Abstract
The primary objective of the current study was to describe micronutrient concentrations in livers collected during postmortem examination from 501 beef calves and fetuses in western Canada. The most frequently identified deficiencies based on reported ranges for aborted fetuses were magnesium, copper, and vitamin E. For stillborn, neonatal, and postnatal calves, the most common deficiencies were magnesium, vitamin A, and vitamin E. However, for calves that died after 3 days of age, selenium deficiency was also very likely. Concentrations of all micronutrients examined in the study except zinc (
Introduction
Trace mineral and vitamin deficiencies contribute to a number of causes of fetal, neonatal, and postnatal losses in beef calves as well as having important impacts on reproductive performance.6,11,20 In addition to well-documented degenerative conditions such as the myocardial and skeletal muscle lesions associated with selenium and vitamin E deficiency in beef calves, many micronutrients also play an important role in the development and function of the immune system.6,11 Liver samples collected during postmortem examination can provide information on the animal’s micronutrient status and the potential role of trace mineral and vitamin nutrition in disease processes.
Although many diagnostic laboratories can provide a comprehensive analysis of micronutrient concentrations from a fresh liver sample, the interpretation of the findings is limited by a lack of understanding of expected levels for beef calves and how these levels change based on the micronutrient and age of the calf. In a study that attempted to address this issue, zinc concentrations were examined in 474 liver samples from dairy and beef calves up to 1 year of age. 18 The authors reported 95% confidence limits to be used for the comparison of group means and 95% prediction limits for the comparison of individual samples. 18 Similar confidence intervals and prediction limits have not been reported for micronutrients other than zinc.
The values used by most reference laboratories represent a collection of data from several studies with many questions that could limit their application in diagnosis and surveillance. 18 The source for the most commonly cited reference values 16 does not clarify whether the samples included were from animals with obvious signs of deficiency or toxicosis or what laboratory methods were used. 18 Other information needed to determine the validity and applicability of the reference range could include when or where the samples were collected, how the reference ranges were calculated, whether the reported ranges were based on data from dairy or beef animals, and how many samples were analyzed per age group to develop the reference ranges.
Micronutrient uptake and metabolism in the calf shift dramatically at birth and through the first few weeks of life. A previous analysis of liver zinc concentrations was not focused on neonatal losses and, therefore, only included predicted reference values for calves at 1 day and 30 days of age. 18 The model predictions were based on the assumption that trace mineral values varied in simple linear and quadratic relationships with calf age. 18 Additional age-related, risk period–specific reference values are required for fetuses and calves within the first 30 days of life because changes near the time of birth are not necessarily monotonic and easily represented with simple continuous functions. For example, the concentrations of several important micronutrients depend on the ingestion of colostrum. 6
There is no data specific to beef calves raised in extensively managed commercial operations with a spring calving season as is typical of many herds in western Canada and the northwestern part of the United States. Neonatal calf losses in these herds tend to be very seasonal. In a previous study, more than 95% of calves that died in 200 extensively managed spring calving herds were lost from birth and 2 months of age between January and May. 29 The results of any investigations of herd micronutrient status during this high risk period will be influenced by the quality of stored forage and effectiveness of supplementation programs.
Reference values are necessary specifically for beef calves because most dairy calves are fed vitamin and mineral supplemented milk replacers; whereas, almost all beef calves are solely reliant on milk from their dam, which will typically be fed stored locally grown forage. Micronutrient deficiencies have been well documented in commercial beef herds in western Canada and the United States.4,5,21,30 However, there is very little information on what animal and management factors influence liver micronutrient concentrations in young beef calves or evidence linking variations in observed micronutrient concentrations to the risk of specific problems in the fetus or neonatal calf.
Specific reference ranges are needed for fetuses, calves that die at or near birth, calves that die in the first few days after birth, and calves that die later but before the start of the summer pasture season. This is the period when most necropsies are conducted in spring calving herds and when liver samples are most likely to be collected from beef calves. The collection of samples from clinically normal live animals to generate true normal ranges would be very difficult. Beef calves from this age group are not routinely slaughtered. The feasibility of obtaining sufficient liver tissue for comprehensive trace mineral and vitamin testing from biopsy of a large number of privately owned newborn and neonatal calves is also limited. The primary objective of the current study was to describe micronutrient concentrations in livers collected during postmortem examination from beef calves in western Canada and to compare these values with current thresholds for interpretation based on calf age category. The second objective was to identify animal and herd management factors that might explain differences in concentrations among animals. Finally, the associations between select liver micronutrient concentrations and the occurrence of plausible histology changes were examined.
Materials and methods
Source of calf liver samples
The liver samples for analysis were collected from privately owned herds in Alberta and Saskatchewan as part of 3 projects to investigate risk factors for abortion and calf mortality in commercial cow-calf operations.
Study 1
Two hundred and three herds were recruited in 2001 as a part of a comprehensive study of factors affecting beef cattle productivity in western Canada. Study investigators invited private veterinary clinics to participate in this project. Within each practice, herds were eligible for enrollment based on completeness of animal identification, existing calving records, presence of animal handling facilities, and a relationship with a local veterinary clinic. Herds of less than 50 animals and herds not using a winter/spring calving season were excluded from the study. 25
From these herds, 29,713 cows were at risk of abortion and there were 29,970 full-term births. All calf losses were summarized for each herd for the complete 2002 calving season. More than 98% of all full-term calves from the 2002 calving season were born in these herds between January 1 and May 31, 2002. Only calves that were part of the 2002 calf crop and died or were aborted before May 31, 2002 were eligible for a necropsy examination and inclusion in subsequent analysis. For every abortion and calf death, participants were asked to record the date and cow identification, and to contact their private veterinarian to arrange for a necropsy examination.
Participating veterinarians were supplied with training materials that described the collection and preparation of tissues for submission and with all necessary supplies for sample submission. A standard set of tissues was requested from every case for histologic examination. The veterinarian completed a form detailing the history, date of abortion or death of the calf, condition of the carcass, stage of development or age of the calf, major morphologic gross diagnoses, and relevant necropsy findings. An overview of the etiologic and morphologic diagnoses in these herds has been reported elsewhere. 29 Each case was uniquely identified to provide consistent linkage between the laboratory findings and the calving record for the calf.
Veterinarians were instructed to collect fresh liver samples from all calves examined postmortem during April 2002. These samples were collected as part of a late protocol addition to the baseline pathology study to supplement the available information on the nutritional status of these herds. The timing of liver sample collection was based on the need to secure funding to support laboratory analysis.
