Abstract
Antemortem diagnosis of neuroborreliosis in horses has been hindered by both the low sensitivity of PCR testing for Borrelia burgdorferi in CSF and the low specificity of serum:CSF ELISA ratios used to determine intrathecal antibody production against the bacterium. PCR testing of the CSF of an adult horse with acute neurologic disease for the B. burgdorferi flagellin gene was negative. However, we enriched B. burgdorferi DNA through nucleic acid hybrid capture, followed by next-generation sequencing, and identified B. burgdorferi in the CSF of the horse, confirming a diagnosis of neuroborreliosis.
Antemortem confirmation of neuroborreliosis caused by Borrelia burgdorferi has been a challenge in both horses and humans.2,5,8,9,17 In contrast to many other bacterial infections of the CNS, there is not an abundance of B. burgdorferi bacteria or detectable bacterial DNA in the CSF of patients with neuroborreliosis.2,17 Consequently, nucleic acid testing (i.e., PCR) of CSF has been an insensitive test for neuroborreliosis in both horses and humans.2,9,11,17 The challenge in accurately diagnosing neuroborreliosis is compounded by the relatively low replication rate of B. burgdorferi, making traditional culture impractical. 2 We describe here an enhancement method for detecting B. burgdorferi DNA in the CSF of a horse suspected to have neuroborreliosis.
An 11-y-old old Swedish Warmblood mare (horse 1) from Connecticut, USA was examined because of an acute onset of fever (40°C) and neurologic signs, including obtundation, ataxia in all limbs, and anisocoria. Two weeks prior, the mare had been treated for Streptococcus equi subsp. zooepidemicus maxillary sinusitis with antibiotics and sinus lavage. The associated unilateral (right side) mucoid nasal discharge resolved within 5 d; follow-up sinus radiographs were normal. One week later, the horse traveled to a nearby horse show where fever and neurologic signs developed. Initial on-farm treatments included the antimicrobial enrofloxacin (Baytril 100, 5 mg/kg IV; Bayer) and a NSAID (dipyrone, 30 mg/kg IV). The horse was then immediately referred to a tertiary care equine hospital for further testing and treatment. Differential diagnoses included West Nile virus (WNV) infection, equine eastern encephalitis (EEE), equine herpesvirus (Equid alphaherpesvirus) myeloencephalopathy, bacterial meningitis, protozoal myelitis, and neuroborreliosis.
Clinical examination at the referral facility confirmed severe ataxia (grade 4 of 5) in all 4 limbs, a right-sided head tilt, and a tendency to fall toward the right. Temperature was normal (37.8°C); heart rate (56 bpm) and respiratory rate (44 bpm) were both elevated. CBC revealed neutrophilic leukocytosis (12.8 × 109/L; RI: 5.5–12.5 × 109/L). Plasma fibrinogen was increased (6.0 g/L; RI: 2–4 g/L), and out-of-range serum biochemistry results included increased serum amyloid A (3,000 mg/L; RI: <30 mg/L) concentration, hyperglycemia (18.0 mmol/ L; RI: 3.7–8.3 mmol/l), and hyperglobulinemia (53 g/L; RI: 24–44 g/L). Initial treatments included IV administration of penicillin (penicillin G potassium, 20,000 IU/kg q6h; Athenex), enrofloxacin (Baytril, 5 mg/kg q24h; Bayer), and prednisolone sodium succinate (Solu-Delta-Cortef, 0.5 mg/kg single dose; Zoetis). A nasal swab taken just prior to referral for equine herpesvirus PCR was negative. After 48 h of treatment, minimal improvement was noted, and hyperesthesia developed throughout the body.
On day 3 of hospitalization, results of a lumbosacral spinal tap revealed neutrophilic pleocytosis with 864 × 106 WBC/L (RI: <5 × 106 WBC/L). Of the WBCs, 85% were non-degenerate neutrophils and 15% were large mononuclear cells (lymphocytes and macrophages). CSF protein was markedly increased at 8.0 g/L (RI: <0.7 g/L). The CSF results were most compatible with EEE or bacterial meningitis, including neuroborreliosis. Horses commonly have neutrophilic pleocytosis with neuroborreliosis, whereas humans routinely have lymphocytic pleocytosis.2,9 No organisms were observed on cytologic examination of the CSF, and aerobic bacterial culture of the fluid had no bacterial growth. Serum:CSF antibody ratio testing for Sarcocystis neurona and Neospora hughesi did not support intrathecal production of antibody against either organism, making equine protozoal myelitis unlikely. 15 Proper vaccination history against viral encephalitis made WNV and EEEV unlikely causes. Although serum multiplex ELISA antibody concentrations against 3 B. burgdorferi surface antigens (OspA, OspC, OspF) were within horse RIs, antibody concentrations against these antigens in the CSF were very high (Table 1). 18
Serum and CSF ELISA results for antibodies against Borrelia burgdorferi outer surface proteins (Osp) in horse 1 diagnosed with neuroborreliosis.

