Abstract
Over a period of 5 years, 10 pure-bred Boxer puppies, 9–16 weeks old, were presented with a history of sudden death and were diagnosed with pyogranulomatous myocarditis. The myocarditis was characterized by a mixed infiltrate composed predominantly of neutrophils and macrophages. In our retrospective study, original case records and archived materials were examined. All dogs were positive for Borrelia burgdorferi on immunohistochemistry (IHC). There was no evidence of infectious agents in formalin-fixed, paraffin-embedded (FFPE) heart tissue sections stained with hematoxylin and eosin, Ziehl–Neelsen, Gram, Grocott methenamine silver, Warthin–Starry, Von Kossa, and Steiner–Chapman stains. IHC for Chlamydia sp., Toxoplasma gondii, Neospora caninum, West Nile virus, and canine parvovirus also yielded a negative result in all dogs. Polymerase chain reaction testing for vector-borne pathogens on heart tissue from 9 of the dogs (1 frozen and 8 FFPE samples) yielded positive results for 1 dog with B. burgdorferi as well as Anaplasma phagocytophilum in another dog. Subsequently, 2 additional cases were found in a French Bulldog and a French Bulldog–Beagle mix that had identical morphology, test results, age, and seasonality to these 10 Boxer dogs. The similarities in the seasonality, signalment of the affected dogs, and the gross and microscopic lesions suggest a common etiology. Positive IHC and morphologic similarities to human Lyme carditis indicate that B. burgdorferi is likely the agent involved. An additional consideration for these cases is the possibility of a breed-specific autoimmune myocarditis or potential predisposition for cardiopathogenic agents in young Boxers.
Myocarditis can occur as part of or in the absence of a disseminated infection with bacteria, fungi, or protozoa. 4 These organisms are easily cultured or seen on microscopic examination. Autoimmune causes of myocarditis have been reported in humans associated with inflammatory myopathies, 15 but a canine equivalent has not been described to our knowledge. However, perinuclear anti-neutrophil cytoplasmic autoantibodies (pANCA) have been detected in the serum of dogs with vector-borne pathogen infections and dogs with immune-mediated hemolytic anemia. 12 The discovery of this potential biomarker brings to light the possibility that there may be an autoimmune or other indirect consequence of vector-borne pathogen infections in dogs.
Infectious causes of myocarditis in dogs include a number of viral, bacterial, protozoal, and fungal pathogens. Canine parvovirus 2 (CPV-2) sporadically causes myocarditis, resulting in sudden death in young dogs under 8 weeks of age. 10 Myocarditis has also been reported in dogs infected with the vector-borne pathogens West Nile virus 3 and Borrelia burgdorferi. 13 Bartonella spp. infections, including B. henselae, can be bacteremic, resulting in myocarditis, but B. vinsonii subsp. berkhoffii more typically causes endocarditis centered on the aortic valve. 9 Canine myocarditis caused by Trypanosoma cruzi infection has been described but occurs exclusively in dogs living in the southern United States (or with a travel history to the southern United States) and neither the parasite nor its vector (Hematophagous triatomines) are known to occur in Minnesota. 2 Neospora caninum is a rare cause of myocarditis in dogs.17,18
Between 2004 and 2009, 10 pure-bred Boxer puppies with a history of sudden death were submitted to the University of Minnesota Veterinary Diagnostic Laboratory (UMVDL; St. Paul, Minnesota) for autopsy. Five of these dogs were reported to have clinical signs of heart failure ranging from minutes to 36 hr before death. The dogs were 9–16 weeks old and lived within 150 miles of St. Paul, Minnesota (Table 1). Eight of the dogs were submitted in the late summer–fall (August 24–October 26) and 2 were submitted in the spring (April 22 and June 15, respectively). There were 4 males, 4 females, and 2 with no sex recorded. Three of the dogs were littermates, but the ancestral relationships of the other 7 dogs are unknown. This is a retrospective study of these 10 cases.
Boxer puppies with myocarditis, 2004–2009.
The 3 dogs were littermates.
