Abstract
Analysis of hair to gain insight into the trace mineral status and exposure to toxic heavy metals of horses is attractive because hair is an easily accessible sample material. To investigate the potential value of hair analysis in horses for determination of trace mineral and heavy metal concentrations, we analyzed mane hair and liver samples from 62 horses presented for slaughter at a facility in the Netherlands that receives horses from all regions of the country. Hair samples were cleaned in warm water. After acid digestion of hair and liver specimens, we quantified, with inductively coupled plasma–mass spectrometry, arsenic, cadmium, chromium, cobalt, copper, iron, lead, manganese, molybdenum, nickel, selenium, vanadium, and zinc in the digests. Based on Pearson product moment correlations, we found no statistically significant correlations between concentrations of trace minerals in liver and hair, with the exception of a slight correlation for copper that was too weak to be of clinical relevance. Our results do not support the use of hair to determine trace mineral status and exposure to toxic heavy metals in the horse under field conditions.
Several trace minerals are essential nutrients, and maintenance of body stores within normal ranges is essential for health and performance in horses. 8 Toxic elements, particularly heavy metals, may also have significant health implications. The term heavy metal has an imprecise definition, but generally refers to metals with relatively high atomic weights. Lead (Pb), cadmium (Cd), and vanadium (V) clearly fall within the general definition, but here we specifically define toxic heavy metals as those metals that can be harmful to health and do not have clear or known nutritional value. For convenience, we also included the toxic metalloid, arsenic (As), in this group.
Analysis of hair to gain insight into the trace mineral status and exposure to toxic heavy metals of horses is attractive because hair is easily accessible. Hair can be sampled, stored, and shipped with relative ease and lower costs compared to alternative sample types such as blood or liver tissue. Additionally, hair is much less invasive to obtain, particularly when compared with liver biopsies. Commercial analytical services have emerged using hair specimens to assess trace mineral status and heavy metal exposure. However, the reliability of results in human hair has been questioned.2,12,15 It has been suggested that the unreliability of results is attributable to a lack of standardized methods and adequate RIs, 6 but this does not appear to be a complete explanation because it only addresses the variation that results from intra- and inter-laboratory variability, and not variation that results from external factors such as environmental variability. 12
Similar to analysis of human hair, there is an apparent lack of peer-reviewed publications supporting the reliable use of hair to determine the mineral status of horses under field conditions. However, some evidence exists indicating that mineral concentrations in horsehair are correlated with identifiable characteristics of horses, including nutritional status and physiologic conditions. For example, both positive and negative correlations of various elements with age, sex, and hair color have been demonstrated.1,9 Positive correlations were found between dietary intake and horsehair mineral concentrations in a study in which mineral exposure was well-controlled. 7 In another study, weak-positive correlations were found between hair concentrations and age for As, copper (Cu), and selenium (Se), but no correlations were demonstrated between trace mineral concentrations in horsehair and whole blood. 3
Although the studies referenced above did not demonstrate any direct clinical relevance of hair trace mineral analysis under field conditions, the studies were not designed to investigate that question specifically. Limited evidence for the use of horsehair under field conditions can be derived from a study using Se concentrations in horsehair for evaluation of Se exposure and risk of Se toxicosis. Se concentrations in segmented hair samples were associated with the timing of Se exposure. 5 Se concentrations in hair also appear to be correlated with dietary intake in other species, for example, in pigs. 10 In mule deer, hair Se was correlated with liver Se, but only when sampled shortly after growth of the winter coat. 14 However, aside from the study in horses on Se at toxicologically relevant exposure levels, evidence for the reliable use of trace mineral analysis in horsehair as a clinically relevant indication of mineral status in horses remains weak. Another important limitation of the available studies is that they failed to report correlations between hair values and liver values in horses. This is important because the liver is a major storage tissue for many trace minerals and is regarded as the preferred tissue for determining trace mineral status. 13
To investigate the potential value of hair mineral analysis with the purpose of determining trace mineral and toxic heavy metal status in horses, we compared mineral concentrations in samples of mane hair and corresponding liver samples from horses presented for slaughter in the Netherlands. The elements selected are part of a panel that is used for routine screening of liver tissue from a variety of livestock species in the Netherlands. It is based on relevance as nutritional elements, potential toxic effects, significant interactions, and indicators of environmental pollution. Therefore, not all elements in the panel are known to be of nutritional or toxicologic importance in horses (e.g., cobalt [Co], nickel [Ni], V).