Study 2
During the months of January to June in 2006 and 2007, there were 7 cow-calf herds participating in a previously described long-term local surveillance project in west central Alberta 23 (1,494 cows and heifers calved in 2006 and 1,505 cows and heifers calved in 2007). Herd owners were asked to record the cow identification and date for every animal known to have or suspected to have aborted. The producer was asked to make every effort to locate the fetus and placenta for laboratory testing. The herd owner was also asked to record the identification and date for any animal that died and to have the animal examined following a suggested protocol. Whenever possible, postmortem examinations were to be completed on farm by the herd owner’s veterinarian. A written report of the complete postmortem examination was required and was to include date of death, animal identification; estimated or reported age, sex, and weight; and a record of the samples submitted for laboratory verification. Veterinarians were asked to collect fresh liver samples from all calves that died within 3 days of birth during 2006 and 2007; some samples were also collected from calves that died later.
For both study 1 and study 2, the veterinarian performing the necropsy categorized each death by the stage of development or calf age based on examination and the history from the herd owner. An abortion was defined as the remains of a fetus resulting from a premature calving, judged to be at least 1 month before full-term. Any calf born with their hair coat complete and long, and incisor teeth erupted was considered full term and classified as a stillbirth rather than abortion. A stillbirth was defined as a full-term calf (≥8 months gestation) that died at or within 1 hr of birth, or a full-term calf that was found dead, had not been observed alive, and was obviously recently born. This definition for stillbirth relied on the history provided by the herd owner and was intended to include calves that were born alive but weak and died immediately after birth. Calves that were alive at 1 hr of age and died before the end of the third day of life were categorized as a neonatal mortality, while calves that were born alive and died between the beginning of the fourth day of life and 3 months of age were classified as postnatal or older calf mortality.
For both study 1 and study 2, tissue samples fixed in 10% buffered formalin were submitted to a diagnostic laboratory. a Fixed tissues were trimmed, processed routinely, stained with hematoxylin and eosin, 29 and submitted to a veterinary pathologist (American College of Veterinary Pathologists) for examination. All results were recorded using a standard electronic data collection form. Fresh liver samples were shipped to the Western College of Veterinary Medicine (University of Saskatchewan, Saskatoon, Saskatchewan, Canada).
Study 3
The final study was undertaken by the Saskatchewan Ministry of Agriculture to investigate the causes of abortion in cow-calf herds from December 2008 through March 2009. Private veterinary practitioners were asked to collect standard questionnaire information and samples from up to 4 aborted or stillborn calves on farms from their practice area. Practitioners could utilize training resources provided by the project and perform the necropsies themselves or submit fetuses directly to the regional laboratory for postmortem examination. Tissue samples and information collected by the veterinarians were to be forwarded to the regional laboratory and processed using a standardized fetal test protocol.
Lung, liver, brain, heart, placenta or caruncle, kidney, eyelid, thymus, spleen, skeletal muscle, tongue, adrenal gland, thyroid, and ileum were fixed in formalin and examined microscopically for lesions. Fresh liver samples were submitted for trace mineral analysis. Fresh liver samples from term stillborn calves with no other lesions were to be tested for vitamin E.
Laboratory analysis of tissue samples
All liver samples were to be shipped to the laboratory on ice, where they were protected from the light and stored at −70°C before analysis. The liver samples were analyzed by a commercial diagnostic laboratory
a
to determine the concentrations of vitamin E and trace minerals. High-performance liquid chromatography (HPLC) methods were applied to determine tocopherol
2
concentration in liver for the assessment of vitamin E. Briefly, 0.3–0.6 g of tissue was homogenized with KCl (0.146 mol/l) made up to a volume 25 ml; 1.5 ml of homogenate was added to a test tube along with 0.5 ml of 0.01 mol/l ascorbic acid; 1 ml of ethanol and the internal standard were added, and the tube was mixed on a vortex mixer for 45 sec, covered with tinfoil, and then put in a 70°C oven for 5 min. One milliliter of 10 mol/l KOH was then added. The tube was mixed with a vortex mixer and again covered with tinfoil and placed in the 70°C oven for 30 min. The tubes were allowed to cool at 4°C and then 4 ml of petroleum ether was added. The mixture was vortexed for another 45 sec and centrifuged
b
at 550 ×
The trace mineral concentrations were measured following an acid digestion technique. Samples were digested in a microwave accelerated reaction system (MARS-5) d byadding 2.5 ml of concentrated nitric acid and 2.5 ml ofdouble-distilled (DD) water to 1.0 ml of sample. Using the appropriate MARS-5 digestion vessels, the samples were placed into the microwave for a total of 20 min at 120°C and 120 psi. Digested samples were transferred to a 25-ml volumetric flask. The MARS containers were rinsed with DD water and this was also added to the flask. The flasks were brought to volume with DD water, covered, and mixed. Trace minerals were determined immediately following acid digestion by analyzing samples using an atomic scan inductively coupled plasma (ICP) spectrometer. e
The selenium concentrations were determined from liver in a similar manner to that described for trace minerals immediately following acid digestion by analyzing samples using an ICP spectrometer coupled with a hydride generator device to assist formation of volatile selenium hydrides. Selenium analysis included the use of a hydride generator to improve instrument sensitivity.
Liver samples from the first study were also analyzed by a second commercial diagnostic laboratory f to determine the concentrations of vitamin A as retinyl palmitate. The samples were analyzed using protocol number FC-LP-220 dated January 31, 2002. Briefly, absolute ethanol was added to the liver samples and then the vitamin A was saponified and extracted with hexane at room temperature. The vitamin A eluate was quantified by HPLC on a C18 column. All analyses and reference ranges for liver tissue were reported on a wet weight (WW) basis.
Statistical analysis
Only the liver micronutrient data, information on potential risk factors, and relevant histology findings were considered for statistical analysis. All data was managed and summarized using commercial spreadsheet and database programs.g,h Summary statistics were calculated for each study and each age group after summarizing across study groups. Age-group–specific summary statistics included percentiles and nonparametric 95% prediction intervals (2.5th–97.5th percentiles).
Additional descriptive statistics included creating a matrix of Pearson correlation coefficients among the log transformed values to look for common patterns in relatively higher or lower concentrations among the measured micronutrients. i This was carried out for all observations and then by age group.
Differences in micronutrient concentrations among age categories were assessed using multilevel mixed models with random intercepts to account for differences among studies and expected similarities among samples from the same herds. j All liver micronutrient concentrations were log transformed before analysis to improve the normality of residuals and model fit. The models were used to calculate 95% confidence intervals for each micronutrient for each age group, which were then back transformed to the original scale.
