Abstract
With the increasing popularity of viscoelastic coagulation analyzers, such as rotational thromboelastometry [ROTEM] and thromboelastography, the need for standardized methodology for appropriate interpretation has become increasingly important. Viscoelastic analysis is heavily influenced by a multitude of pre-analytic factors, both in vivo and in vitro, leading to a large amount of variation between institutions. We investigated the effect of room temperature during a 30-min sample rest time on ROTEM, which analyzed both intrinsic and extrinsic pathways. We also evaluated the feasibility of using ROTEM to assess coagulation in non-anesthetized domestic rabbits. Rabbits were selected because they are a common companion animal that could benefit from the use of viscoelastic analysis for various disease processes that could lead to coagulopathies. Citrated whole blood was collected from 10 rabbits and allowed to rest upright for 30 min either at room temperature (~ 21°C) or in a tube warmer (37°C) before analysis. There was no significant difference in results between room temperature and warmed samples, which suggests that allowing samples to rest at room temperature is acceptable clinically. Additionally, blood collection and analysis were feasible in all rabbits.
Introduction
Viscoelastic coagulation (VEC) analysis, including thromboelastography (TEG) and rotational thromboelastometry (ROTEM), have become increasingly popular in both human and veterinary medicine given that they provide a global assessment of hemostasis by analyzing whole blood samples for clotting initiation, amplification, propagation, and fibrinolysis. This global assessment is advantageous in comparison to traditional methods such as prothrombin time (PT) and activated partial thrombin time (aPPT) because the traditional methods are platelet-poor, plasma-based tests that exclude cellular components of coagulation such as platelets. 12 In addition, in the face of clinical challenges such as hypothermia or hemorrhage, VEC allows for more sensitive and specific measures that can help tailor therapy based on specific clotting abnormalities.7,10,16 Despite the advantages of using VEC in practice, results are heavily influenced by a multitude of in vivo factors, such as hematocrit and patient body temperature, and in vitro factors, such as quality and method of sample collection.4,8,13 To reduce variability, the Partnership on Rotation ViscoElastic Test Standardization (PROVETS) 4 was developed to help create guidelines for better reproducibility and comparability of data between institutions. Although the PROVETS document helps assess available literature, it highlights the need for additional supportive data. Additionally, these guidelines focus predominantly on traditional companion animals such as dogs and cats. 4
VEC was initially developed to be performed on fresh whole blood samples. A rest period is commonly practiced, however, because of clinical factors, such as facilities that need to transport samples to external laboratories, or availability of staff to consistently run samples immediately after collection. 4 Based on the literature, PROVETS suggests a consistent rest period of 30 min. 4 Allowing samples to rest adds several additional variables including temperature, timing, and chemicals used to prevent coagulation prior to analysis, which also require standardization for consistent results. 4
The effects of rest temperature on samples can be difficult to discern from studies given the large amount of variation that can occur in VEC methodology. Two publications in canine species were used by PROVETS to recommend a 30-min rest period at room temperature.4,13,17 Of these, one study focusing on TEG found that samples rested at 37°C as compared to room temperature appeared more hypercoagulable. However, these differences were judged to be clinically minor given that results from either methodology were within accepted canine reference intervals (RIs). 13 A second study, which investigated pre-analytic factors of ROTEM, noted that samples left at room temperature for 20 min had no significant difference to those rested at 37°C for 30 min. 17 Beyond these publications, research is limited on the effects of rest temperature in veterinary medicine. Several studies, in human, equine, and canine samples, indicate that actively cooled samples will result in a hypocoagulable sample.4,8,14,15,19 Additionally, in vivo–induced hypothermia has been shown to result in hypocoagulable states in humans, pigs, dogs, and rabbits.1,5–7,9,10,16,18 The significance of the induced hypocoagulability, however, varies among the studies. In addition, methodology in these studies had significant variation.