Mane hair and liver samples were obtained from 62 horses presented for slaughter at a facility in the spring of 2021. This slaughter facility receives horses from all regions of the Netherlands. A minimum of 100 g of liver tissue was taken from the right liver lobe, sent to the laboratory in insulated containers cooled with ice packs, and stored at −20°C until analysis. Mane hair samples from all 62 horses were taken from an area of at least 1 cm in diameter behind the poll where mane was at least 10 cm in length. These hair samples were sent to the laboratory at room temperature and stored at −20°C until analysis. Coat color and sex of sampled horses were recorded.
Hair samples were cut at 5 cm (measured from the base), and 1 g of each cut sample was mixed with 40 mL of ultrapure water at 60°C and placed on a roller (Greiner) for 15 min. This was repeated 6 times. Hair samples were then dried overnight at 55°C in an oven (Heratherm OM60; Thermo Electron); ~200 mg of each cleaned sample was used for digestion in the next step. Liver samples were homogenized, and 5-g samples were dried at 103°C to determine the dry weight. Drying times for hair and liver were validated by confirming that sample weights were stable, and did not reduce further with longer drying times. We used ~1 g of liver tissue for digestion in the next step. Hair and liver samples were digested in trace mineral grade 65% nitric acid using a microwave oven (Discover SP microwave synthesizer; CEM Microwave Technology) to heat the samples to 200°C for 5 min, with a ramp time of 4 min, at a maximum pressure of 40 MPa. Digested samples were diluted with ultrapure water, and internal standards (germanium, scandium, and thallium; NIST traceable, Inorganic Ventures) were added. The internal standards were chosen because they are elements that do not generally occur in tissue samples at detectable concentrations.
Sample analysis was done with inductively coupled plasma–mass spectrometry (ICP-MS 7700x; Agilent Technologies), using a published tissue method. 4 Method validation (data not shown) included measurement uncertainty, robustness, and measurement range. Quantification was based on the ratios between the elements of interest and the internal standard with the closest matching mass, and calibration with external standards (NIST traceable; Inorganic Ventures). All analyses were done using helium collision to reduce interferences. Instrument stability was monitored by analyzing a control sample (freeze-dried and homogenized tissue derived from bovine liver) at the onset of analysis, and then once for every 20 analyzed samples. Hair and liver results were corrected for dry matter content. Hair and liver were analyzed for a panel of elements that are used for routine monitoring of trace minerals and heavy metals in animal tissues in the Netherlands, 4 including As, Cd, chromium (Cr), Co, Cu, iron (Fe), Pb, manganese (Mn), molybdenum (Mo), Ni, Se, V, and zinc (Zn). Not all elements in the panel are known to be of nutritional or toxicologic importance in horses (e.g., Co, Ni, V).
Statistical analyses included descriptive statistics, Pearson product moment correlations (PPMC), and one-way ANOVA on ranks, performed using SigmaPlot for Windows (v.11.0, build 11.2.0.5; Systat). Correlations between hair and liver trace mineral concentrations and differences were regarded as statistically significant at p ≤ 0.05. Trace mineral concentrations below the limits of quantification (LOQs) were included in the statistical analyses, and were assigned values of half the LOQ for each element.