MFI = median fluorescent intensity.
Lyme multiplex assay; Animal Health Diagnostic Center, Cornell University. 18
No RIs are published for CSF.
PCR testing of CSF for the B. burgdorferi flagellin gene using forward primer sequence TCTTTTCTCTGGT GAGGGAGCT, reverse primer sequence TCCTTCCTGTT GAACACCCTCT, and probe sequence FAM-AAACTGCT CAGGCTGCACCGGTTC-BHQ1, as described previously, 12 was negative. Regardless, based on the clinical findings and possible exposure to ticks in a Lyme-endemic area, in addition to CSF serology, cytology, and bacterial culture test results, a tentative diagnosis of neuroborreliosis was made and oxytetracycline (Liquamycin LA-200, 6.6 mg/kg IV q12h; Zoetis) was initiated. By day 14, the neurologic signs had improved, and the horse was able to walk and graze, although transient signs of narcolepsy developed for an 8-d period before resolving. Narcolepsy has been reported previously in horses with neuroborreliosis. 13 Treatment was transitioned to minocycline (minocycline hydrochloride, 4 mg/kg PO q12h; Aurobindo) as the sole antimicrobial for an additional 60 d. Three months later, the horse was clinically normal and competing at a high level as a show jumper without notable residual neurologic deficits.
Although diagnosed clinically with neuroborreliosis, PCR nucleic acid detection of B. burgdorferi was negative in this horse, prompting us to investigate retrospectively a more sensitive method of detecting B. burgdorferi DNA in equine CSF. A nucleic acid hybrid capture assay was devised and performed on a frozen (−80°C) CSF sample from horse 1 collected on day 3 of hospitalization. We tested by the same method a stored sample of CSF (−80°C) from a 6-y-old Oldenburg gelding (horse 2) from New Jersey that had been collected after 2 mo of antibiotic treatment and resolution of neurologic signs suspected to be the result of neuroborreliosis; the volume of CSF collected early in the disease was insufficient for testing. Hybrid capture testing on the CSF samples was performed as described previously. 3 First, custom capture oligo probes were designed using both the NimbleGen SeqCap EZ (Roche), and SureSelect (Agilent) systems. The probe design covers 95–99.9% of the reference B. burgdorferi genomes, which comprises most of the known Borrelia genomic diversity. 4 Next, total genomic DNA was extracted from the <1-mL CSF sample of each horse. A sequencing library was prepared from the extracted genomic DNA, after which custom capture enrichment of B. burgdorferi DNA was performed with the NimbleGen SeqCap method. The captured DNA fragments were sequenced (HiSeq 4000, Illumina; 150-bp paired-end, 1/12 to 1/14 HiSeq 400 lane per sample), and sequences were analyzed using pipelines adopted from a prior study 3 and already in place at the laboratory of C.M. Fraser (Institute of Genome Sciences, University of Maryland School of Medicine, Baltimore, MD, USA).
Sequence-capture on the CSF sample from horse 1 yielded 10,622,080 HiSeq reads (150-bp reads; 1,603,934,080 bp in total). Over 63% of the reads from the captured DNA could be mapped to B. burgdorferi, confirming the presence of the bacteria in the CSF sample (Fig. 1). In the CSF sample from horse 2, which had been collected after 2 mo of treatment for suspected neuroborreliosis, 7,233,684 reads were sequenced (150-bp reads; 1,092,286,284 bp in total), and BLAST searches (https://blast.ncbi.nlm.nih.gov/Blast.cgi) revealed the absence of Borrelia sequence hits, confirming that this sample was negative for the bacterium. Most sequences in the sample from horse 2 were confirmed to be horse or human hits, likely to be nonspecific pulldown DNA (Fig. 2). No sequence hits for Streptococcus spp. were identified.