We selected rabbits for our study because rabbits are a commonly owned companion animal that likely have clinically unappreciated coagulopathies associated with documented disease processes including rabbit hemorrhagic disease, hepatopathies such as liver lobe torsion, or other underlying causes for sepsis.3,20,21 RIs have been established for PT and aPPT in healthy pet rabbits; however, these have limitations for interpretation. 11 Rabbits have been used previously for translational studies analyzing hypothermia and VEC. Although these studies did demonstrate hypocoagulability secondary to induced patient hypothermia, procedures including anesthesia-induced hypothermia and partial exsanguination limit their clinical application in most cases. Additionally, these studies used TEG and did not investigate other methodologies such as ROTEM.7,16 Finally, although the 2 available publications that use room temperature during rest period indicate minimal differences compared to those kept warmed, it is unknown if this holds true for other species, such as rabbits, that generally have a higher body temperature than humans or dogs.13,17
We determined the feasibility of ROTEM in non-anesthetized rabbits as well as analyzed the effect of resting temperature on sodium citrated whole blood measured by rotational VEC analysis. The tested hypothesis was that resting samples at room temperature would result in relative hypocoagulability compared to those kept at 37°C.
Materials and methods
Animals
We included 10 purpose-bred, ~ 4-mo-old, intact male, New Zealand White rabbits in our project. Rabbits were allowed a 2-wk acclimation period prior to the study. Animals were deemed healthy based on physical examination, food and water intake, and fecal output. Additionally, complete blood count, blood chemistry (alanine aminotransaminase, alkaline phosphatase, urea, creatinine, glucose, total protein, albumin, and globulin), and PT and aPTT were performed concurrently with sample collection for the study, with analysis performed by a commercial laboratory (Antech Diagnostics, Stillwater, OK). These animals were concurrently used for other coagulation studies (data not shown). No drugs or supplements were administered throughout the study or during the acclimation period. The study was approved by the Oklahoma State University (OSU; Stillwater, OK) Institutional Animal Care and Use Committee (protocol VM-19-8).
Blood collection and handling
Each rabbit was transported from the OSU Animal Resource facility to the OSU Boren Veterinary Teaching Hospital in pet carriers. The animals were allowed at least 1 h resting period prior to handling. Each animal was restrained physically, and the phlebotomy site was shaved and prepared aseptically. We collected 12 mL of blood from a jugular vein using a 21-gauge, 1.9 cm (0.75-in) butterfly catheter (SURFLO winged infusion set; Terumo) attached to 6-mL syringes (SUR-VET syringe with needle; Terumo). If venipuncture was not successful on the first attempt, the animal was allowed to recover for 1 wk before re-attempting venipuncture. Only one attempt was made to collect the entire blood sample, and redirection of the needle was not allowed. Upon reattempt, either jugular vein was considered acceptable. The first 5.4 mL of whole blood were used for a different coagulation study (data not shown), and the second syringe was used for our study reported herein. A volume of 2.7 mL of whole blood was placed in 2 commercial blood tubes containing 3.2% sodium citrate with the final concentration of blood to sodium citrate at 9:1 (Vacuette sodium citrate coagulation tube; Greiner Bio-One). The remaining 1.2 mL of blood was placed in EDTA (Vacuette MiniCollect K3 EDTA coagulation tube; Greiner Bio-One) and lithium heparin (Vacuette lithium heparin coagulation tube; Greiner Bio-One) tubes for complete blood count and blood chemistry. When placing blood within the blood tubes, the butterfly catheter and the blood tube caps were removed, and the samples were manually transferred into the blood tubes. After 8 manual inversions, one tube containing citrated blood was allowed to rest stationary and upright at room temperature (~ 21°C); the second tube of citrated blood was allowed to rest stationary and upright in a tube warmer (Isotemp FSGPDO2; Fisher Scientific) at 37°C (99°F).
ROTEM
All analyses were performed on a commercial rotational thromboelastometer (ROTEM Delta; Instrumentation Laboratory) by 1 of 2 investigators (D. Pathak or R. Maranville). Routine maintenance and quality control procedures were performed as recommended by the manufacturer.
