Abstract
Albendazole is a widely used anthelmintic drug that is labeled for the treatment of specific nematodes and flukes in ruminants. Albendazole is approved for the treatment of liver flukes in goats (10 mg/kg PO for a single dose), but is commonly used extra-label in situations in which parasite resistance is an issue. Albendazole toxicosis has been reported in pigeons, doves, alpacas, humans, dogs, and cats. Here we report an adverse event in a 6-mo-old goat associated with extra-label use of albendazole (35.7 mg/kg PO daily for 3 d). Clinicopathologic findings included severe diarrhea and death, with small intestinal crypt necrosis and dysplasia, and severe bone marrow hypoplasia. Microbial and molecular testing and transmission electron microscopy ruled out infectious organisms. The described pathologic changes are similar to those reported in other species that have experienced toxicosis associated with albendazole. To our knowledge, bone marrow and intestinal lesions associated with albendazole use in the goat have not been reported previously. Veterinarians should be aware of potential adverse events and toxicoses associated with anthelmintic drugs, especially as parasite resistance increases, and extra-label usage, and the use of such drugs without veterinary supervision, becomes more common.
Keywords
Albendazole is a broad-spectrum anthelmintic benzimidazole that is used widely in both human and veterinary medicine. Albendazole is used to treat lung and gastrointestinal nematodes and liver flukes in cattle and sheep in a single dose orally. 12 Albendazole is also approved for the treatment of adult liver flukes in goats at 10 mg/kg as a single dose orally (https://animaldrugsatfda.fda.gov/adafda/app/search/public/document/downloadFoi/11659). Nematode resistance, particularly by gastrointestinal nematodes, is a significant problem for small ruminant owners and veterinarians. 14 Parasite resistance in small ruminants has resulted in the extra-label use of several anthelmintic drugs, such as albendazole. 14 Generally, benzimidazoles are thought to have low toxicity to mammals and are regarded as a safe class of drugs. 12 Bone marrow hypoplasia and small intestinal crypt necrosis and dysplasia associated with albendazole use have not been reported in the goat, but similar lesions have been reported in other species after the use of albendazole.8–10 We describe here the pathologic findings associated with presumed albendazole toxicosis in a goat that was treated in an extra-label manner.
A 6-mo-old, 31-kg, male Boer goat was found dead on a premises that had a history of parasite resistance and heavy burden of intestinal helminths. Two goat kids had died in the current year, and the remaining goat kids and some adults appeared lethargic and had pale mucosae. Because of perceived worsening clinical signs observed in the goat kids, the owner initiated a combination anthelmintic treatment that included moxidectin (Quest Plus Gel, 20 mg/mL; Zoetis) and albendazole (Valbazen, 113.6 mg/mL; Zoetis) without consulting a veterinarian. This goat received a single dose of moxidectin (0.44 mg/kg) and albendazole (35.7 mg/kg) PO on day 1. Additional doses of albendazole (35.7 mg/kg) were administered consecutively on days 2 and 3 (single dose/day) for a total cumulative dose of ~3,320 mg of albendazole. This goat continued to do poorly and died on day 8 (5 d after receiving the last dose of albendazole). The goat was submitted to the Iowa State University Veterinary Diagnostic Laboratory (ISU-VDL; Ames, IA, USA) for postmortem examination. A second goat kid received the same treatment and died on day 11 but was not submitted for postmortem examination. The submitting veterinarian’s primary antemortem differential was severe Haemonchus contortus infection and associated anemia given that other animals on the premises had been diagnosed with hemonchosis previously.
At postmortem examination, the carcass had adequate-to-thin body condition with moderate autolysis. The mucous membranes were moderately pale, and there was marked staining of the perineum and legs with brown, watery diarrheic feces. A standard small ruminant postmortem examination was completed. The liver and kidneys were pale. The intestinal and colonic contents were liquid in consistency and dark brown. Parasites were not observed on gross examination. No other significant gross pathologic findings were observed. Differentials after gross postmortem examination included bacterial enterocolitis (Clostridium perfringens, Salmonella spp., Escherichia coli) and/or intestinal parasitism (Eimeria spp., Cryptosporidium spp., Haemonchus contortus).