All dogs received full postmortem examinations, and tissue samples were collected from all major organs. All diagnostic tests were performed at the UMVDL, unless noted otherwise. Tissue samples were fixed in neutral buffered, 10% formalin solution and routinely processed for paraffin embedding. All formalin-fixed, paraffin-embedded (FFPE) tissues were stained with hematoxylin and eosin (HE). As a result of the presence of myocarditis, additional microsections of FFPE heart tissue were stained with Ziehl–Neelsen, Gram, Grocott methenamine silver, Von Kossa, and Steiner–Chapman stains. Immunohistochemistry (IHC) was performed on FFPE heart tissue using antibodies for B. burgdorferi, Chlamydia sp., Toxoplasma gondii, Neospora caninum, West Nile virus, and canine parvovirus in an attempt to identify infectious agents (Table 2). Because B. burgdorferi antigen is more commonly seen in the kidneys of dogs with Lyme disease, IHC was also performed on paraffin-embedded kidney tissue from the 10 dogs despite the absence of renal lesions. Additionally, IHC for B. burgdorferi was performed on heart tissue from 3 healthy dogs (16–17-week-old German Shepherd Dog, Labrador Retriever, and Labrador Retriever mix) to look for endogenous cross-reactivity, along with heart tissue from 5 dogs diagnosed with myocarditis. These additional cases of myocarditis were in two 7-week-old Cocker Spaniels with canine parvovirus, one 1-week-old Newfoundland with Streptococcus sp. septicemia, and 2 Labrador Retrievers (3 and 4 years old, respectively) with Blastomyces dermatitidis infections.
List of products and sources for the immunohistochemistry performed on formalin-fixed, paraffin-embedded tissue after microsectioning and adherence to a glass slide. All protocols used 1 of 3 peroxidase-based signal amplification kits a with 3-amino-9-ethylcarbazole (AEC) chromagen a and Mayer hematoxylin counterstain.*
ProtK = proteinase K (5-min incubation); DC-TR = decloaking chamber with target retrieval solution a ; H2O2 = hydrogen peroxide h (3%; 15-min incubation); EnV+Rb = EnVision+ a peroxidase-labeled polymer conjugated to goat anti-rabbit immunoglobulins (30–45-min incubation); EnV+Ms = EnVision+ a peroxidase-labeled polymer conjugated to goat anti-mouse immunoglobulins (45-min incubation); LSAB+ = For the LSAB+ kit, a biotin-labeled affinity isolated swine anti-goat, swine anti-rabbit, and swine anti-mouse immunoglobulins is followed by streptavidin conjugated to horseradish peroxidase (30-min incubation for each step).
Polymerase chain reaction (PCR) testing for agents causing canine vector-borne disease was performed at the University of California–Davis, Real-time PCR Research and Diagnostics Core Facility on fresh heart tissue (dog 10) and FFPE tissue (dogs 1, 2, 4–9). The panel includes Anaplasma phagocytophilum, Anaplasma platys, Bartonella spp., B. burgdorferi, Ehrlichia canis, Rickettsia spp., Babesia spp., Mycoplasma haemofelis, and Mycoplasma haemocanis. Additionally, fresh heart tissue from dog 7 was tested for B. burgdorferi alone in a research laboratory at the University of Minnesota. 6 Aerobic culture of heart tissue was performed at the time of autopsy for 5 dogs (dogs 3, 5, 7, 8, and 10). Clinicopathologic data was not available for any of the dogs, but a serum sample from the dam of dogs 4–6 was collected shortly after their deaths. The dam had a B. burgdorferi–specific immunofluorescence antibody titer of >1,280.