Because individual animal and herd management data were most complete for study 1, further analysis was undertaken to explore other risk factors that might explain differences in micronutrient concentrations among calves and herds. This analysis also used multilevel mixed models with random intercepts to account for clustering within herd. Unconditional associations were estimated between each risk factor and log transformed micronutrient concentrations of interest (selenium, copper, molybdenum, vitamin A, and vitamin E). Risk factors considered in this analysis included: ecological region, 25 cow age (heifer or older cow), precipitation during the previous growing season, 26 cow body condition score at pregnancy testing, cow body condition score at calving, whether any trace minerals were fed before calving, whether any vitamin A was fed before calving, whether the herd injected calves with selenium at birth, whether the herd were fed 2- or 3-year-old stored hay from previous growing seasons, if herd winter water supply was >1,000 ppm sulfate, and if herd winter water supply was >40 ppm iron. 24 Information on herd average serum trace mineral concentrations in cows at pregnancy testing was available for 34 herds.21,30
Finally, because the tissue samples were examined by the same pathologist using the same histology scoring system in study 1 and 2, the data was used to estimate the association between liver concentrations and tissue changes. Specifically, the association between concentrations of selenium and vitamin E in the liver and the occurrence of myopathy, necrosis, or mineralization in either any skeletal muscle or heart muscle were examined. The unconditional association between liver selenium, copper, zinc, vitamin A and vitamin E concentrations and thymic atrophy was also examined. All associations were estimated using generalized linear mixed models with a logit link function and binomial distribution and random intercept to account for clustering by herd. j
Results
Description of study sample and comparison to existing reference values
A total of 501 beef calf liver samples from Alberta and Saskatchewan were analyzed as part of the 3 studies between 2002 and 2009; 225 from study 1, 80 from study 2, and 196 from study 3 (Table 1). There was no significant variation among the micronutrient concentrations of the liver samples from the 3 studies for selenium (
Liver micronutrient analysis from each of the 3 studies included in the current report summarized as the mean.*
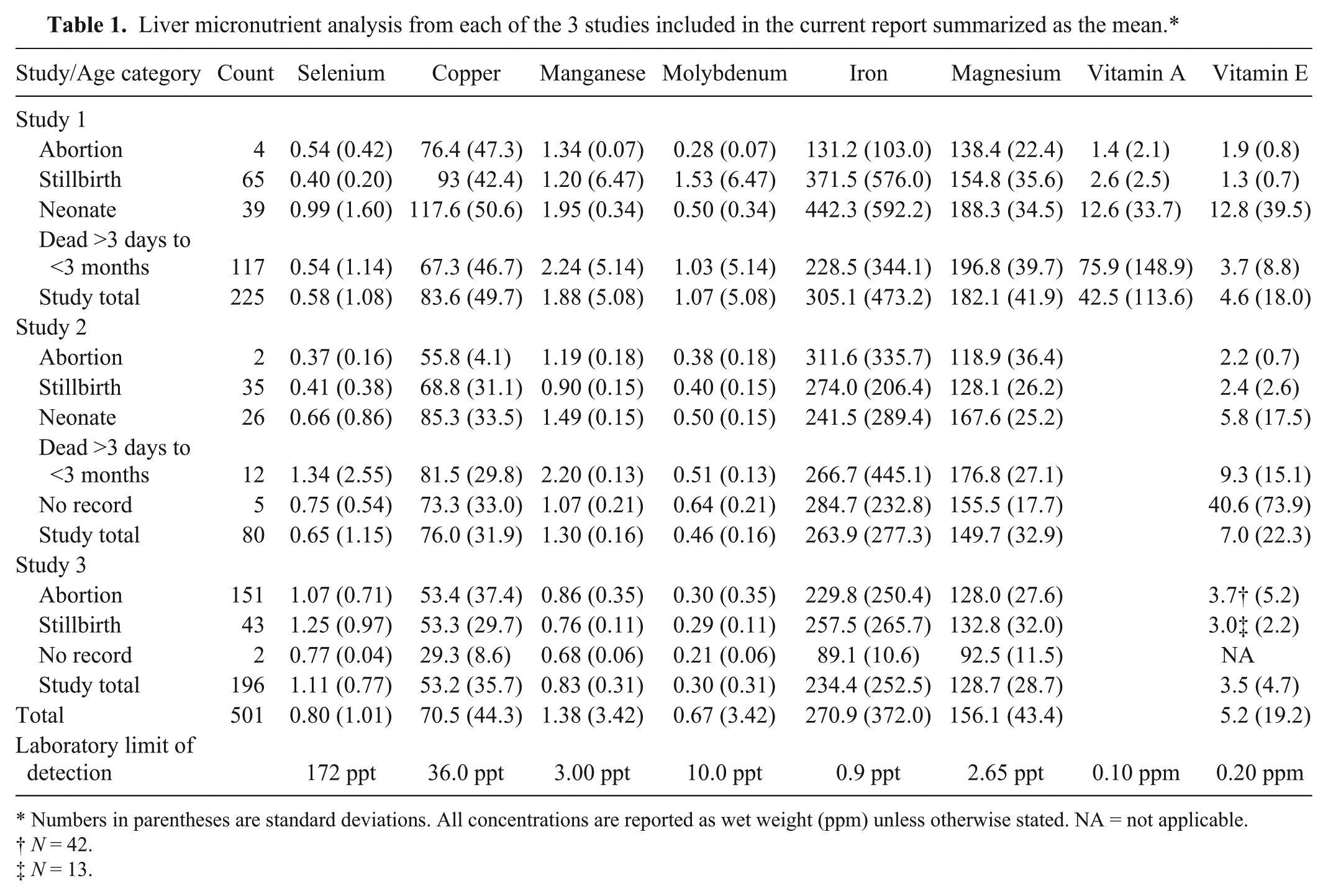
Numbers in parentheses are standard deviations. All concentrations are reported as wet weight (ppm) unless otherwise stated. NA = not applicable.
The samples from all studies were then combined for subsequent analysis and compared with reported reference ranges for deficient and adequate status16,17 (Tables 2–5). The most frequently identified deficiencies based on reported ranges for aborted fetuses were magnesium, copper, and vitamin E. For stillborn, neonatal, and postnatal calves, the most common deficiencies were magnesium, vitamin A, and vitamin E. However, for calves that died after 3 days of age, selenium deficiency was also very likely.