Some elements were below their LOQs, including As and V in hair and liver, Cd and Co in hair, and Ni in liver (Table 1; Fig. 1). There were no statistically significant correlations between trace element concentrations in liver and hair, except for a slight positive correlation for Cu (PPMC coefficient = 0.265, p = 0.037; Table 2). Several statistically significant intra-tissue correlations occurred (8 correlations in liver; 9 correlations in hair), occasionally with highly significant p-values and high correlation coefficients (Tables 3, 4). ANOVA on ranks revealed no statistically significant differences in hair trace mineral concentrations based on hair color or sex.
Descriptive statistics for 62 paired hair and liver samples from horses in the Netherlands.
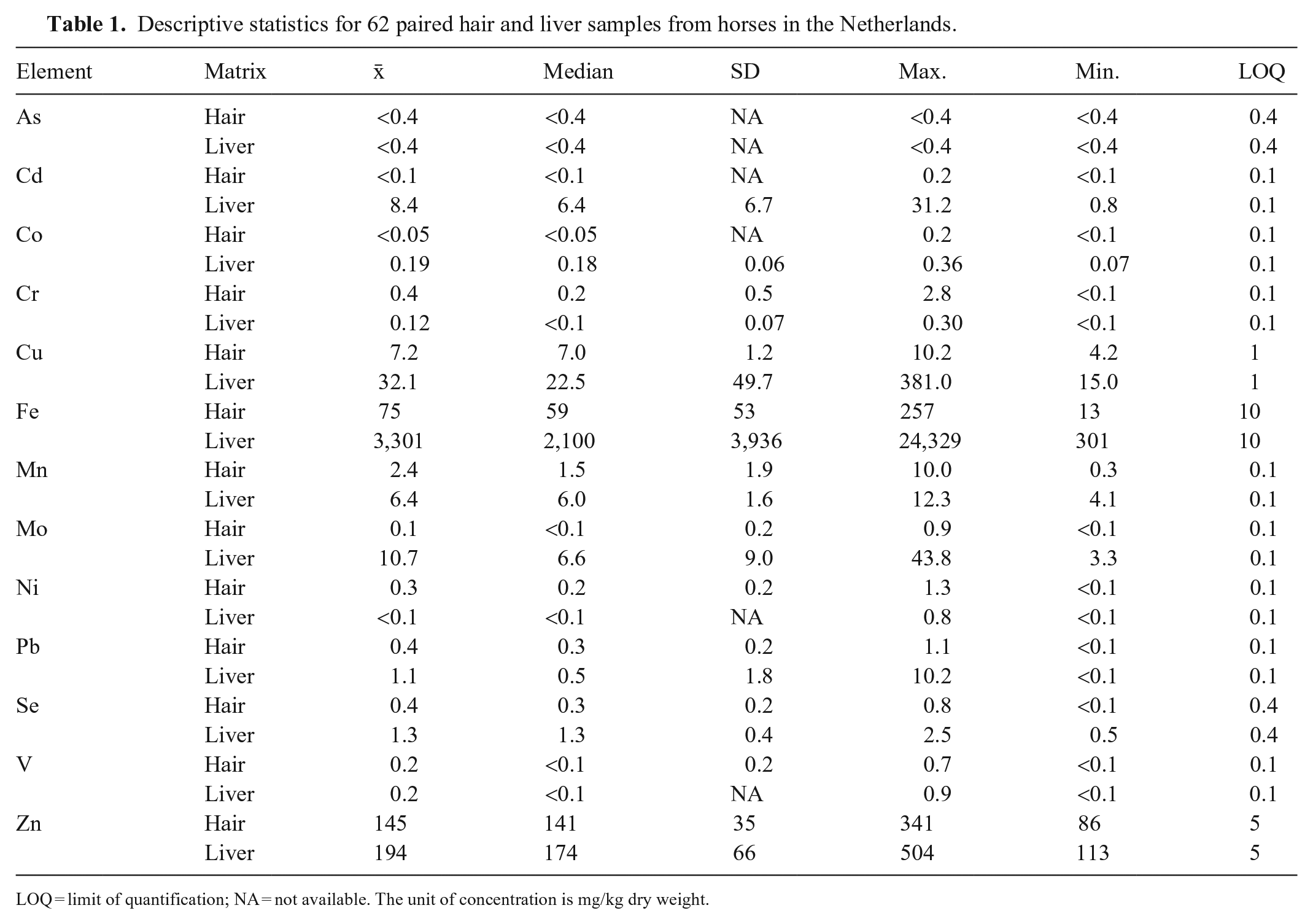
LOQ = limit of quantification; NA = not available. The unit of concentration is mg/kg dry weight.