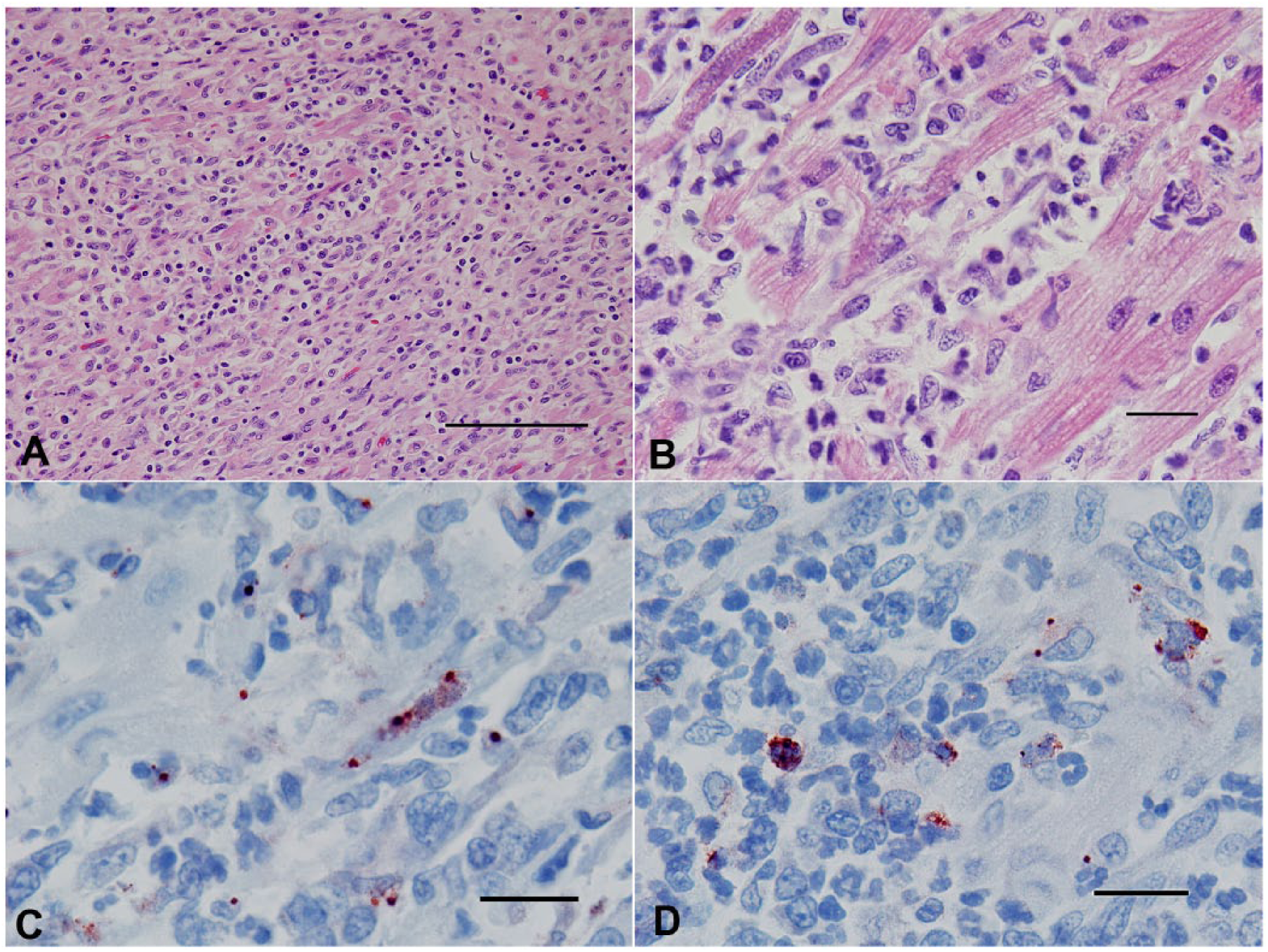
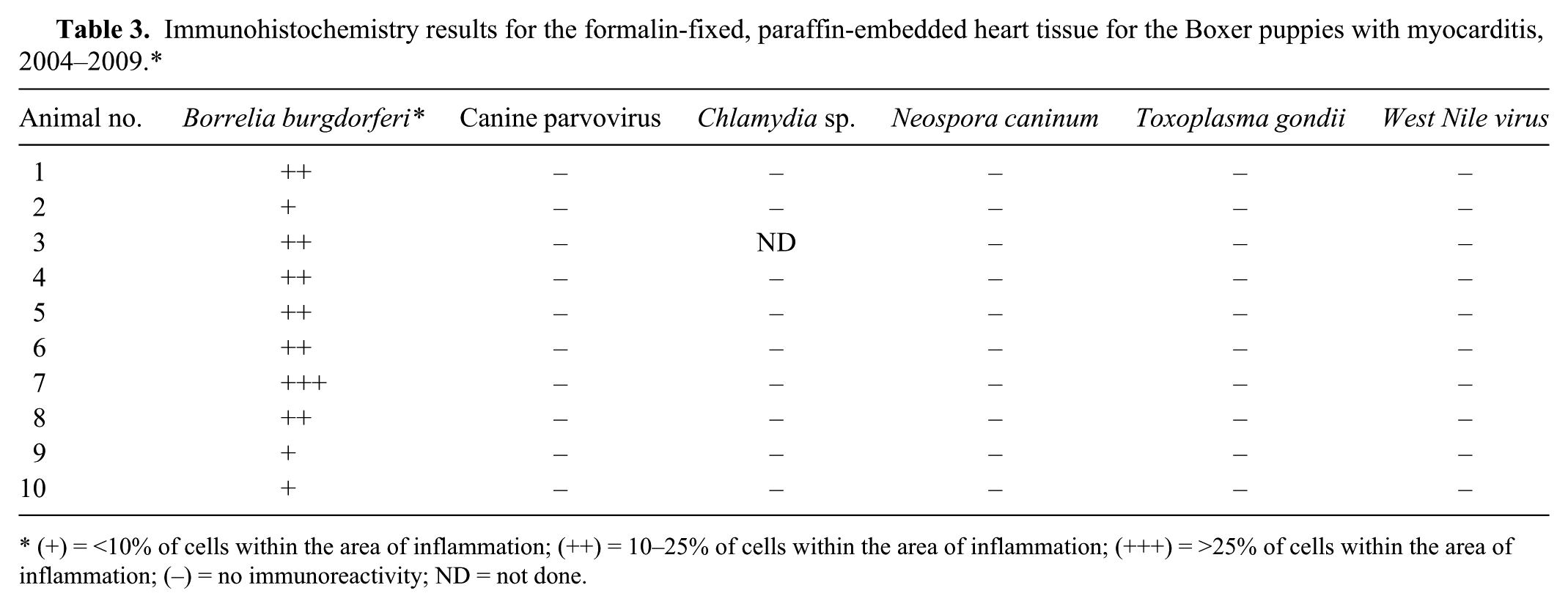
Grossly, all hearts had multifocal to coalescing, white to tan areas within the myocardium that occasionally extended transmurally (Fig. 1). Lesions of the heart were similar in all 10 cases. The dogs had marked myocarditis characterized by multifocal to coalescing separation and replacement of cardiomyocytes by an infiltrate mainly composed of neutrophils and macrophages with scattered clusters of lymphocytes and plasma cells (Fig. 2A, 2B). In the most severely affected areas, there was degeneration, necrosis, and mineralization of the cardiomyocytes adjacent to inflammatory infiltrates admixed with scant fibroblast proliferation. Von Kossa stain demonstrated mild to moderate multifocal calcification in dogs 1 and 7. Infectious agents were absent in HE-stained sections and the employed special stains in all cases.
