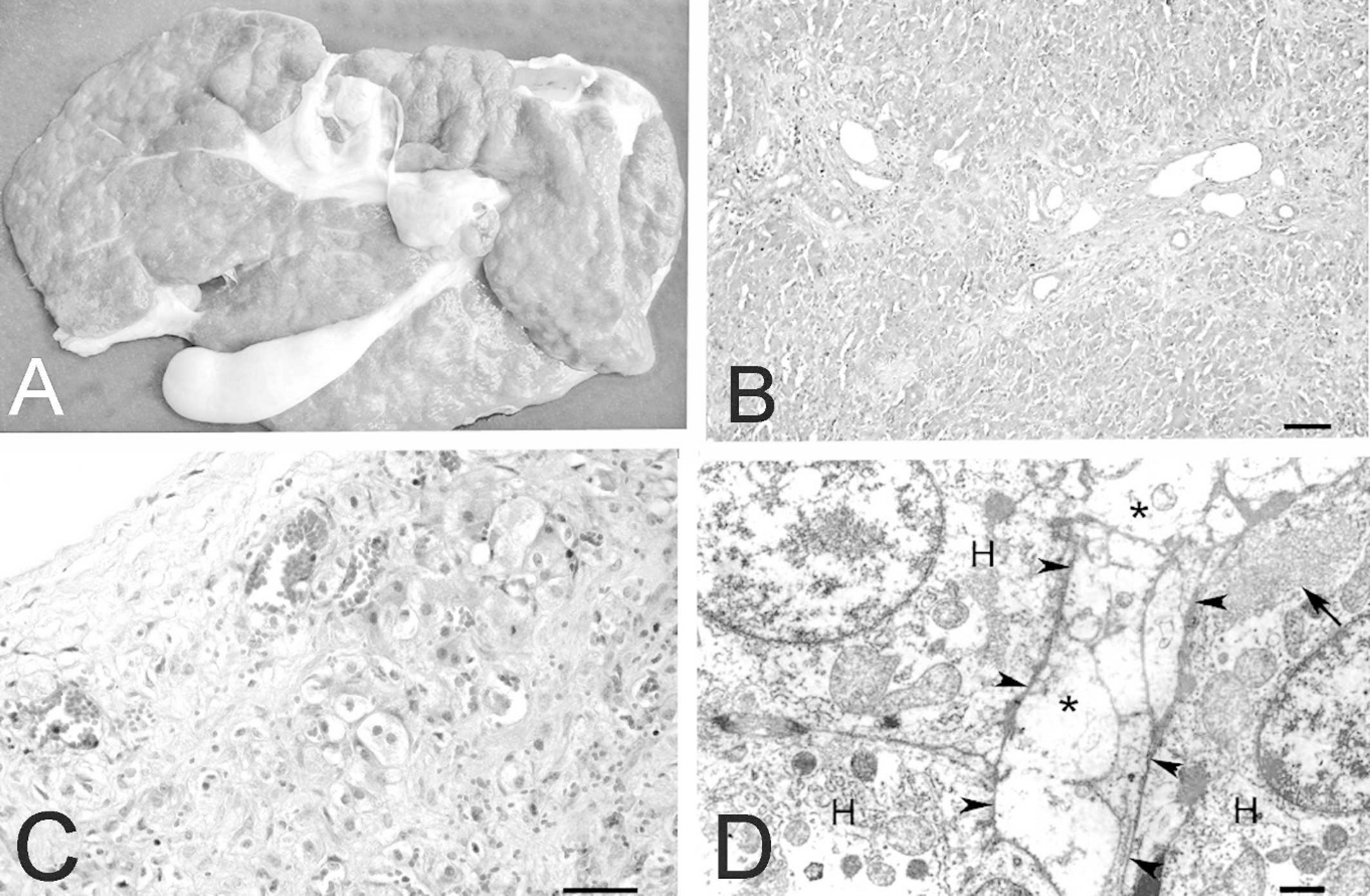
Liver of sheep intoxicated by T. cinerea.
Formulation and timing of administration of Tephrosia cinerea in sheep no. 3.

Percentage mixed in the concentrate ration.
Amount in percentage of live weight.
Hay made with the whole aerial plant including seeds.