Distribution of liver micronutrient concentrations (ppm) for beef calf fetuses aborted before 9 months gestation from privately owned cow-calf herds in Alberta and Saskatchewan (Canada;
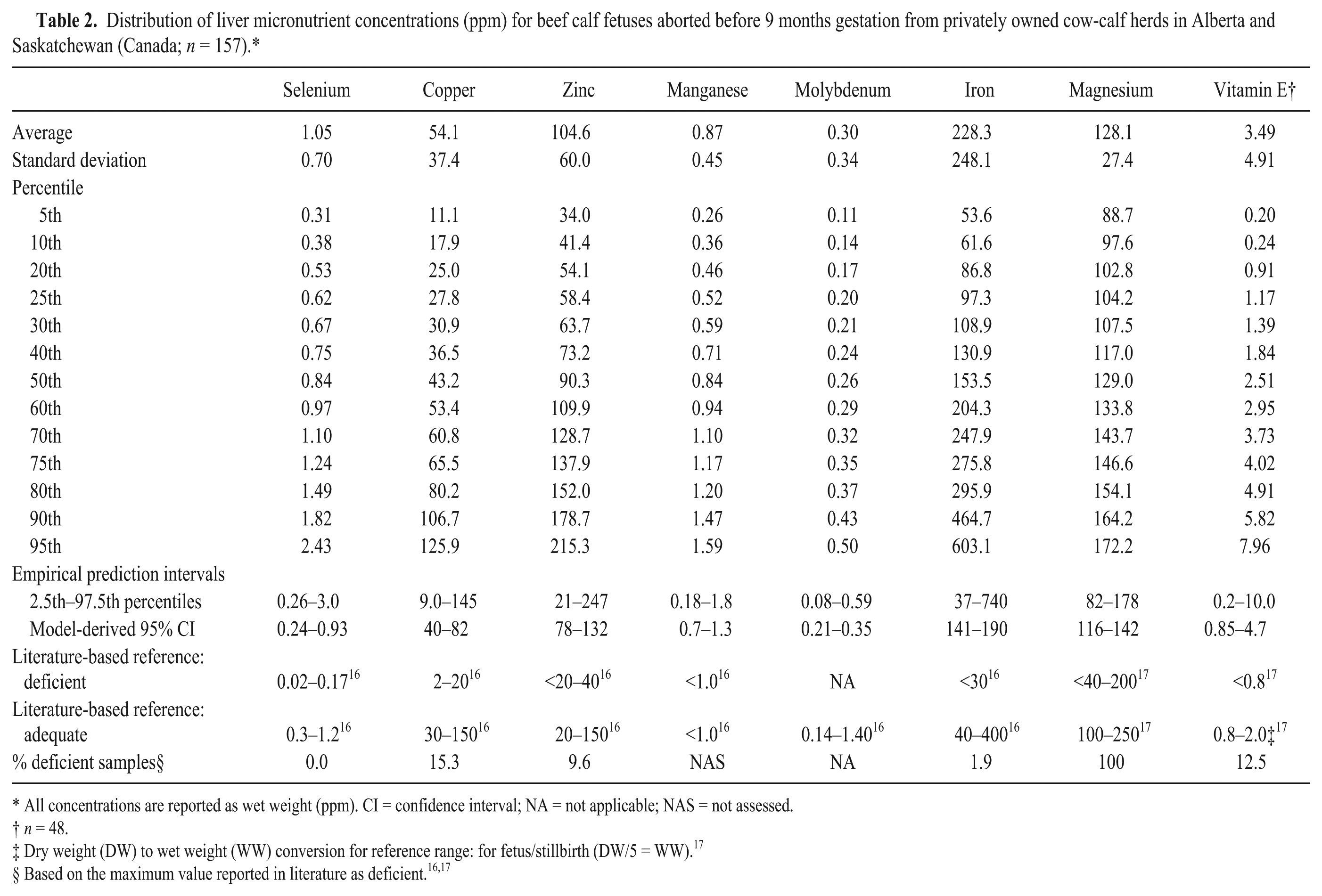
All concentrations are reported as wet weight (ppm). CI = confidence interval; NA = not applicable; NAS = not assessed.
Dry weight (DW) to wet weight (WW) conversion for reference range: for fetus/stillbirth (DW/5 = WW). 17
Based on the maximum value reported in literature as deficient.16,17
Distribution of liver micronutrient concentrations for full-term beef calves dead within 1 hr of birth from privately owned cow-calf herds in Alberta and Saskatchewan (Canada;
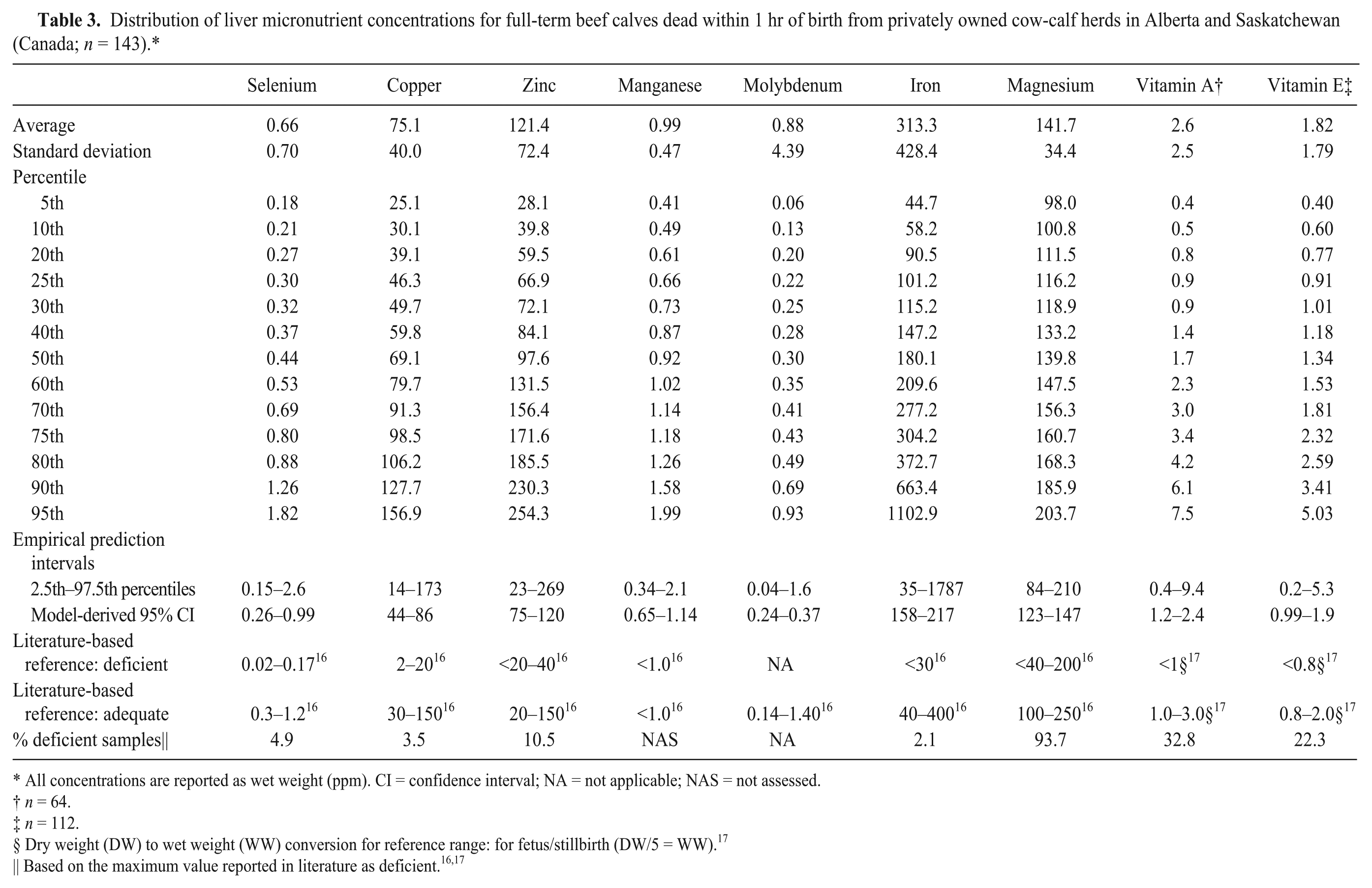
All concentrations are reported as wet weight (ppm). CI = confidence interval; NA = not applicable; NAS = not assessed.