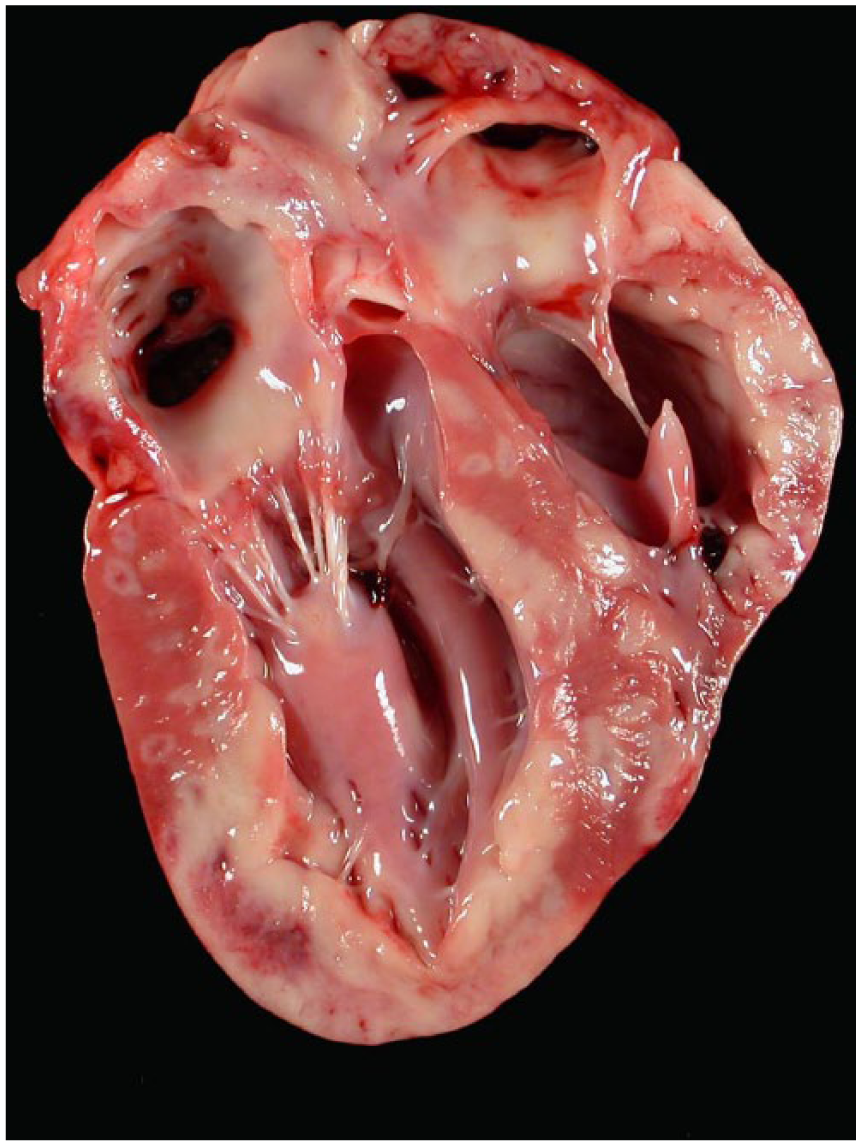
Pyogranulomatous myocarditis in Boxer dog 4. Multifocal to coalescing and occasionally transmural tan areas are present in the sagittal section through atria and ventricles.
Pyogranulomatous myocarditis in Boxer dog 7.
Extracardiac lesions included pulmonary edema (6 dogs) and centrilobular vacuolar hepatocyte degeneration and necrosis (5 dogs), hydrothorax (5 dogs), hydropericardium (2 dogs), and ascites (3 dogs). All of these lesions were attributed to heart failure. Dog 7 also had mild lymphoplasmacytic interstitial nephritis and an ileocecal intussusception without underlying intestinal disease. Dog 10 had a subcapsular hematoma on the liver. Gross lesions were absent in the skeletal muscle.
There was positive immunoreactivity using the B. burgdorferi antigen–specific antibodies in the hearts of all dogs (Table 3). The immunoreactivity rarely extended outside the areas of inflammation, into the unaffected myocardial tissue, and was qualitatively assessed by 2 pathologists (SE Detmer and A Wünschmann) as (+) = <10% of cells within the area of inflammation; (++) = 10–25% of cells within the area of inflammation; (+++) = >25% of cells within the area of inflammation; (–) = no immunoreactivity. The immunoreactivity was predominantly punctate and within the cytoplasm of macrophages (Fig. 2C) but was also noted as granular in the cytoplasm of a small number of cardiomyocytes (Fig. 2D) and as linear staining in degenerating and shrunken cardiomyocytes (5 dogs). All other IHCs were negative for the 10 dogs, including the IHC for B. burgdorferi in the kidneys of these dogs. Aerobic cultures yielded a light growth (1+) of beta-hemolytic Streptococcus sp. and Enterococcus sp. from dog 3 and 7, respectively. Aerobic cultures of heart from dogs 5, 8, and 10 yielded no growth.
