Abstract
Common variable immunodeficiency (CVID) is a rare condition in adult horses characterized by hypogammaglobulinemia and increased susceptibility to parasitic and bacterial infections, including recurrent respiratory diseases, septicemia, and meningitis. Lyme disease is often included as a differential diagnosis in CVID horses with signs of meningitis; however, the
A 7-y-old American Quarter Horse mare from the northeastern United States was presented to a referring veterinarian in mid-June 2015 with bilateral mucopurulent ocular discharge and was treated for conjunctivitis. Two days later, the horse was lame on the right forefoot with moderate distal limb edema and a rectal temperature of 38.1°C (normal 37.5–38.6°C). Hoof abscess was suspected, and she was treated appropriately. Although the lameness and ocular discharge resolved within 1 wk of presentation, enlarged submandibular lymph nodes and moderate mucopurulent discharge in the left nostril were noted. Temperature was still within normal range at 37.9°C. Oral examination revealed no significant findings. Bloodwork at this time revealed anemia, lymphopenia, hypoproteinemia, and hypoglobulinemia (Table 1). Fibrinogen was also elevated (7.8 g/L; reference interval: 1.5–4.0 g/L) and would remain persistently elevated for the next 6 mo. A Lyme multiplex assay
19
performed at the New York State Animal Health Diagnostic Center (AHDC, Ithaca, NY) to measure serum antibodies to the outer surface proteins (Osps) of
Hematology values in a CVID horse with neuroborreliosis from the time of presentation (June 2015) and the time of euthanasia (December 2015).
By early July 2015, acute uveitis with an anterior chamber fibrin clot was noted in the left eye. There was still left nasal discharge, although the horse remained afebrile (38.0°C). Blood was submitted for
Upon return from the rehabilitation facility in late July 2015, the horse became febrile (39.0°C), had continued bilateral ocular discharge, and began shifting weight in the forelimbs. Thoracic ultrasound suggested pneumonia. Injections of ceftiofur crystalline-free acid (Zoetis, Kalamazoo, MO) antibiotic and flunixin meglumine (Aspen Veterinary Resources, Liberty, MO) were begun as needed for control of fever. Repeat PCR for
Initial lymphocyte immunophenotyping was performed at Cornell University Equine Immunology Laboratory (Ithaca, NY) in mid-August, and results were consistent with common variable immunodeficiency (CVID). The final peripheral blood lymphocyte phenotyping (Table 2) showed a markedly decreased B-cell concentration measured by flow cytometry with a 1.0% distribution of CD19+ B cells in the lymphocyte-gated area, 0.2% CD21+ B cells, and 0.9% IgM+ B cells. The CD4+ and CD8+ T-cell distributions were normal to slightly increased, and the CD4-to-CD8 ratio was within the low normal reference interval. Serum IgG concentrations measured by immunoturbidimetric assay in August 2015 and December 2015 were markedly decreased; serum IgM concentrations measured by radial immunodiffusion at the same times were within the normal reference interval (Table 3).
Phenotyping of peripheral blood lymphocytes using flow cytometry, December 2015.
Serum immunoglobulin concentrations at initial immunophenotyping (August 2015) and near the time of euthanasia (December 2015).

In late August 2015, the horse exhibited narcolepsy signs such as slow head movements and abrupt lying down on the knees and sides. No seizures were noted. On neurologic examination, there was weak bilateral tail pull; the left pupil was mildly dilated as a result of atropine treatments while at the referral hospital. There was also continued nasal discharge, but no lameness or fever. Oral doxycycline (Wedgewood Pharmacy, Swedesboro, NJ) and tapering doses of injectable flunixin meglumine were started. The horse remained relatively stable over the next 3 mo with monthly examinations and monthly 7-d courses of oral sulfamethoxazole and trimethoprim (Vista Pharmaceuticals, West Orange, NJ). Bloodwork in December showed hypoproteinemia and hypoglobulinemia, as well as markedly elevated gamma-glutamyl transferase activity (Table 1). By January 2016, bilateral ocular discharge was still noted, and the horse was reported to be falling down. Given the potential danger to caretakers and the poor prognosis, the horse was submitted for euthanasia under the owner’s consent. An autopsy was performed on the same day at the anatomic pathology service at the AHDC for complete tissue collection.