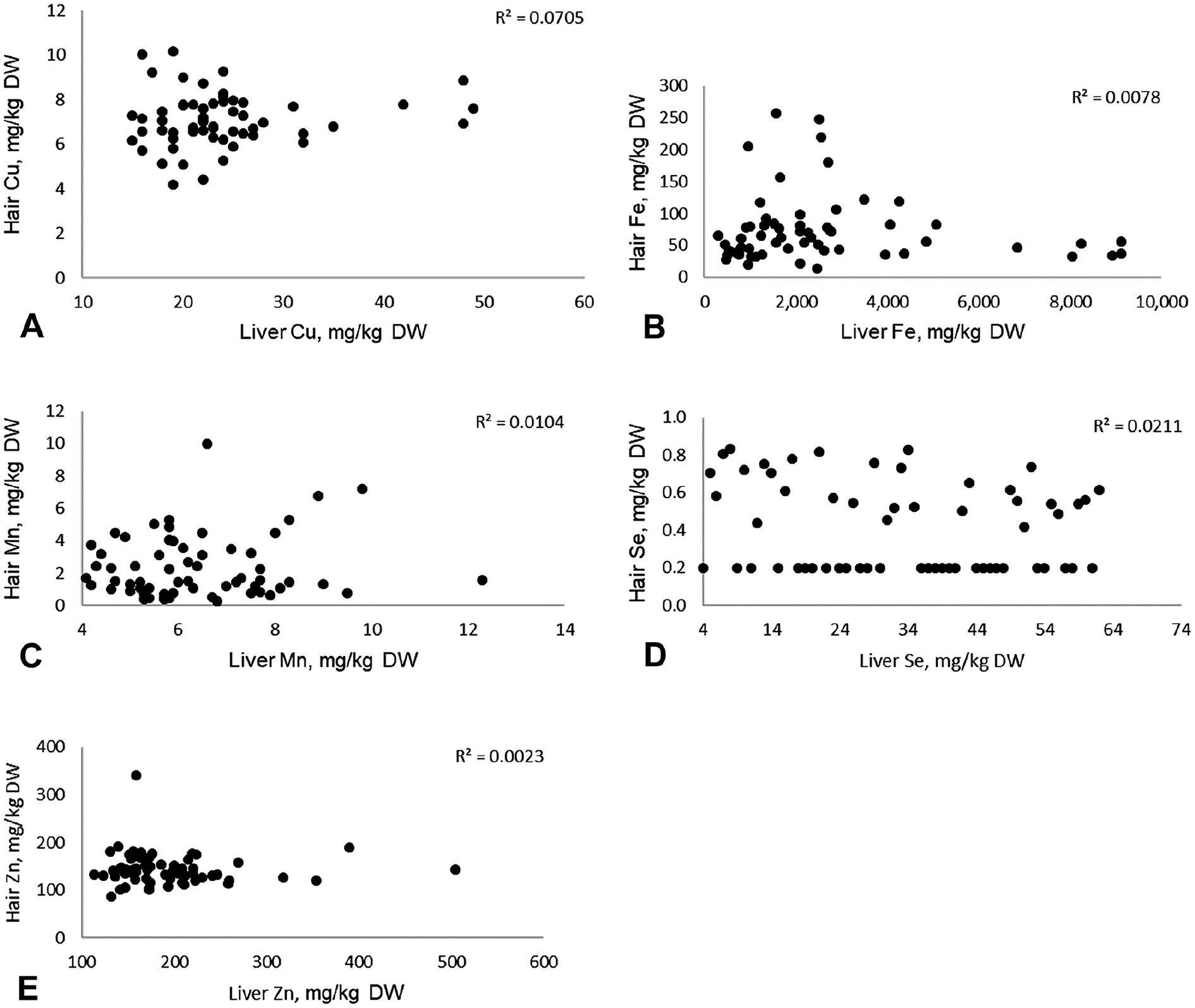
Scatter plots with R
2
-values for
Pearson product moment correlation (PPMC) coefficients and p-values for 62 paired hair and liver samples from horses in the Netherlands.