After a 30-min rest time, samples were manually inverted 8 times then added to cuvettes for ROTEM analysis, with a random channel assignment using an online random number generator (www.randomizer.org). Cuvettes were pre-warmed to 37°C per the manufacturer’s instructions, and samples were run at 37°C during analysis per machine standardization. Activators for both intrinsic (INTEM; Tem Innovations) and extrinsic pathways (EXTEM; Tem Innovations) were used for each sample. The remaining whole blood in the sodium citrate was submitted for PT/aPTT panel. Citrated samples (300 µL of blood) were mixed with 20 µL of 0.2 M/L calcium chloride for recalcification for a total of 320 µL per sample, and either added to intrinsic or extrinsic pathway activators as automatically set by the device. Each rabbit had 4 analyses performed, including room temperature and warmed samples for both intrinsic and extrinsic pathways. Samples were randomly allocated to 1 of the 4 analyzer channels. Duration of analyses was set at 60 min or until the lysis index at 60 min was reported. Data were recorded for future analysis.
Statistical analysis
Continuous variables were reported as median ± SD (range). Generalized linear mixed models (GLMM) were built to explore the effect of type of analysis (intrinsic or extrinsic pathway activators) and temperature (room vs. warm) on each of the 6 outcomes, including clot time (CT), clot formation time (CFT), alpha, clot amplitude at 10 min (A10), clot amplitude at 20 min (A20), and maximum clot firmness (MCF), accounting for paired rabbit analyses (Fig. 1). For each outcome, “temperature” and “type of analysis” were included as fixed effect, and “rabbit ID” was included as a random effect. The random effect block included the intercept and used the variance component as a random effect covariance type. The advantage of GLMM is that it accounts for changes in temperature and type of analysis in the same analysis, reducing the chance of type I statistical errors. However, sensitivity analyses were performed with more traditional techniques, namely nonparametric related–samples Friedman 2-way ANOVA by ranks. No significant differences in the interpretation of the results were observed, and therefore the analysis based on mixed models was retained. Statistical significance was considered
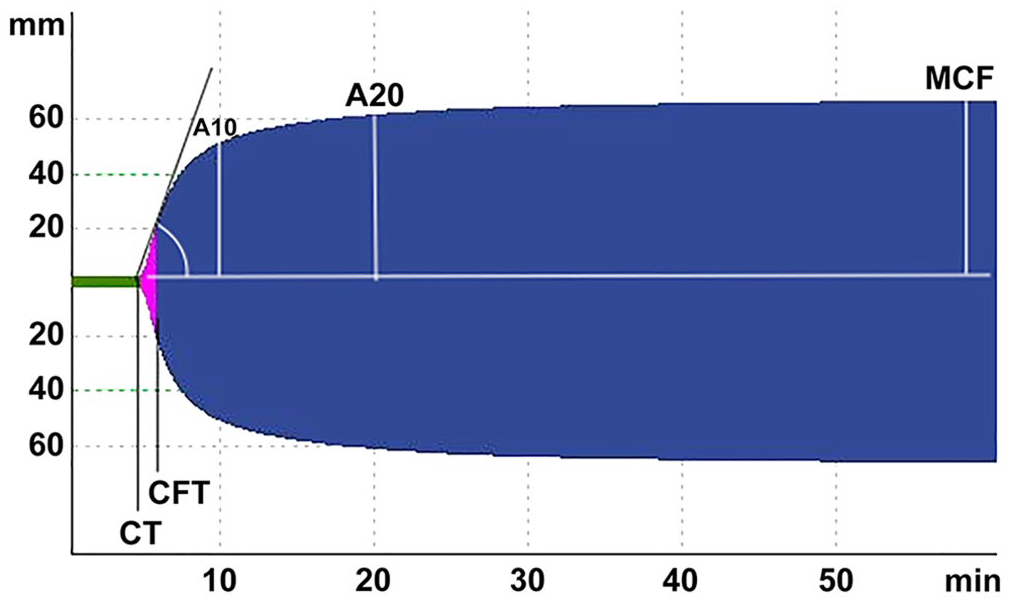
Rotational viscoelastic analysis of a New Zealand White rabbit.