Dry weight (DW) to wet weight (WW) conversion for reference range: for fetus/stillbirth (DW/5 = WW). 17
Based on the maximum value reported in literature as deficient.16,17
Distribution of liver micronutrient concentrations for beef calves dead between 1 hr and 3 days of birth from privately owned cow-calf herds in Alberta and Saskatchewan (Canada;
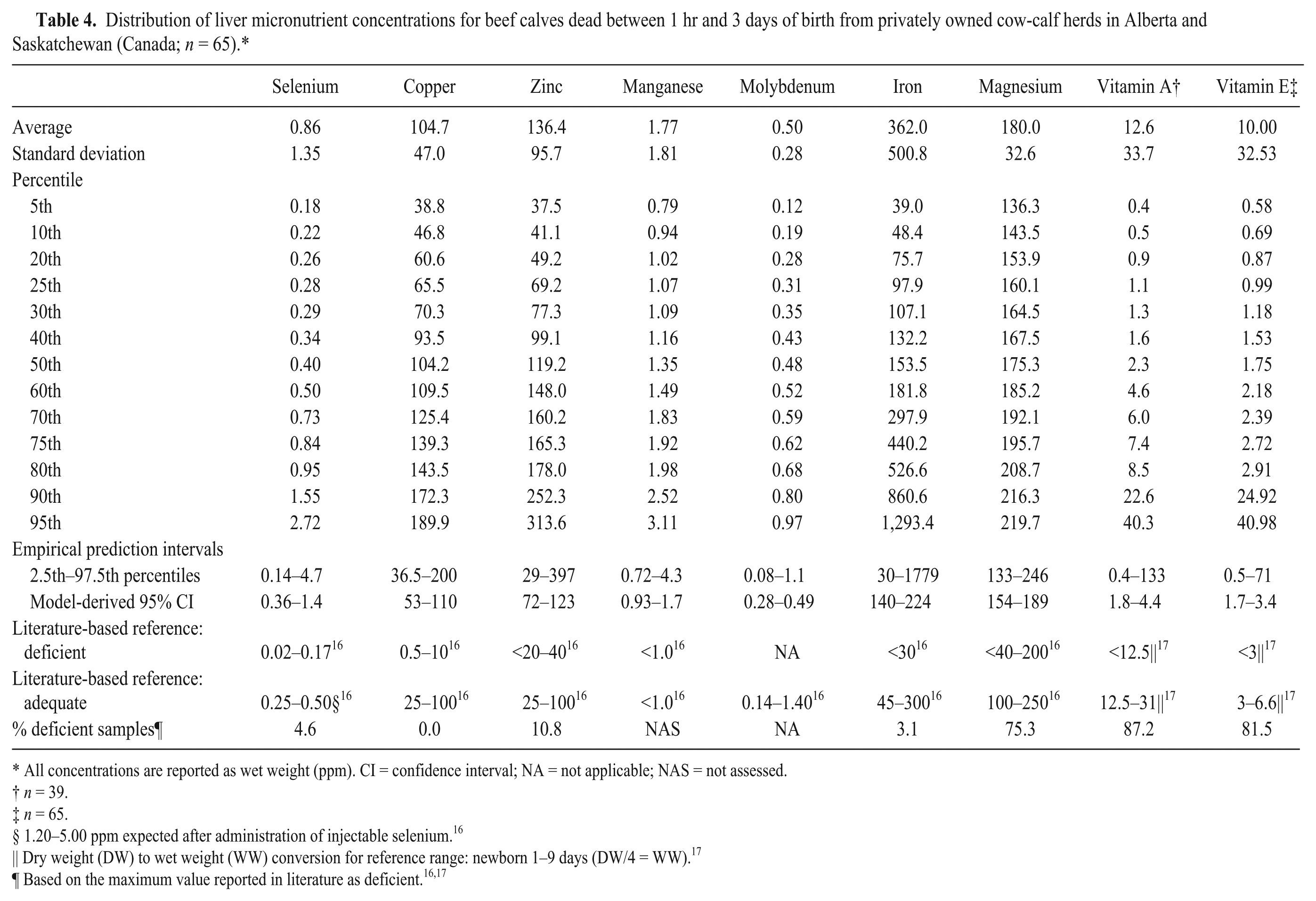
All concentrations are reported as wet weight (ppm). CI = confidence interval; NA = not applicable; NAS = not assessed.
1.20–5.00 ppm expected after administration of injectable selenium. 16
Dry weight (DW) to wet weight (WW) conversion for reference range: newborn 1–9 days (DW/4 = WW). 17
Based on the maximum value reported in literature as deficient.16,17
Distribution of liver micronutrient concentrations for beef calves dead between 3 days and 3 months of age from privately owned cow-calf herds in Alberta and Saskatchewan (Canada;
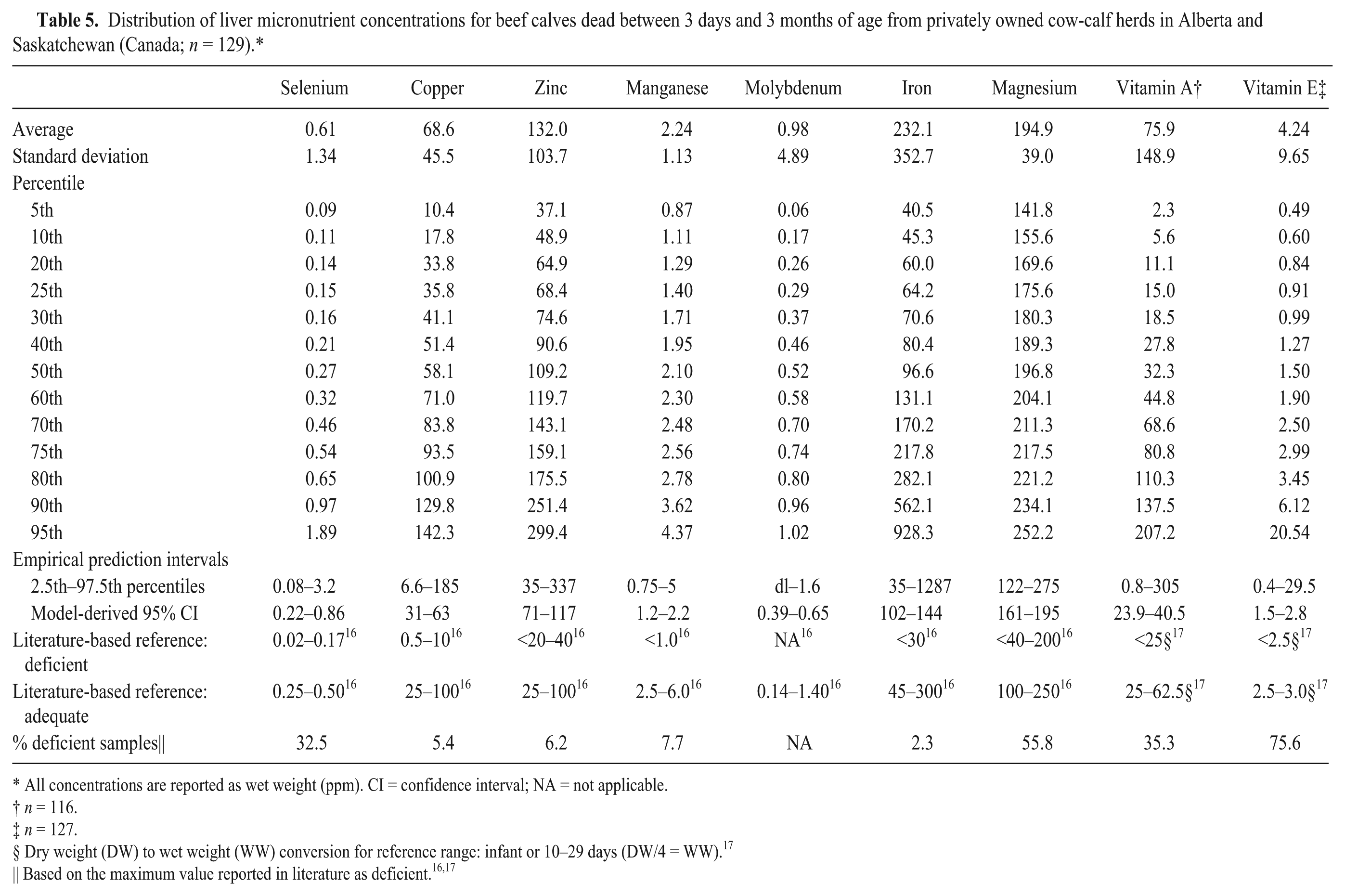
All concentrations are reported as wet weight (ppm). CI = confidence interval; NA = not applicable.
Dry weight (DW) to wet weight (WW) conversion for reference range: infant or 10–29 days (DW/4 = WW). 17
Based on the maximum value reported in literature as deficient.16,17
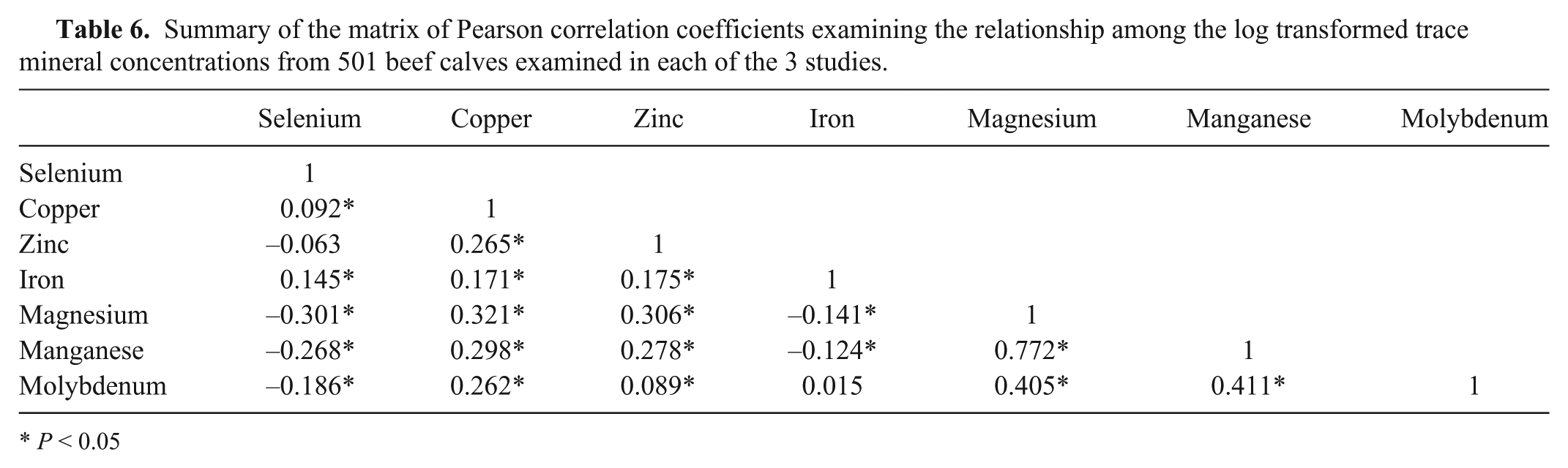
Correlations among the measured micronutrients
The only pair of micronutrients with a Pearson correlation coefficient of >0.5 was manganese and magnesium (Table 6). All other coefficients reflected little if any or low correlation.
12
This pattern did not change when correlation matrices were examined for each age group (data not shown). The Pearson correlation coefficients between selenium and vitamin E concentrations (
Summary of the matrix of Pearson correlation coefficients examining the relationship among the log transformed trace mineral concentrations from 501 beef calves examined in each of the 3 studies.
Effect of calf age
Calf age was not available for 5 calves from study 2, and 2 calves from study 3. These calves were excluded from further analysis. Concentrations of all micronutrients examined in the present study except zinc (
Liver selenium concentrations were higher in neonates, than in abortions (
Liver vitamin A was higher in calves that died after 3 days of age than in abortions (