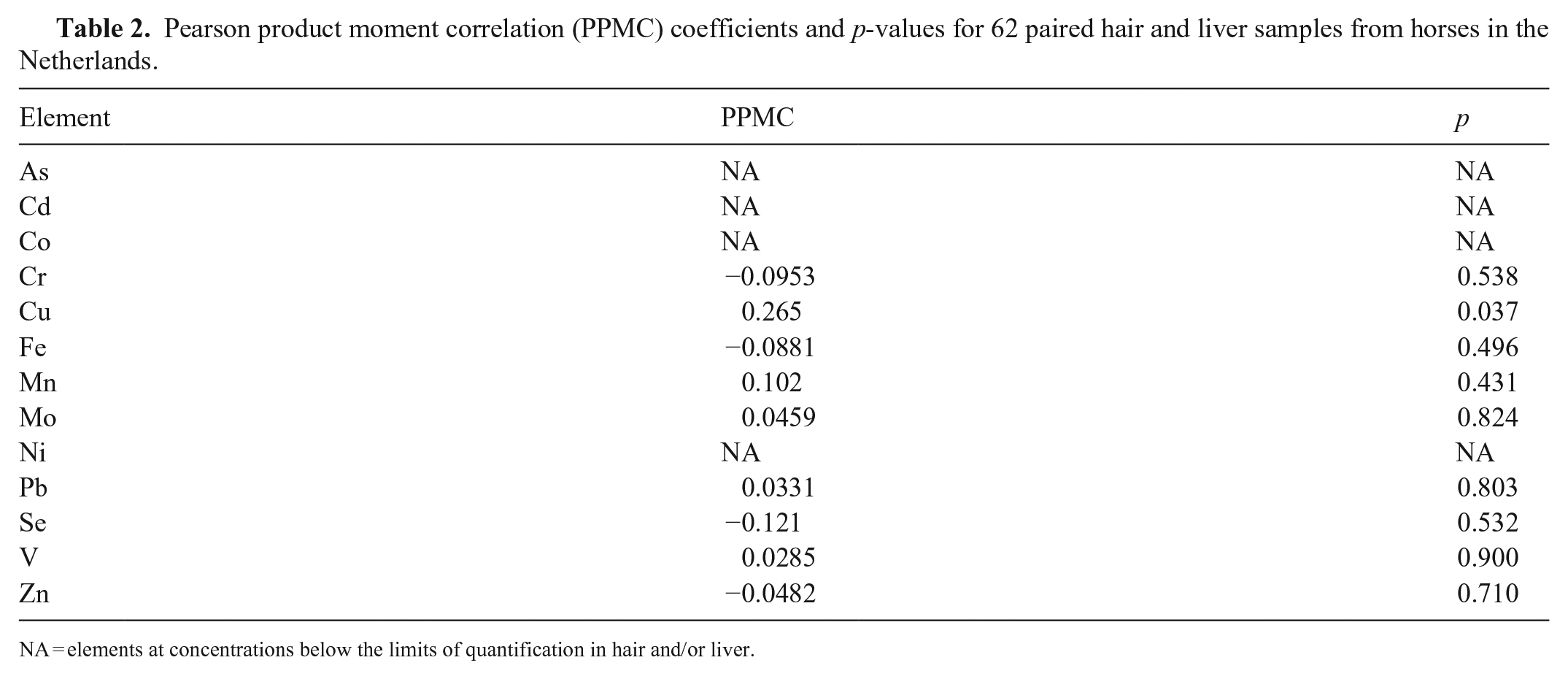
NA = elements at concentrations below the limits of quantification in hair and/or liver.