Immunohistochemistry results for the formalin-fixed, paraffin-embedded heart tissue for the Boxer puppies with myocarditis, 2004–2009.*
(+) = <10% of cells within the area of inflammation; (++) = 10–25% of cells within the area of inflammation; (+++) = >25% of cells within the area of inflammation; (−) = no immunoreactivity; ND = not done.
There was no immunoreactivity to anti–B. burgdorferi antibodies in the heart tissue for 2 of the healthy dogs or for the 2 dogs infected with canine parvovirus. There was rare, nonspecific staining present for the apparently healthy 4-month-old Labrador Retriever cross that died under anesthesia, the neonate with Streptococcus sp. septicemia, and the 4-year-old Labrador Retriever with disseminated blastomycosis. For the 3-year-old Labrador Retriever with disseminated blastomycosis, there was a focal area of inflammation surrounded by numerous fungal organisms. Within this area, there were small numbers of macrophages with punctate cytoplasmic staining similar to that seen with the 10 Boxer cases.
For the 9 cases that had heart tissue tested for the canine vector-borne pathogens by PCR (1 frozen, 8 FFPE), A. phagocytophilum was detected in 1 sample (dog 4). The only positive result for B. burgdorferi was for the frozen heart tissue from dog 7 that was tested in a research laboratory 6 and could not be confirmed using FFPE heart tissue with the canine vector-borne pathogen testing panel. It is important to note that the canine vector-borne panel has only been validated for ticks and whole blood submitted in a lavender top tube, and there is a caution regarding the use of whole blood for B. burgdorferi quantitative PCR by this laboratory. The positive result for A. phagocytophilum indicates that detection may be possible, but without proper validation of FFPE tissues on this assay, the sensitivity is not known.
The morphology of the cardiac lesions was highly suggestive of an infectious disease process. Furthermore, the similarities in signalment and seasonality for these 10 dogs suggest a common etiology. The absence of infectious organisms in the histologic sections and the absence of inflammation in skeletal muscle and central nervous system made the possibility of infection with N. caninum highly unlikely. A case report of Citrobacter koseri in 12-week-old sibling Boxers details findings similar to our cases. 4 However, Citrobacter sp. was not isolated from the heart or any other tissue in the 5 dogs in which aerobic culture was performed. Additionally, microscopic examination of the heart tissues of these 10 dogs failed to detect bacteria in HE-stained tissue sections and a variety of special stains. In these 10 cases, all of the hearts had cytoplasmic immunoreactivity within macrophages and occasional cardiomyocytes for B. burgdorferi antigen.
The heart tissue of the dog with disseminated blastomycosis had a focal area of inflammation that contained macrophages with cytoplasmic immunoreactivity for B. burgdorferi. This dog was a hunting dog within a region with a high rate of endemic B. burgdorferi infections, and concurrent B. burgdorferi infection cannot be ruled out in this case. The positive controls used for the B. burgdorferi IHC test are kidney tissues from Lyme nephritis cases that were confirmed by antemortem blood tests. For the healthy dog, the septicemia dog, and the other blastomycosis dog, the minimal (a few cells) staining was likely a nonspecific result that can occur with rabbit-derived antibodies. 21 Still, cross-reactivity to an epitope in canine cardiac myosin should be explored because gene sequence homology between the OspA gene of B. burgdorferi and human cardiac myosin has been identified. 20
Eight of the 10 cases occurred in late summer and early fall, whereas 2 cases occurred in the middle to end of spring. According to the Centers for Disease Control and Prevention (CDC), onset of clinical signs of human cases of Lyme disease is most common in June, July, or August (CDC, 2015, http://www.cdc.gov/lyme/stats/). Other information shows that the seasonality of the tick vector in most geographical regions results in cases of Lyme carditis in humans peaking between June and December. 19 The cases reported herein fall into the same seasonal range for human cases. Additionally, a correlation has be shown between canine seroprevalence (C6 protein–based test) and reported human incidence of Lyme disease, 16 indicating that both species are at higher risk of infection in the endemic areas of the Northeast, mid-Atlantic, and upper Midwest regions of the United States.