Effect of other cow and calf factorsand herd management
Information on cow, calf, and herd management attributes were available for 221 samples from 106 herds collected as part of study 1. However, after accounting for clustering by herd and differences associated with calf age, there were no factors associated with liver concentrations of selenium, copper, manganese, magnesium, or vitamin E. For example, there was no association between whether the herd owner reported using injectable selenium in calves following birth and liver selenium concentrations (
Calves from herds that were fed any trace mineral supplement before calving (112 ppm, 95% confidence interval [CI] = 99–126) had higher liver zinc concentrations than calves from herds that were fed none (44 ppm, 95% CI = 19–100;
Trace mineral supplementation before calving (
There was an unconditional increase in vitamin A associated with the change from the lowest to highest level of precipitation during the previous growing season (
Association between liver micronutrient concentrations and histology findings
There were no significant associations (
Discussion
The liver samples available for the current study provided a unique opportunity to describe micronutrient concentrations in livers collected during postmortem examination from beef calves in western Canada and to re-examine existing reference ranges for liver micronutrient concentrations in very young beef calves that die on farm. Unlike most submissions to diagnostic laboratories, the calves included in the present study were not preferentially submitted based on history or clinical signs suggestive of deficiency or toxicity. All calves that died on farm during the eligible spring calving period were to be examined in the first 2 projects.23,25 For the third study, veterinarians were simply instructed to submit abortions from herds in their practice area. The projects were designed to document a number of different risk factors for fetal and calf loss in representative commercial beef herds, not just nutritional status. Measures of cow productivity and calf survival in herds from the first 2 projects were reported within expected ranges.23,27,29 None of the 3 study protocols included criteria that overtly biased selection to herds with micronutrient imbalances.