Further testing included histopathology performed on femoral bone marrow, brain, esophagus, trachea, heart, lung, liver, spleen, adrenal gland, kidney, rumen, abomasum, small intestine, and large intestine fixed in 10% neutral-buffered formalin; McMaster fecal flotation; bacterial culture (simple aerobic and tetrathionate enrichment); multiplex real-time PCR (rtPCR) for select bovine enteric pathogens (bovine coronavirus [BCV; Betacoronavirus 1], bovine viral diarrhea virus [BVDV; Pestivirus], Cryptosporidium parvum, E. coli K99, group A bovine rotavirus, Salmonella spp.); and transmission electron microscopy (TEM) performed on fecal material. Routine processing and staining of fixed tissues, bacterial culture, and rtPCR were performed at the ISU-VDL according to standard operating procedures (SOPs). TEM was performed in the Diagnostic Virology Laboratory at the National Veterinary Services Laboratories (NVSL) in Ames, IA, USA. McMaster fecal flotation identified ~750 Strongyle spp. ova/g of feces and low numbers of Eimeria sp., Trichuris sp., and Moniezia sp. ova. According to the submitting veterinarian, this parasite burden was significantly lower than that observed in McMaster fecal flotations performed previously on other goats on the premises. Routine bacterial culture identified C. perfringens type A, which was interpreted as nonpathogenic. All other tests were negative, and no viral particles were detected by TEM performed at the NVSL.
Histologic examination identified lesions in the bone marrow and intestines. A low-power (100× or 3.79-mm2 area) examination of the bone marrow identified hypocellularity (15% cellular, 85% adipose) with an expansion of the hematopoietic elements by edema and extravasated mature erythrocytes (Fig. 1). A high-power (400× or 0.237-mm2 area) examination identified marked trilineage hypoplasia and a paucity of differentiated cells (Fig. 2). Microscopic examination of the small intestines identified diffuse villus blunting and fusion with marked intestinal crypt necrosis and stromal collapse (Figs. 3, 4). The remaining crypt epithelial cells were attenuated, numbers of mitotic figures were increased, and bizarre mitotic figures were present (Fig. 4), which was interpreted as epithelial dysplasia. Significant microscopic pathologic findings were not observed in the other tissues examined.
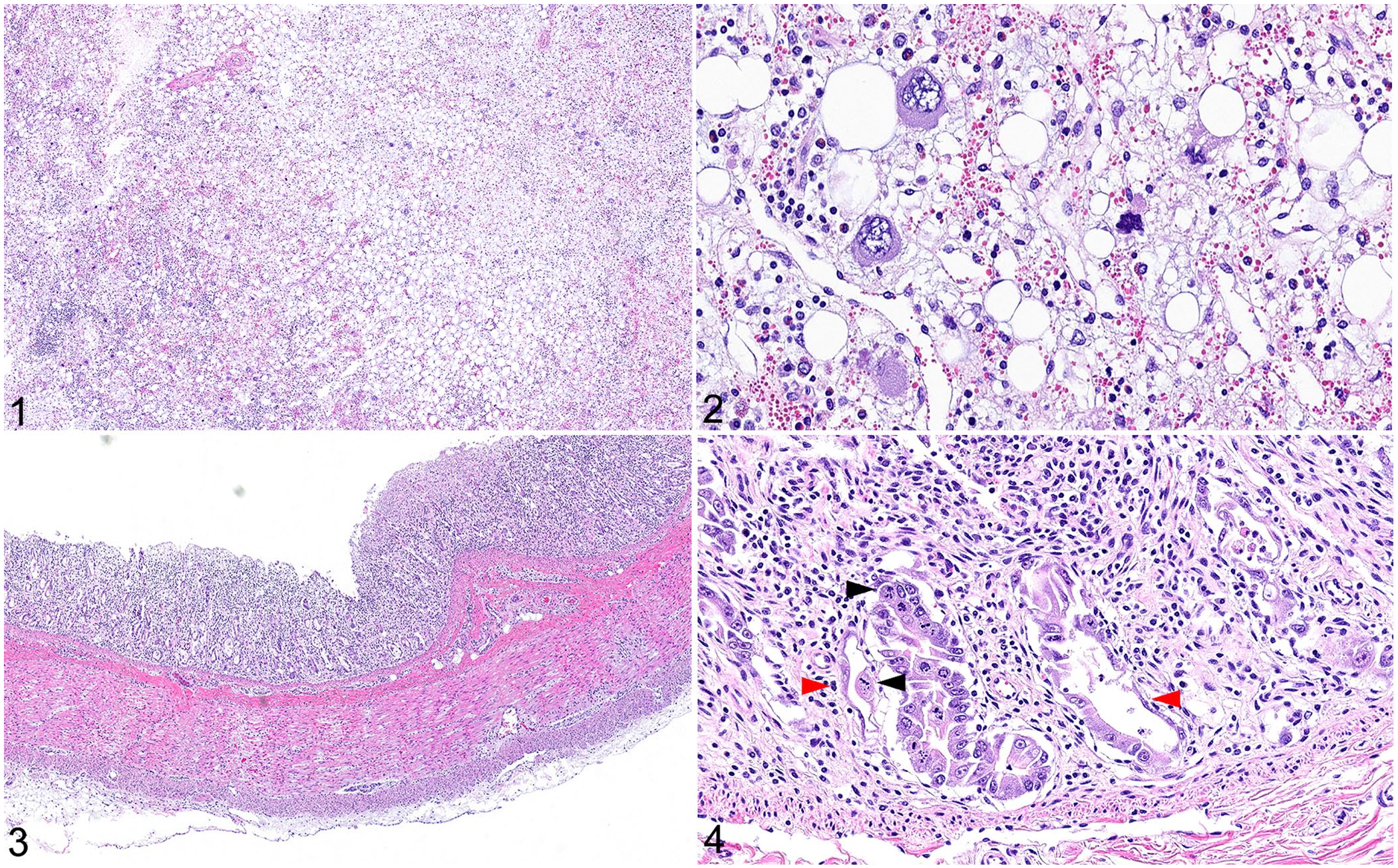
Microscopic pathologic changes observed in a juvenile Boer goat with presumptive albendazole toxicosis.