Another sheep, a crossbred Santa Inês ram (no. 4), weighing 27 kg, was from a farm where the disease occurred. The farmer stated that this sheep had recovered from the disease after having been taken from a paddock with T. cinerea. Seeds and pods of the plant were mixed at 50% in a concentrate commercial ration, which was given daily in an amount equivalent to 1% of its live weight, for 40 days, and, simultaneously, it received T. cinerea hay ad libitum. Both sheep were euthanized and necropsied at the end of the experiment. Two sheep (nos. 5 and 6), used as controls, ingested commercial ration in a daily amount equivalent to 1 % of their live weight and C. dactylon hay ad libitum, for 250 days.
Experimental sheep no. 3 had anorexia, depression, and mild ascites, 2 to 3 weeks before the end of the experiment, on day 232. Experimental sheep no. 4 had anorexia, ascites, dyspnea, and exercise intolerance, starting 10 to 20 days before euthanasia. A few days after the start of the experiment, sheep no. 3 had a purulent lymphadenitis in the right prescapular lymph node. This lesion was surgically drained, and the animal was treated with tetracycline and recovered. The lesion recurred twice, each time producing purulent exudate. Both times, the sheep was treated in the same way as before, but the lymph node stayed enlarged, with hard consistency and sometimes draining purulent exudate. Staphylococcus aureus and Corynebacterium pseudotuberculosis were isolated from the lesion.
Serum activity of AST and GGT, and values of TP and AL in sheep nos. 3 and 4 intoxicated experimentally by Tephrosia cinerea and 2 control sheep.∗
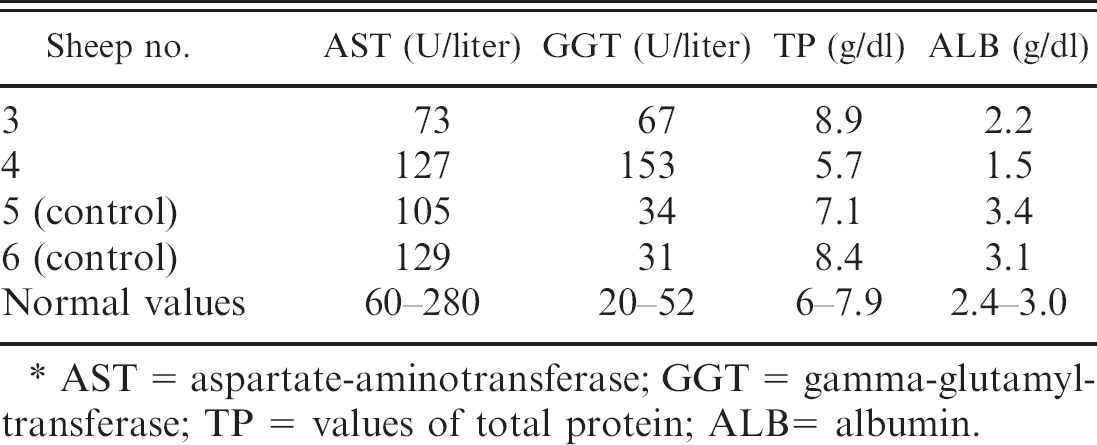
AST = aspartate-aminotransferase; GGT = gamma-glutamyltransferase; TP = values of total protein; ALB= albumin.
Blood samples obtained immediately before euthanasia in sheep nos. 3 and 4 were sent to a private laboratory for determination of serum activity of aspartate aminotrans-ferase and gamma glutamyltransferase, and serum concentrations of total proteins and albumin, with the methods of Schmid and von Forstner. 5 Samples from sheep nos. 5 and 6 were used as controls. Results of those examinations are presented in table 2.
Experimental sheep no. 3 had ascites and a whitish hard liver with an irregular surface. Whitish areas mixed with normal colored areas were observed at the cut surface of the liver. The wall of the gallbladder had mild edema, with dilated lymphatic vessels. A chronic fistulated abscess surrounded by fibrous tissue was observed in the right prescapular lymph node, which measured 6 cm in diameter. Histologic lesions were chronic periportal and subcapsular fibrosis with bridging. Dilated veins and lymphatic vessels; thickening of the Glisson capsule; bile-duct proliferation; vacuolation; and, occasionally, individual necrosis of hepatocytes and mild bile stasis were also observed. An eosinophilic homogeneous hyaline substance with morphologic characteristics of amyloid was deposited between hepatocytes. A similar substance was observed in the kidney deposited extracellularly in glomeruli and between tubules in the medullar region. Rare eosinophilic casts and some interstitial infiltration of mononuclear cells were also observed. An eosinophilic homogeneous substance was also observed in the wall of the small arteries of the spleen. In all of these tissues, the substance was strongly positive to Congo red. Macroscopic and histologic lesions observed in sheep no. 4 were similar to those observed in sheep nos. 1 and 2. Multifocal centrolobular or midzonal hemorrhages were also observed.