Pearson product moment correlation (PPMC) coefficients and p-values for trace element and heavy metal concentrations in 62 liver samples from horses in the Netherlands.
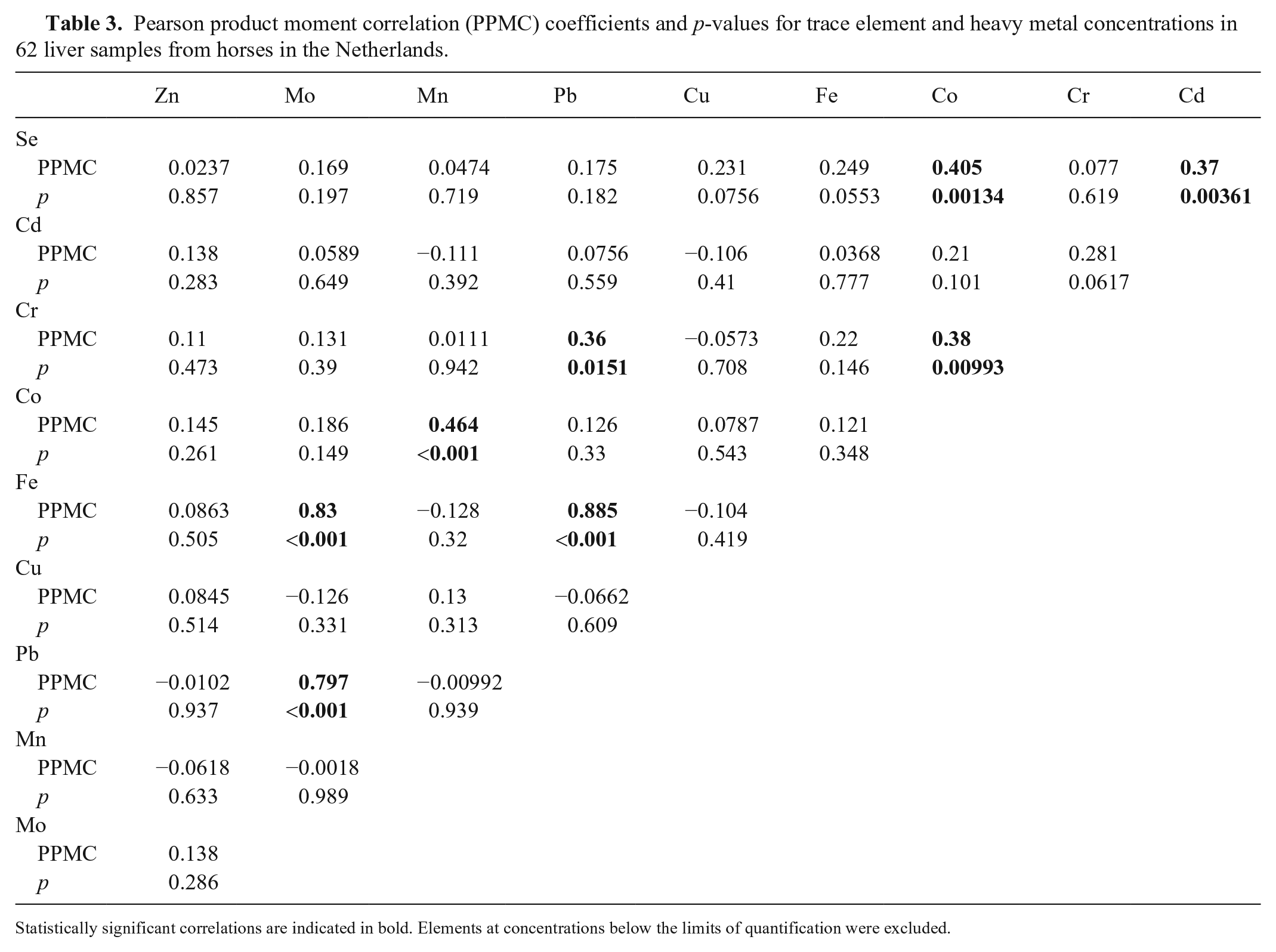
Statistically significant correlations are indicated in bold. Elements at concentrations below the limits of quantification were excluded.