Differential diagnoses following ancillary testing included a systemic viral infection (coronavirus, parvovirus, pestivirus, rotavirus) or damage resulting from a radiomimetic agent. Bacterial pathogens were considered a less likely cause based on the lack of isolation of a significant enteric bacterial pathogen and the lack of necrosuppurative histologic lesions. Parasite ova were detected via fecal flotation and may, in part, have been the cause of the reported diarrhea. Intestinal parasitism is a major cause of diarrhea in young goats. 19 Intestinal parasitism is commonly associated with various degrees of villus atrophy, crypt hyperplasia, and mixed inflammatory response. 19 The latter features were not observed in our case, suggesting that the contribution of parasitism to the clinical signs and observed lesions may be minimal. In addition, intralesional parasites were not observed microscopically.
Several viral pathogens have been reported to cause enteritis in goats. 11 To our knowledge, there are no reported systemic viruses in the goat that cause selective destruction of intestinal crypt epithelium with concurrent bone marrow depletion. In cattle, BVDV can cause ulcerative enterocolitis and lymphoid and bone marrow depletion; however, the ability of BVDV to cause disease in goats is unclear, and BVDV rtPCR was negative in our case.15,19 BCV can cause ulcerative enteritis in goats, but bone marrow depletion has not been reported, and BCV rtPCR was negative. 2 In dogs and cats, parvovirus can cause crypt necrosis, villus blunting and fusion, and bone marrow depletion similar to the lesions in our case. 19 Parvoviral infection has yet to be reported in goats, and TEM performed on the feces did not identify any viral particles. Rotavirus can result in villus blunting and fusion, but crypt necrosis and bone marrow depletion are not a feature of rotaviral infection; rotavirus was also not detected by rtPCR. 6 Microscopic examination and ancillary testing effectively ruled out the most common viral causes of crypt necrosis and bone marrow hypoplasia.
The concurrent lesions in crypt epithelial cells and hematopoietic cells are suggestive of a radiomimetic agent, which attack mitotically active and rapidly dividing cells. 7 Radiomimetic agents can be infectious agents (e.g., parvovirus), toxic compounds (certain chemotherapeutic drugs), or radiation. We ruled out infectious agents in our case. This goat had been treated with the anthelmintic drugs moxidectin and albendazole. Moxidectin is not reported to be a radiomimetic agent. Albendazole is a benzimidazole chemotherapeutic agent that works by binding to and stabilizing microtubules and effectively stopping cellular division and, thus, acts as a radiomimetic agent. 12 Considering the extra-label use of albendazole, we suspected albendazole overdose.
Liquid chromatography–tandem mass spectrometry (LC-MS/MS) was performed on fresh liver according to an SOP at the ISU-VDL Analytical Chemistry Laboratory (Table 1; Suppl. material). After oral administration, albendazole undergoes first-pass oxidation to produce albendazole sulfoxide, which is converted to albendazole sulfone and albendazole-2-aminosulfone.4,5 This process is mediated by cytochrome P450 and flavin-containing monooxygenases within hepatic microsomes.1,4,5 Albendazole sulfoxide is considered to be the active metabolite and is thought to be largely responsible for the therapeutic effect.1,5 Previously, albendazole sulfoxide was thought to be responsible for the toxicity, as observed in studies utilizing zebrafish embryotoxicity testing. 4 However, in vitro tests have demonstrated that the parent compound, albendazole, is more toxic to cultured mammalian hepatocytes than the metabolite, and the metabolism of albendazole leads to detoxification. 16
Quantification of albendazole and associated metabolites in the liver of a goat using liquid chromatography–tandem mass spectrometry.