For electron microscopy, samples of the liver from sheep nos. 3 and 4 were fixed, immediately after euthanasia, in 2% glutaraldehyde with 2% paraformaldehyde, in 0.4 M cacodylate buffer (pH 7.4), post-fixed in 1% osmium tetroxide buffered in 0.4 M sodium cacodylate (pH 7.4), and embedded in Epon 812. Semithin sections were stained with methylene blue. Ultrathin sections were stained with lead citrate and uranyl acetate and examined with an EM 109 Zeiss transmission electron microscope at 80 kV. On ultrastructure examination, severe swelling of the sinusoidal endothelial cells, obstructing the sinusoidal lumen (Fig. 3D) with erythrodiapedesis to the Disse space was observed in both sheep examined. The hepatocytes were degenerated or necrotic with conspicuous hyperplasia of the smooth endoplasmic reticulum associated with glycogen granules. The mitochondria had a granular more electrodense matrix. Proliferation of collagen fibers was present in the space between hepatocytes and in the Disse space. In sheep no. 3, large amounts of fibrillar material characteristic of amyloid were observed in the interstitium.
Samples of seeds and leaves of the plant were examined by gas chromatography-mass spectrometry (GC-MS) for the presence of pyrrolizidine alkaloids 3 with negative results. The nonprotein amino acids, homoarginine, and gamma-hydroxyhomoarginine, which have been reported to occur in T. cinerea and many other Tephrosia species 2 were also not found, probably because they would not be detected under the extraction procedure and GC-MS conditions used for pyrrolizidine alkaloids. However, there are no reports of these nonprotein amino acids causing liver damage.
The occurrence of the disease exclusively in paddocks where T. cinerea represented 80% to 100% of the vegetation and its absence in paddocks where the plant does not exist, the experimental production of the intoxication in 1 sheep, and the induction of clinical signs in a sheep previously recovered from the spontaneous intoxication suggest that the disease is caused by the ingestion of T. cinerea. Another species of Tephrosia, Tephrosia apollinea, was reported as experimentally toxic for goats, but clinical signs and pathology, affecting the nervous and digestive systems, 6 are different than those observed in T. cinerea poisoning.
Despite some similarities between this disease and the intoxication by pyrrolizidine alkaloids (PA), results of the GC-MS analysis indicate that it is unlikely that the hepatotoxicity is caused by pyrrolizidine alkaloids, and there are no literature reports of this type of compound occurring in the genus Tephrosia. Furthermore, clinical signs of ascites without other signs of liver disease, observed in the spontaneous and experimental intoxication by T. cinerea, are different from PA intoxication and other plant intoxications causing liver fibrosis. These intoxications had other signs of liver disease, including jaundice, photosensitization, and hepatic encephalopathy.
The large amount of the plant necessary to cause the disease agrees with the field information that intoxication only occurs in areas where the plant is dominant and after a long grazing period. These conditions occur in the Brazilian semiarid areas during the long dry periods. The Brazilian semiarid region consists of nearly 1 million km2, and T. cinerea is found in almost the whole area. It proliferates in degraded areas under intensive agriculture and becomes dominant after a few years. To prevent the intoxication, it is important to eradicate the plant immediately on its detection in the paddocks, because control is very difficult when it becomes dominant in the pasture. The only way to minimize mortality in areas invaded by the plant is to move the flock from the areas infested by T. cinerea immediately after the observation of the first clinical signs.
Acknowledgements. This work was financially supported by the Programa de Apoio a Núcleos de Excelência (PRONEX), grant 001/04, CNPq, FAPESQ, MCT, and by Institutos do Milénio, grant 420012/2005–2), CNPq.