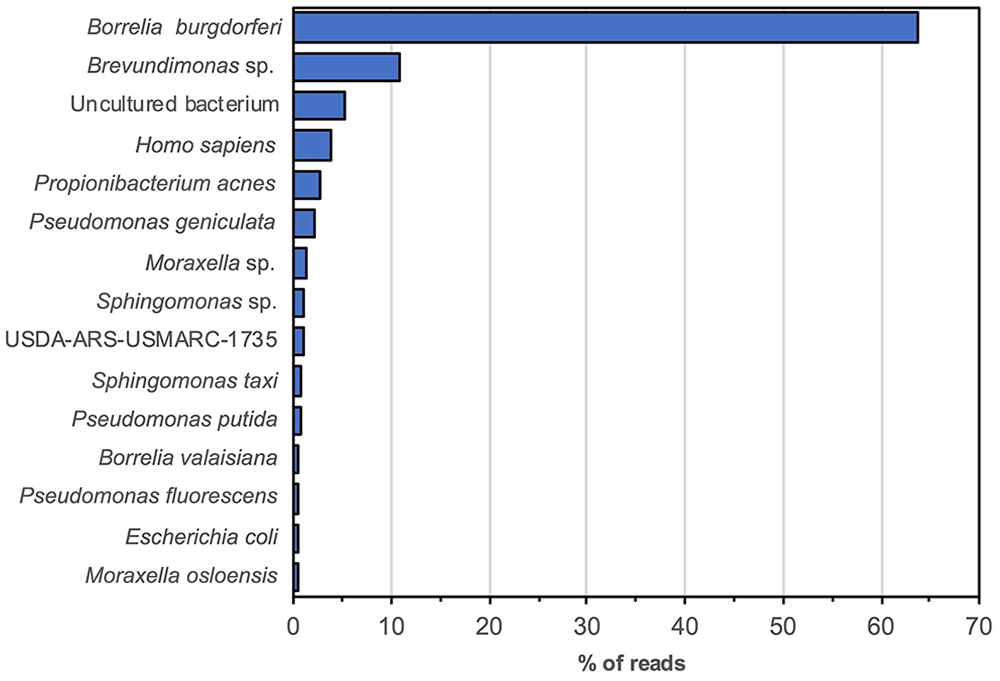
Targeted (hybrid capture enrichment) metagenomic findings from the CSF from horse 1 diagnosed with neuroborreliosis.
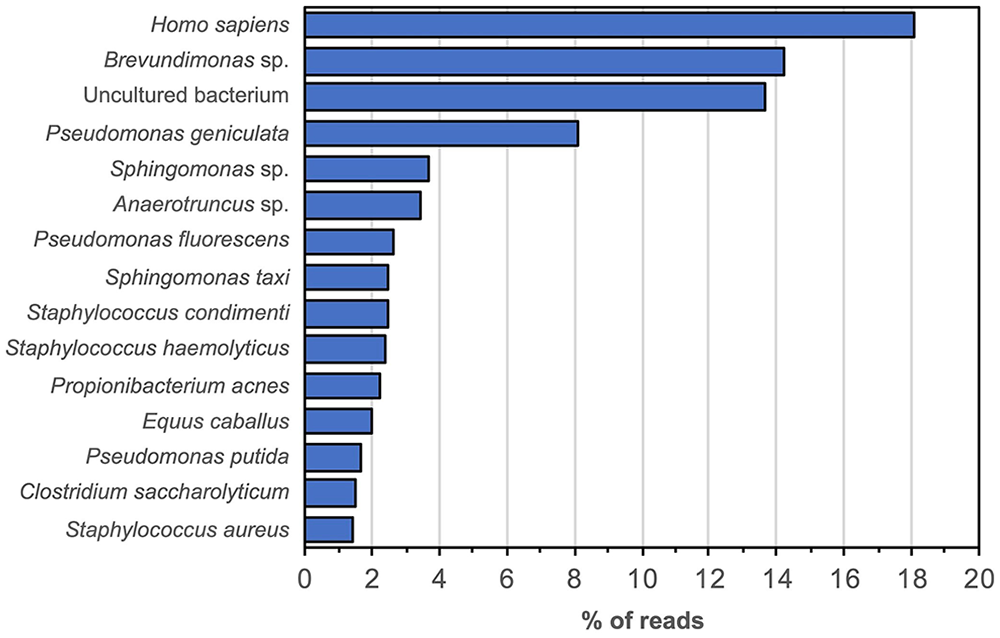
Targeted (hybrid capture enrichment) metagenomic findings from the CSF from horse 2 after 2 mo of treatment for suspected neuroborreliosis.