The study did not explicitly define normal or adequate ranges from the data as all of the values result from the analysis of samples that have come from calves that died on farm. Therefore, none of the liver data is derived from truly clinically normal animals. Reference metrics similar to the comparison values reported in the current study were also reported for dairy and beef calves by the researchers summarizing calf liver zinc analysis. 18 While the authors of the zinc study carefully reviewed calf postmortem records to exclude animals with overt clinical signs of zinc deficiency or toxicity, the same approach was not considered appropriate in the present study.
Given the wide range of trace minerals and vitamins evaluated in the study, there are very few causes of death in either the fetus or young calf that are not directly or potentially indirectly linked to micronutrient status of the dam or calf. For example, calves that died from accidental trauma could be considered a potential control group. However, many of these calves could still have subclinical evidence consistent with micronutrient deficiencies. Of the calves that died from trauma in the larger study cohort, 33% that died between 1 hr and 3 days had histologic evidence of skeletal muscle degenerative myopathy or necrosis as well as 39% of calves that died between 3 and 30 days of age. 28
The results of the laboratory analysis were summarized as percentiles with prediction intervals that can be compared to samples from individual beef calves that died on farm. For example, it was expected that 95% of all calves that died on farm fall within the reported prediction intervals and 90% of all calves that died on farm to have liver micronutrient concentrations above the 10% percentile. Ninety-five percent CIs were also reported that can be used for the comparison of means from groups of calves. The mean of a group of comparable calves would be expected to fall within the confidence interval for their age group 95 times out of 100.
The prediction limits, confidence intervals, and percentiles for each age group resulting from the current study are intended as tools for relative comparisons of future liver data from fetal and early calf losses in beef herds. While it was not reasonable to restrict the analysis to clinically normal calves and present the results as normal ranges, there was no clear link in the present study between trace mineral or vitamin concentration and expected clinical or pathological findings. Many of the calves were considered deficient based on existing reference limits, yet none of the concentrations examined were significantly associated with reported histologic lesions in the calves. The comparison ranges reported in the study could be used for the identification of outliers or very high risk herds.
In addition to describing the range of concentrations in these calves and examining the associations between micronutrient concentrations and histology changes, the objectives of the present study were to compare micronutrient concentrations to current thresholds for interpretation based on calf age category and to identifying animal and herd management factors associated with concentrations differences. The age-specific, percentile-based prediction intervals reported herein were wider than the currently used definitions for adequate liver concentrations for most of the nutrients examined including selenium, which has recommended values specific for the fetus and newborn. 16 In an earlier report, liver selenium concentrations in fetuses were higher than in the dams and increased during pregnancy. 10 More than 97.5% of selenium concentrations in the livers of fetuses and newborns were within the high but not toxic threshold limit of 3.0 ppm currently used in many laboratories, 16 suggesting there was very little evidence of selenium toxicity in these herds. Similarly, liver concentrations for neonatal calves that died within 3 days of birth were less than the 5.0 ppm concentration expected after the selenium injection many producers give to newborn calves. 16
Although liver selenium concentrations were significantly higher in neonates than in any other age group, there was no association between liver concentrations and whether the herd reported using selenium injections in newborn calves or history of supplementation to the cows. Previous studies have documented an association between selenium supplementation and calf liver selenium concentration. 1 The lack of an association in the current study could be related to the absence of individual level mineral intake data. It was not clear from the herd records which of the liver samples were from calves that had actually received an injection of selenium before they died.
Similarly, feed supplementation practices were reported at the herd level and most of the herds used provided them free choice. Only 34% of herd owners in the first study provided measured quantities of trace mineral supplements mixed in grain or silage. 24 Individual animal intake of free-choice trace mineral supplements is highly variable and has been previously documented to range between 0 and 974 g per head per day. 3
Liver selenium concentrations were deficient when compared with published criteria in more than 30% of calves that died after 3 days of age. In an earlier study, selenium concentrations were lower in fetuses with heart lesions than in aborted fetuses with no lesions. 15 However, there were no associations in the present study between selenium concentrations and documented lesions in either the skeletal muscle or heart.29,30
Liver is the primary storage organ for copper and is considered to be the best indicator of copper depletion.7,31 Copper deficiency has been primarily associated with immunosuppression, anemia, weak and coarse hair fibers, alteration in hair color, skeletal abnormalities, and growth retardation.11,19 Copper levels were low in more than 15% of aborted fetuses relative to criteria for deficiency currently reported for fetal liver samples 16 ; however, fetal liver copper levels can be difficult to interpret. Copper metabolism in the newborn calf does not mature until 4 days of age. 19 This is consistent with the finding that liver copper concentrations were highest in neonatal losses and stillbirths in the present study.
An earlier study reported that the level of copper progressively increased in the fetal liver during gestation and decreased in the maternal liver. 9 A store of liver copper is important to meet postnatal requirements as bovine milk is low in copper. Therefore, a critical concentration of 300 mg/kg dry matter (or 75 mg/kg wet weight) was proposed for the newborn calf. 8 At this threshold, more than 50% of newborn calves in the present study would be considered less than adequate. This is substantially higher than 10% of samples that contained less than 30 ppm wet weight referenced as the minimal adequate concentration. 16
Zinc was classified as deficient based on currently used guidelines in 10% of fetuses, stillbirths, and neonatal losses. 16 Previous prediction limits reported by other researchers for stillborn calves (36–439 ppm) were slightly higher than those observed in the current study. 18 Those for calves 30 days of age (32–365 ppm), however, were very similar to what was observed in the present study. The earlier study of zinc included calves up to 1 year of age and found that concentrations increased with increasing calf age. 18 In a slaughter plant study, zinc concentrations were higher in the fetus than the dam and progressively increased in the fetal liver during gestation. 10 There was no significant difference in liver concentrations among the age categories examined herein, though calf liver zinc concentrations were significantly higher in herds that supplemented trace minerals to the cows than in those that did not.
Liver concentration can reflect low zinc status but is not considered a good indicator of zinc adequacy. 11 Clinical signs of severe zinc deficiency include stiff gait and swelling of the distal extremities, parakeratosis, deformed hooves, abnormal appearing hair, and hair loss around the muzzle, ears, and eyes. Lymphoid depletion of the thymus, lymph nodes, Peyer patches, and gut-associated lymphoid tissue can be observed at postmortem examination. 11 There was, however, no association between liver zinc concentrations and thymic atrophy in the present study.