Results
Based on the health assessment, all 10 rabbits were considered healthy and were included in this project. There were no complications noted during the blood collection. A single rabbit had sample collection delayed to a different day because of poor venipuncture on first attempt. None of the rabbits had clinical signs of bleeding abnormalities, such as excessive hematoma formation after venipuncture. No lipemic or hemolytic samples were noted based on visual assessment and via the submitted blood chemistry analysis.
The models built for each dependent variable (CT, alpha, CFT, A10, A20, and MCF) including rabbit as random effect and temperature and analysis methodology (intrinsic or extrinsic pathway activators) as fixed effects, demonstrated no difference in any of the variables depending on sample temperature and differences in all of the variables depending on the methodology used (Table 1).
Rotational viscoelastic analysis of 10 New Zealand White rabbits analyzed after a 30-min resting period at room temperature or warmed (37°C), using intrinsic (INTEM) and extrinsic (EXTEM) pathway activators.
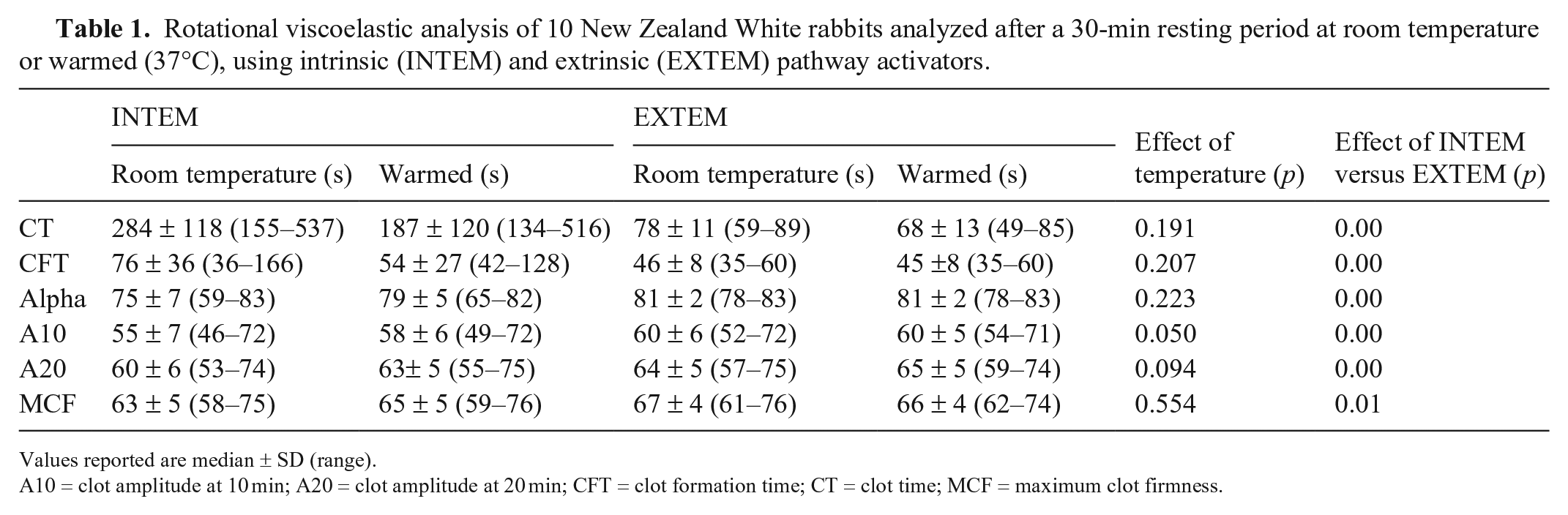
Values reported are median ± SD (range).