Given that conventional PCR testing for B. burgdorferi DNA in CSF has low sensitivity, serum and CSF antibody ratios are often used to confirm intrathecal production of B. burgdorferi antibody and document neuroborreliosis in humans.2,10 A similar use of the serum:CSF ELISA antibody ratio has been used successfully in equine practice to detect intrathecal production of antibodies to S. neurona in horses to confirm a diagnosis of equine protozoal myelitis. 15 Unfortunately, serum:CSF antibody ratios using the currently available multiplex ELISA for B. burgdorferi surface antigen antibodies (New York Animal Health Diagnostic Center) have low specificity for equine neuroborreliosis. 8 Possible reasons for the low specificity have been reported and include: dilution of serum but not CSF for the assay; unknown linearity of the assay results; and the marked damage to the blood-brain barrier (BBB) that may occur in horses with neuroborreliosis, increasing transfer of serum antibodies into CSF. 8 When mathematically correcting for the serum dilution, CSF antibody ELISA values 4 times greater than serum values could suggest intrathecal antibody production in horses with an intact BBB. 8 However, using this ratio, the same authors could not separate neuroborreliosis from other equine neurologic diseases. 8
The low serum antibody value for B. burgdorferi in horse 1 does not rule out equine neuroborreliosis given that, in one report, only 6 of 13 horses with autopsy-confirmed neuroborreliosis had serum antibodies to B. burgdorferi.9 The reasons might be early infection prior to production of measurable serum antibody, an immune deficiency, or infection only in privileged sites, such as the eye and CNS.9,13,16
Detecting B. burgdorferi in the CSF can be crucial in the antemortem confirmation of equine neuroborreliosis. Antigen or DNA detection tests such as PCR, fluorescent in situ hybridization, and silver staining of diseased tissue have successfully confirmed neuroborreliosis in deceased horses.7,9,13 Antemortem detection of B. burgdorferi DNA by PCR in ocular fluid has been successful in documenting Lyme uveitis in horses and, in some cases, the organism could be seen on cytologic examination of the aqueous fluid.14,16 As noted, conventional PCR testing of CSF from horses and humans with B. burgdorferi neuroborreliosis has low sensitivity, likely because of low numbers of B. burgdorferi in CSF.2,9,17 Although the specific genomic hybrid capture methodology that we used was able to identify B. burgdorferi in the CSF of a horse with acute onset neurologic signs, finding DNA of an organism in fluid from a diseased animal does not confirm causation of disease. However, in our case, finding B. burgdorferi in the CSF, and the absence of molecular evidence for any other bacterial pathogen, make neuroborreliosis the most likely diagnosis.
One other bacterial rule-out considered in horse 1 was meningitis secondary to sinusitis or treatment thereof. Meningitis following surgery for primary streptococcal sinusitis has been reported in one horse in which surgical complications associated with abnormally high lavage pressure led to convulsions during surgery, followed by streptococcal meningitis postoperatively. 1 The absence of streptococcal DNA in the CSF of horse 1 was helpful in ruling out streptococcal meningitis. The failure to find B. burgdorferi nucleic acid sequences in the CSF from horse 2 does not imply successful treatment for Lyme neuroborreliosis in that horse given that the diagnosis had not been confirmed prior to treatment. Testing of this sample was helpful however, given that it served as a control sample for the B. burgdorferi amplification that we used.
Our results demonstrate that these relatively low-cost, custom solution-based, oligo-capture platforms might be helpful in detecting B. burgdorferi in CSF of horses with neuroborreliosis. The capture kits can be used to quickly and effectively isolate pathogen DNA from host DNA and enrich the targeted genomes of B. burgdorferi in CSF prior to deep sequencing, thereby enhancing detection of nucleotides of interest. In addition, analysis of the sequences could allow evaluation of B. burgdorferi strain variation and functional profiling analyses similar to functional analysis of a complete genome sequence. 6 Next-generation sequencing of the CSF from horses with meningitis of suspected infectious origin would allow testing for additional pathogens.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Disclaimer
Emmanuel F. Mongodin contributed to this work as an employee of the University of Maryland School of Medicine; the views expressed in this manuscript are his own and do not necessarily represent the views of the National Institutes of Health or the United States Government.
Funding
This study was supported in part by NIH grants U19AI110820, AI114009-02 (Emmanuel F. Mongodin, Claire M. Fraser, Steven E. Schutzer).