On autopsy, gross anatomic findings included moderate serous abdominal effusion, extensive fibrosis of the hepatic capsule and parenchyma, numerous coalescing white hepatic nodules, variably sized pulmonary and thymic white nodules, 2 chronic gastric ulcers, mild bilateral forelimb laminitis, and a chronic sole abscess of the left forelimb. Microscopically, meninges of the spinal cord, cerebellum, and cerebrum were diffusely expanded by marked infiltrates of lymphocytes, histiocytes, fewer neutrophils, and rare plasma cells (Figs. 1, 2). Within the parenchyma of the white and gray matter, the Virchow–Robin spaces of blood vessels had cuffs of similar inflammatory cells, and blood vessels were prominent with endothelial hypertrophy and variable branching. Other significant histologic findings included moderate, chronic lymphocytic hypophysitis and lymphohistiocytic eosinophilic choroid plexitis; thymic lymphoid depletion; absent splenic lymphoid follicles and small periarteriolar lymphoid sheaths; loss of normal architecture and abundant homogeneous acellular eosinophilic material within a mesenteric lymph node interpreted as amyloid-like material; severe myeloid and erythroid hypoplasia of the bone marrow; chronic lymphoplasmacytic portal hepatitis with portal-to-portal and portal-to-central bridging fibrosis and acute lobular hepatocellular necrosis; and chronic mineralized granulomas in the liver, lung, and thymus. Although no metazoan parasites were noted in any of the tissue sections examined, the mineralized granulomas were presumed to be caused by chronic parasitic migration, given that excessive gastrointestinal tract parasitism has been noted in another CVID horse. 3
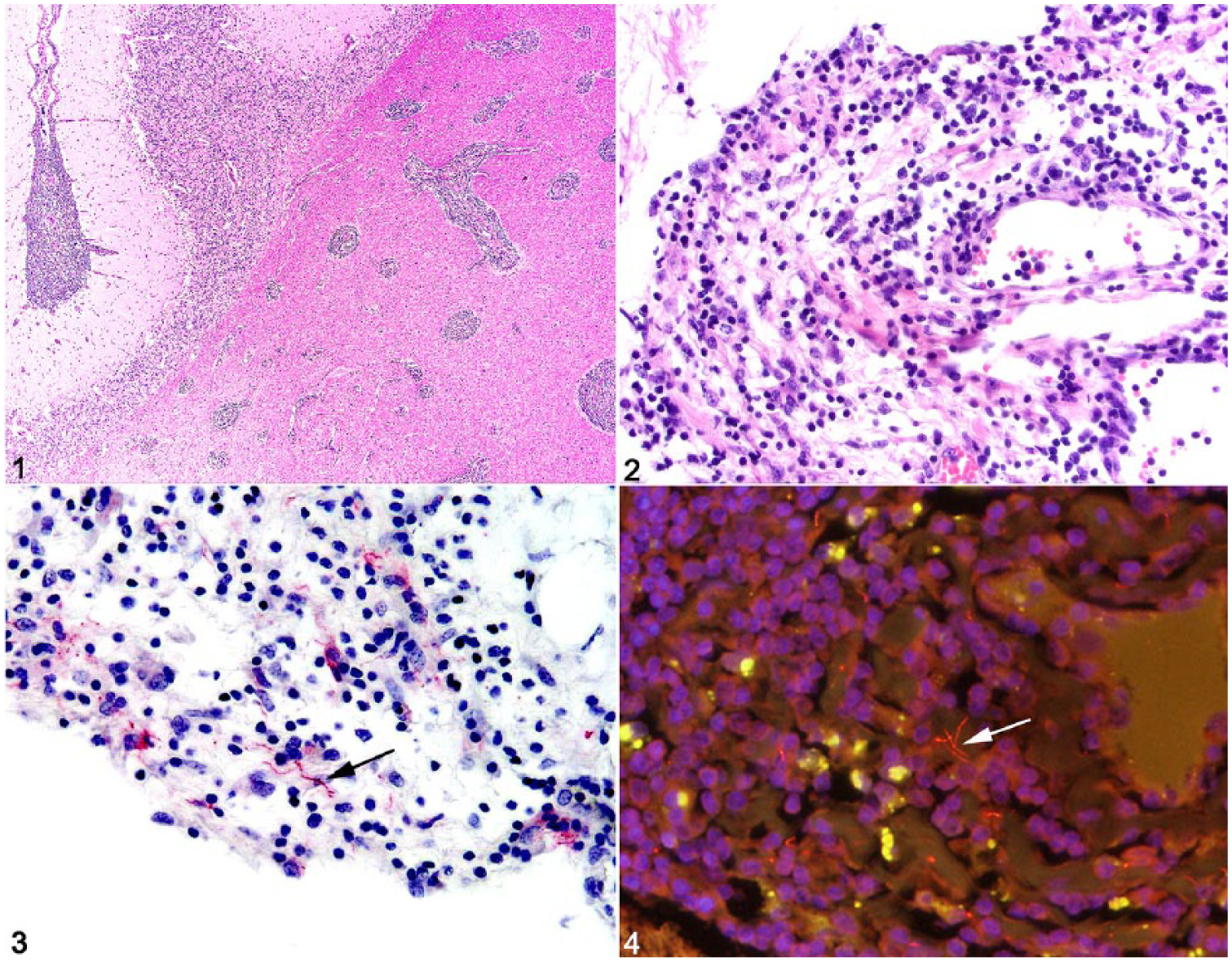
Neuroborreliosis in a horse with common variable immunodeficiency.
The constellation of gross and histologic findings, namely chronic parasitic migration and depletion of lymphoid tissues in the spleen, thymus, and mesenteric lymph node, was consistent with a severely immunocompromised animal with meningoencephalomyelitis. Given the histologic findings and clinical history of uveitis, lymphadenopathy, and, finally, narcolepsy,