The hepatic concentration of albendazole in our goat, ~5 d after the last dose of albendazole, was 17,800 ng/g (Table 1). The albendazole freedom of information (FOI) summary supplemental to NADA 110-048 demonstrated mean liver concentrations of 138 ng/g in treated goats 5 d after PO administration of albendazole (11.36 mg/mL) at 10 mg/kg (https://animaldrugsatfda.fda.gov/adafda/app/search/public/document/downloadFoi/11659). The FOI summary and analytical chemistry results suggest that our goat had been administrated albendazole at a dose higher than the FDA-approved dose of 10 mg/kg. The LC-MS/MS results do not definitively prove albendazole toxicosis, given that extensive toxicologic studies in the goat have not been carried out, and there is no reported threshold for the development of toxicosis in goats. A 10 mg/kg PO dose is approved by the FDA for use in goats; however, this dose was extrapolated from studies in cattle and sheep (https://animaldrugsatfda.fda.gov/adafda/app/search/public/document/downloadFoi/11659).
A review of the literature regarding albendazole toxicosis identified a species-specific sensitivity to the anthelmintic. Albendazole, similar to other benzimidazoles, is considered to be a relatively safe class of drug. Toxicologic and pharmaceutical studies have demonstrated high median lethal doses (LD50) for mice (500–1,250 mg/kg), rats (>3,000 mg/kg), and rabbits (1,320–2,400 mg/kg). When adverse events were observed in these trials, they included intestinal hemorrhage, excessive intestinal fluid, leukopenia, and/or reversible anemia. 4 In cattle, the maximum tolerated dose is 45–75 mg/kg, and death has been reported after administration of a 300 mg/kg dose (5–6 d post-administration). Doses >45 mg/kg result in lethargy, anorexia, diarrhea, and weight loss (https://animaldrugsatfda.fda.gov/adafda/app/search/public/document/downloadFoi/11657). In sheep, the maximum tolerated dose is 37.5 mg/kg. Doses >37.5 mg/kg resulted in lethargy, anorexia, diarrhea, and wool break (https://animaldrugsatfda.fda.gov/adafda/app/search/public/document/downloadFoi/495). Toxicosis in the dog has been observed with doses of 30–60 mg/kg administered PO daily for up to 6 mo. The toxic responses observed in the dog include bone marrow suppression, hepatomegaly, and testicular atrophy (https://animaldrugsatfda.fda.gov/adafda/app/search/public/document/downloadFoi/11657). Safety and toxicologic studies examining the effects of albendazole have not been carried out on goats.
Albendazole toxicosis and adverse events have been reported in other species. Pigeons and doves developed bone marrow hypoplasia and intestinal crypt necrosis when treated with albendazole at 50 mg/kg PO for 2–9 d. 10 Alpacas treated with doses as low as 19 mg/kg PO for consecutive days demonstrated bone marrow hypoplasia, crypt necrosis, and alopecia similar to that described in pigeons and doves.8,9 Adverse events have been reported in humans, dogs, and cats treated with albendazole. The adverse events include neutropenic leukopenia, thrombocytopenia, pancytopenia, gastrointestinal signs (dogs), hepatitis (humans), and/or alopecia (humans).13,17,18,20 The lesions in our case are similar to those described in pigeons, doves, and alpacas with albendazole toxicosis.
Diarrhea, small intestinal crypt necrosis and dysplasia, and bone marrow hypoplasia associated with albendazole use have not been reported in the goat. Neurologic clinical signs similar to tetanus have been reported with presumptive albendazole toxicosis (380 mg PO) observed in an Anglo-Nubian Alpine cross goat. 3 However, the final diagnosis in that case was based solely on a favorable response to therapy and signs occurring in conjunction with albendazole treatment. The authors did not state if other causes for neurologic clinical signs were ruled out in the goat.
Awareness of potential toxicosis and adverse events associated with anthelmintic drugs is vital for small ruminant veterinarians. Parasite resistance continues to be a major problem for the small ruminant industry. Many veterinarians and owners have resorted to extra-label drug use to maintain parasitized herds. It is important to note that adverse reactions and toxicosis associated with albendazole may mimic enteric pathogens, particularly those elicited by viruses. Obtaining a detailed history from the owner about anthelmintic treatment and developing a thorough diagnostic plan is vital to making a diagnosis of albendazole toxicosis.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387221121122 – Supplemental material for Enteropathy and bone marrow hypoplasia associated with presumptive albendazole toxicosis in a juvenile Boer goat
Supplemental material, sj-pdf-1-vdi-10.1177_10406387221121122 for Enteropathy and bone marrow hypoplasia associated with presumptive albendazole toxicosis in a juvenile Boer goat by Tyler A. Harm, Scott L. Radke, Laura E. Burns and Dwayne E. Schrunk in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We acknowledge the excellent work performed by the ISU-VDL analytical chemistry and pathology staff. Their fine work significantly aided in the production of this report. In addition, we acknowledge Dr. Erich Hodges for referring the case and providing excellent clinically relevant information.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