Manganese deficiency was not assessed for fetuses and stillborn calves as there were no clear existing standards. Fetal liver concentrations from a slaughter plant study were 0.6–0.7 times the maternal levels and did not change with stage of gestation. 10 Fetal liver concentrations are not expected to reach normal levels until the calf is 2–3 weeks of age. 16 This is consistent with the current finding that liver manganese was higher in both neonates and postnatal losses than in either abortions or stillbirths. Signs of deficiency have been reported to include skeletal damage in calves 11 ; however, skeletal deformities were very rare in the study herds and were not present in any of the calves with liver micronutrient analysis. 29
Molybdenum concentrations were below the elevated and toxic threshold limit of 2.0 ppm. 16 Liver molybdenum concentrations were highest in calves that died after 3 days of age. As previously reported for western Canada, herds in some areas were more likely to have high molybdenum levels than others. 21 Excess dietary molybdenum can interfere with copper absorption as can excess dietary iron. Between 2% and 3% of calves in each age group had iron concentrations approaching the threshold for the toxic range (1,800 ppm). 16 All of these calves had hemosiderosis of the liver noted on their histology reports. A similar proportion of stillborn and neonatal calves had low liver iron concentrations. Unlike many of the other nutrients examined, fetal liver iron concentrations do not change during gestation. 10 In the present study, liver iron concentrations were lowest in calves that died after 3 days of age. Also, calves from heifers had lower liver iron concentrations than calves from older cows. Milk alone is not a sufficient source of iron; however, calves that are deficient can quickly recover if they have access to soil and forage. 11
Although most of the samples were considered deficient for macronutrient magnesium when compared to current standards, 16 liver concentrations are not considered to be a good indicator of magnesium status. These values were included in the study because they are part of the panel, but do not appear to provide useful information and will not be considered further. To the authors’ knowledge, clinical magnesium deficiency has not been reported in fetuses and newborn calves.
Liver contains approximately 90% of the body vitamin A stores. 6 Calves are born with very limited vitamin A stores and must get most of their vitamin A from colostrum ingestion. Current guidelines provide specific values for the fetus, newborns, and infants to account for expected increases with colostrum absorption. 17 More than 30% of stillborn calves were considered deficient based on existing guidelines. Vitamin A analysis was not available for the calves examined as part of the second and third project, limiting the data from aborted fetuses. The very high percentage of neonatal calves in the present study with low vitamin A levels when compared to existing reference ranges suggests that many of these calves had died without ingesting colostrum.
Liver vitamin A concentrations in the present study were higher in postnatal losses than in any other age group. However, more than 30% were still considered deficient based on existing reference values. Cows can have depleted vitamin A stores following the winter feeding period and, therefore, colostral vitamin A might not be sufficient. 17 The pathological lesions associated with vitamin A deficiency in the calf could include loss of vision due to a failure of rhodopsin formation in the retina, defects in bone growth, and defects in growth and differentiation of epithelial tissues, frequently resulting in keratinization. 6 There were a small number of calves in the first study where histologic evidence of vitamin A deficiency was documented in changes in the salivary glands. 28 As salivary gland was not specifically requested in this study, and only sporadically submitted, the actual frequency of these lesions was not reported.
The major source of vitamin A for cattle is carotene, a precursor of vitamin A found in green feed. Cattle build up vitamin A stores while on pasture during the summer, but these begin to deplete in 2–3 months. Vitamin A is not stable in stored feed and loss from stored feed can exceed 50% in a year. 6 Lower liver vitamin A concentrations from the first study were unconditionally associated with lower levels of precipitation during the previous growing season and the use of 3-year-old stored feeds. Higher liver levels were unconditionally associated with feeding loose trace mineral mix during gestation. After adjusting for calf age, calves from heifers had higher liver vitamin A levels than calves from cows. This could be related to the tendency of many herd owners to provide heifers with the best available feed, often greenest or best appearing hay, to address their increased nutritional requirements during gestation.
Using existing reference intervals, more than 75% of calves that were born alive and died later were classified as deficient based on liver vitamin E concentrations. Green grass is an important source of vitamin E to the cow and later to the grazing calf. 17 Most calves died and most samples were collected for analysis before movement of cows to summer pasture and while the cows were still being provided with stored feed. As for vitamin A, the fetus primarily depends on colostrum for transfer of vitamin E from the dam because there is limited placental transfer 13 ; therefore, the reference ranges for calves born alive are higher than for those that are aborted or stillborn. Vitamin E declines in the liver during gestation. 22 Liver vitamin E concentrations in the present study were higher in calves that were born alive than in calves that died at birth. Unlike for vitamin A, the liver is not a primary, long-term storage site for vitamin E, and levels reflect recent uptake.6,14 Although a large proportion of calves were classified as deficient, there were no associations between liver vitamin E concentrations and histologic lesions in the skeletal muscle or heart in these calves.
In conclusion, the data summarized herein can provide an index for comparing micronutrient concentrations from beef calves that die on farm in spring calving herds, but cannot be used as a definition of normal. It is critical to consider the age of the calf, the potential for colostrum ingestion, and whether the cow is reliant on stored feed or has green grass available when interpreting these values. Cow age, supplementation history, and herd location were associated with the concentration of some of the micronutrients examined. The current study is unique in the number of samples included from young calves that were submitted as part of routine surveillance and not based on suspicion of either deficiency or toxicity. Most previous studies have been focused on older animals or fetuses and have contained a mix of beef and dairy animals with little or no information on their supplementation history. Laboratories can use the reported percentiles, prediction limits, and confidence intervals to provide age-specific reference point for identifying extreme values for the micronutrients most commonly at the center of issues related to poor calf health and herd productivity.
Footnotes
Acknowledgements
The authors appreciate the dedication of all of the veterinarians and producers who contributed to this project, and would also like to thank the Saskatchewan Ministry of Agriculture for providing the data for study 3.
a.
Prairie Diagnostics Services Inc., Saskatoon, Saskatchewan, Canada.
b.
International refrigerated centrifuge PR–6, International Equipment Co., Needham, MA.
c.
Ultrasphere TM ODS HPLC column, Beckman Coulter Canada Inc., Mississauga, Ontario, Canada.
d.
Microwave accelerated reaction system (MARS-5), CEM Corp., Matthews, NC.
e.
Trace Scan, Atomic scan 16/25 inductively coupled plasma spectrometer, Thermo Jarrell Ash Corp., Franklin, MA.
f.
Laboratory Services Division, University of Guelph, Guelph, Ontario, Canada.
g.
Microsoft Excel, Microsoft Corp., Redmond, WA.
h.
Microsoft Access, Microsoft Corp., Redmond, WA.
i.
PROC CORR, SAS version 9.3, SAS Institute Inc., Cary, NC.
j.
PROC MIXED, SAS version 9.3, SAS Institute Inc., Cary, NC.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding support was provided through the Western Interprovincial Scientific Studies Association (WISSA) for study 1 and the Alberta Beef Producers for study 2.