CVID is a primary immunodeficiency disease of humans and horses that encompasses a group of heterogeneous disorders characterized by hypogammaglobulinemia and increased susceptibility to bacterial and parasitic infections. Generally, at least 2 isotypes of antibodies are affected, although IgG deficiency alone is also recognized. 9 Human CVID patients have a high frequency of autoimmune and lymphoproliferative disease, and such patients often are seen clinically with recurrent respiratory disease 18 and persistent herpesviral infection. 14 B cells may be present or absent in human patients with CVID; however, more commonly, differentiation into plasma cells is disrupted by an unknown mechanism that results in reduced humoral immunity to common pathogens. 9
In horses, CVID is a rare condition. Relatively few cases have been reported,2,3,6,12,17 though the Equine Immunology Laboratory at Cornell College of Veterinary Medicine (Ithaca, NY) has identified this condition in >50 horses since 2002 and has been actively investigating potential genetic and epigenetic mechanisms of the disease.
16
Age of onset is 2–23 y, with an average of 11.3 y. Like human patients, horses with CVID most often have recurrent infections of the respiratory tract. In addition, persistent bacterial meningitis caused by common skin contaminants such as
Clinical syndromes of
Another histologic feature that supports the diagnosis of CVID, in addition to lymphoid depletion in the mesenteric lymph node, spleen, and thymus, was rare B-cell immunoreactivity within sections of cerebrum with florid inflammation. This finding mirrors the antemortem peripheral blood immunophenotyping that reported <1% B cells of total lymphocytes. Indeed, to date, research on equine CVID focuses on the disruption of B-cell development in bone marrow and has identified decreased mRNA expression and incomplete demethylation of the
A low Lyme antibody concentration should be expected in humoral immunodeficiency, and a negative result should not rule out CVID. Nevertheless, the lack of humoral immunity, the primary host defense mechanism against
Footnotes
Acknowledgements
We thank the staff of the New York State Animal Health Diagnostic Center histology, bacteriology, virology, and molecular diagnostic laboratories at Cornell University for technical assistance, including Drs. Amy Glaser and Laura Goodman for interpretation of and information on the
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