A10 = clot amplitude at 10 min; A20 = clot amplitude at 20 min; CFT = clot formation time; CT = clot time; MCF = maximum clot firmness.
Discussion
We hypothesized that samples left at room temperature would be relatively hypocoagulable compared to those kept at 37°C. We found no significant difference between samples rested at room temperature versus kept warm at 37°C, and thus rejected the hypothesis. Clot firmness at 10 min (A10) had a
Although our hypothesis was rejected, these findings were consistent with 2 previous studies on canine blood.13,17 These studies concluded that the effects of allowing samples to remain at room temperature were minimal. It is reasonable to assume that samples rested at room temperature have some degree of cooling effect, however, the degree to which this occurs is not established. Assessing actual temperatures of the samples left to rest at room temperature for 30 min would be useful but was not pursued in our study because obtaining the temperature of these samples may affect the results of VEC analysis. Based on these findings and the belief that standardization for samples at room temperature would be more feasible clinically, PROVETS has recommended viscoelastic samples to be rested for 30 min at room temperature. 4 Our study is consistent with PROVETS recommendation.
Our results suggest that the use of ROTEM to assess coagulation in rabbits is feasible, with all but one sample being obtained and analyzed on first attempt. One sample was delayed because of our inability to obtain a clean venipuncture sample, which is a potential complication in clinical practice that should be considered in patients of any species. 4 As a result of invasive interventions, TEG studies in rabbits7,16 likely did not produce clinically applicable values and are not appropriate for comparison with our study.
It should be noted that ROTEM coagulation tests are based on human body temperature (37°C, 98.6°F), whereas companion exotic mammals often have higher temperatures. For instance, normal rabbit body temperature is generally considered to be 38–39.9°C (100.4–103.8°F). 2 To date, there is limited data in companion animal medicine to suggest whether analysis should be run based on patient body temperature. 4 Previous studies analyzing in vivo hypothermia, both in humans and rabbits, have shown that samples run at patient temperature compared to the standardized 37°C trended toward being more hypocoagulable.6,16 Performing the analysis at 37°C is relatively hypothermic for rabbit patients and more hypocoagulable tracings may result. Further studies should compare the standardized temperature versus relevant patient body temperature.
Our study was limited by the number of rabbits and is not appropriate to establish RIs. It was also assumed, via physical exam and hematologic analysis, that all rabbits were healthy without undiagnosed coagulopathies. Based on the lack of abnormal physical exam findings or abnormalities during venipuncture, underlying coagulopathies are unlikely but cannot be completely excluded. In addition, the sample population in our study was specific to purpose-bred juvenile male New Zealand White rabbits and may not reflect the population of rabbits seen by a regular practitioner. It is our hope, however, that these data can add to a general knowledge base of rabbit coagulation and will help initiate further investigations of rabbit viscoelastic analysis. Given the large number of pre-analytic factors that influence VEC, it is our recommendation, and that of PROVETS, to establish institutional standardized methodology and RIs for individual species. 4
Although our initial hypothesis of hypocoagulable tracings secondary to room temperature samples at rest was rejected, our findings were similar to studies in canine VEC analysis as well as the recent PROVETS suggestions for standardization.4,13,17 We, therefore, conclude that rotational viscoelastic samples may be left at room temperature during rest. Additionally, we demonstrated analytical feasibility in non-anesthetized rabbits. Institutional standardization, including rest temperature, and establishing RIs for any species to be analyzed is crucial before instituting VEC in practice.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the Oklahoma State University (OSU) College of Veterinary Medicine and the Boehringer Ingelheim Veterinary Research Scholars Summer Program, the OSU Joan Kirkpatrick Chair in Small Animal Medicine, the OSU Dr. Kristie Plunkett Exotic Animal Fund, and the OSU Patricia Henthorne Clinical Professorship in Small Animal Medicine.