Pearson product moment correlation (PPMC) coefficients and p-values for trace element and heavy metal concentrations in 62 hair samples from horses in the Netherlands.
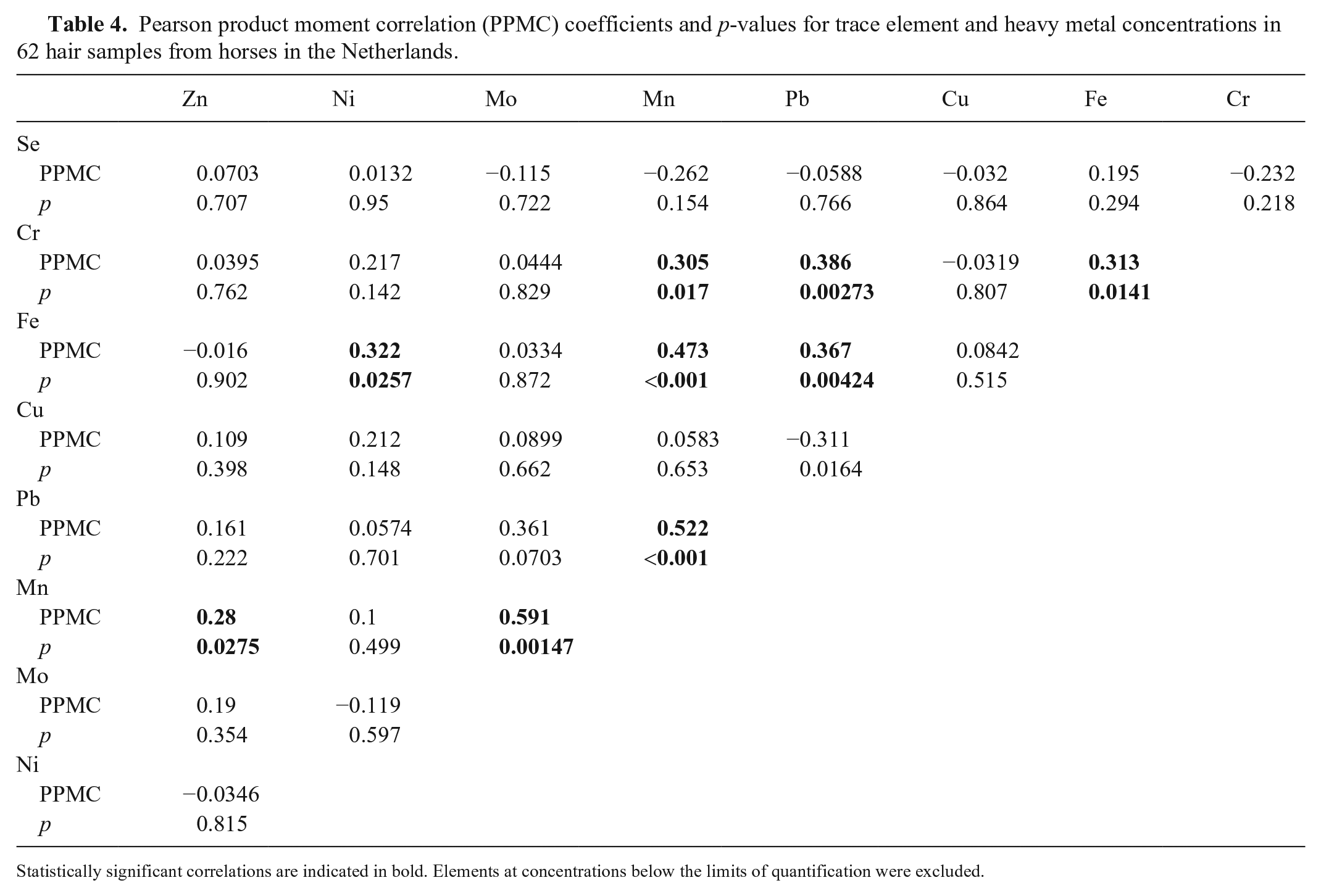
Statistically significant correlations are indicated in bold. Elements at concentrations below the limits of quantification were excluded.