A conundrum for this study was that the cases initially appeared to only occur in the Boxer breed. Although there is a genetically transmitted ventricular arrhythmia associated with sudden death in mature (7–11 years old) Boxers, 1 there is no susceptibility to inflammatory or degenerative heart disease for immature Boxers reported in the literature. Furthermore, although there initially appeared to be a breed predisposition for Boxers, 2 additional cases were subsequently found. The first case was a 12-week-old French Bulldog from Minnesota (October 2008) and the most recent case was an 11-week-old French Bulldog and Beagle mix from Alberta, Canada (October 2014; Fig. 3A, 3B). Both of these cases were subjected to the same testing as the Boxers and were positive for only B. burgdorferi antigen IHC at UMVDL.
Pyogranulomatous myocarditis in French Bulldog and Beagle mix.
The cardiac manifestations of Lyme disease caused by B. burgdorferi were first described in humans in 1980. 22 The microscopic lesion is usually transmural, and the inflammatory infiltrate is primarily composed of neutrophils and macrophages in the early stage followed by lymphocytes later. 8 Individual cardiomyocyte necrosis, fibrosis, and scattered vasculitis can also be seen. 8 Fatalities caused by Lyme carditis are rare in humans, but the conduction system in the heart is affected, resulting in atrioventricular block diagnosed by electrocardiogram. 19 The gross and microscopic lesions described for human cases of Lyme carditis 23 are similar to the findings in these Boxer puppies and a single case report in an 11-year-old German Shepherd Dog. 13
Experimental infections of dogs with B. burgdorferi caused acute to chronic arthritis, enlarged lymph nodes, and focal skin lesions at the site of tick attachment. Fulminant renal disease has been reported less often, and rarely periarteritis and perineuritis are found.5,7 Lyme carditis has only been reported in dogs in a small number of field cases13,14 where the spirochetes were detected by IHC and modified Dieterle silver stain. Although B. burgdorferi has been detected by PCR in the myocardium and pericardium of 5 out of 6 experimentally infected dogs 9 months after the initial infection, Lyme carditis has not been experimentally reproduced in dogs. 5
Nonetheless, the diagnostic efficacy of PCR for B. burgdorferi has produced inconsistent results in tissue samples, particularly in cases of Lyme nephritis.5,11 Although the negative PCR results contradict the IHC results in our study, the PCR test used was not validated for fresh or fixed heart tissue and the IHC has consistently detected B. burgdorferi antigen in cases of Lyme nephritis at UMVDL. Additionally, underlying Lyme disease could not be ruled out in the blastomycosis case that was IHC positive. For this reason, the IHC results may be considered to be more sensitive in these cases.
In our report, immunoreactivity for B. burgdorferi antigen in combination with the possible seasonality of the lesion suggests that this bacterium or a closely related organism is contributing to the myocarditis in these dogs. Additionally, the morphologic similarities of these cases to human and dog Lyme carditis cases indicate that B. burgdorferi is likely the agent involved. An additional consideration for these cases is the possibility of a breed-specific autoimmune myocarditis or potential predisposition for cardiopathogenic agents in young Boxer dogs. However, additional studies are needed in order to determine the etiology and prevalence of myocarditis in Boxer puppies and to more fully understand the pathogenesis of this condition.
Footnotes
Acknowledgements
We thank Jan Shivers, Ronda Aho, Angela Kimmel, Laurie Landgren, and Michelle Nelson for sharing their technical assistance and expertise.
Authors’ contributions
SE Detmer contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; and critically revised the manuscript. M Bouljihad and A Armien contributed to acquisition and interpretation of data, and critically revised the manuscript. DW Hayden and A Wünschmann contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and critically revised the manuscript. JM Schefers contributed to acquisition of data. All authors gave final approval, and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by the University of Minnesota Veterinary Diagnostic Laboratory.
a.
Dako Denmark A/S, Glostrup, Denmark.
b.
US Biologicals, Swampscott, MA.
c.
Custom Monoclonals International, West Sacramento, CA.
d.
Biodesign/Meridian Life Science Inc., Saco, ME.
e.
VMRD Inc., Pullman, WA.
f.
LabVision, Kalamazoo, MI.
g.
BioReliance, Rockville, MD.
h.
Fisher Scientific, Pittsburg, PA.