We found no statistically significant correlations between mineral concentrations in liver and hair, with the exception of a slight correlation for Cu. The correlation between Cu concentrations in hair and liver were, however, considered to be too weak to be of clinical relevance. Our results do not preclude the possibility that correlations of mineral concentrations between hair and liver may occur under specific conditions, such as Se poisoning or in well-controlled feeding trials.
Various factors may contribute to the mineral variability of mammalian hair. Skin and hair are the interface between the body and the outer environment, and are therefore in constant contact with substances in the environment, including fluids and solids, with variable element concentrations. This contact may influence uptake of environmental elements into the hair, and leaching of elements from the hair. Results from controlled experiments that would allow quantification of environmental effects under various realistic exposure scenarios in horses are, however, not available in the scientific literature. Physiologic processes that impact the skin and hair through cell formation and secretions may also play a role, and could have some influence on correlations between body mineral status and minerals in hair. These may include apocrine, eccrine, and sebaceous secretions, and the continuous formation of new keratin during hair growth or self-renewal. 11 We chose hair sample cleaning using repeated warm water rinsing to partially preserve non-polar skin secretions that may contribute to trace element and heavy metal contents of hair. As in the case of environmental factors, however, quantification of the contributions of various physiologic processes to hair minerals in horses is not feasible based on available data.
Intra-tissue correlations occurred in liver and hair. This suggests that conditions of high or low exposure to heavy metals and mineral intake often involve multiple elements. The intra-tissue correlations were different for liver and hair. This could indicate that different drivers are important in the development of intra-tissue correlations within liver and hair. Further investigations regarding the drivers of such co-exposures could be of interest. For example, it could indicate location-based differences in environmental exposure to heavy metals, or differences in rates of mineral supplementation.
We conducted our study in apparently healthy horses presented for slaughter. Trace element intake and exposures to toxic heavy metals could be assumed to be within typical ranges for horses in the Netherlands. High exposure levels were therefore outside the scope of our study, and potential hair and liver correlations under conditions of high exposure cannot be excluded based on our results. Paired samples of hair and liver under conditions of clinically relevant heavy metal exposures will be necessary to draw conclusions regarding the potential usefulness of horsehair as an indicator of heavy metal exposures under such conditions. We also did not study differences in hair segments, whereby potential differences in correlations when using proximal or distal hair segments might be determined. Intra-hepatic and intra-hair correlations were not a specific goal of our study, but several such correlations were observed and quantified. Examples of correlations include Cr/Pb and Fe/Pb in liver and hair, as well as Fe/Mo, Fe/Pb, and Pb/Mo in liver. These correlations may reflect co-exposures or interactions, but more data, including exposure data, would be needed to assess the likely drivers of these correlations. We could not determine the clinical significance of intra-tissue correlations based on our data. Although our data indicate expected concentration ranges in liver and hair under field conditions in the Netherlands, further data, including more horses and various seasons, will be required to confidently define RIs.
Our results suggest that, given the lack of correlation observed between mineral concentrations in liver and hair samples of horses under field conditions, external environmental factors and/or physiologic processes unrelated to liver mineral concentrations may play a relatively large role in hair trace mineral concentrations. Our results do not support the use of hair to determine mineral status in the horse under field conditions in the Netherlands.
