Abstract
Meningioangiomatosis (MA) is a rare proliferative meningovascular entity that has been described mainly in humans and dogs. Here we describe MA in a 13-y-old spayed female domestic shorthaired cat that died 5 d after acute change in behavior, open-mouth breathing, seizures, hyperthermia, and inability to walk. On MRI, the lesion appeared predominantly as extraparenchymal hemorrhage. Autopsy changes consisted of a dark-red, hemorrhagic plaque that expanded the leptomeninges and outer neuroparenchyma of the right piriform and temporal telencephalic lobes, chalky white nodules in the peripancreatic fat, and yellow fluid in the abdomen. Histologically, the lesion in the brain consisted of leptomeningeal thickening by spindle cells that effaced the subarachnoid spaces and extended perivascularly into the underlying cerebral cortex. Spindle cells were arranged as streams or whorls around blood vessels, and had slender eosinophilic cytoplasm and elongated nuclei with coarsely stippled chromatin and 1 or 2 distinct nucleoli. There was extensive hemorrhage, clusters of hemosiderin-laden macrophages, and mineralization throughout. Spindle cells had positive immunolabeling for vimentin. A striking MRI and gross feature in our case was the extensive hemorrhage associated with the MA lesion. Additional findings included suppurative pancreatitis with peritonitis and supraspinatus myonecrosis.
Meningioangiomatosis (MA) is a rare and poorly understood proliferative lesion of the leptomeninges and superficial neuroparenchyma that has been described mainly in humans and dogs,2,5,6,9,12,13,17,18 with additional single case reports in a horse, 4 cow, 8 and mouse. 1 MA is a poorly understood lesion.2,19 Although developmental and dysplastic mechanisms have been proposed, the pathophysiology and histogenesis of MA remain widely elusive in human and veterinary medicine.10,19 The characteristic lesion of MA consists of a proliferation of meningothelial and spindle cells (presumably meningeal fibroblasts) that efface the subarachnoid spaces and typically extend along the perivascular spaces into the outer encephalic or spinal cord neuroparenchyma.2,6,15
MA was first described in humans in 1915 and has been reported in nearly 200 patients.15,19 Lesions affect mainly children and young male adults, and occur sporadically or in association with meningiomas, focal cortical dysplasia, or neurofibromatosis type 2.15,19 The most frequent clinical sign in patients with MA is epileptic seizure, but cases can be clinically silent. 15 Diagnostic imaging features are often nonspecific and difficult to recognize because of the subtle and variable nature of most MA lesions.15,19 For these reasons, diagnostic confirmation typically relies on histologic evaluation of surgical biopsy or autopsy samples. 15 MA typically affects the frontal, temporal, parietal, and occipital telencephalic lobes, with no spinal cord involvement described to date. 19 Although postoperative deterioration and seizures persist in some cases, most patients respond well to surgical resection of lesions, with recurrence in <10% of cases. 15
In veterinary medicine, MA was first described in a dog in 1988, 18 with 12 canine cases reported since then.2,5,6,9,12,13,17,18 Based on limited evidence from these cases, MA appears to affect younger dogs <1-y-old; no apparent breed or sex predisposition has been observed. Clinical signs vary according to the anatomic location of the lesions, with affected dogs displaying seizures and variable neurologic deficits in postural reactions and gait.2,5,6,9,12,13,17,18 A presumptive clinical diagnosis is challenging given that too few cases of MA have been described to develop characteristic imaging findings. MRI typically reveals an extraparenchymal lesion that is primarily T2-hyperintense with focal areas of hypointensity, and T1-hypointense with variable degrees of contrast enhancement. 9 A predilection for the brainstem has been proposed in dogs based on the neurolocalization of lesions in 6 published cases. 2 However, the number of MA diagnoses in dogs is too small to draw conclusions about possible clinical disease or anatomic pathology patterns. In addition, lesions can also occur in other neuroanatomic locations, including telencephalon, cerebellum, and spinal cord.2,5,6,9,12,13,17,18 As reported in humans, MA has been reported concomitantly with a meningioma in a dog. 5
A search of Google, PubMed, CAB Direct, Web of Science, and Scopus for cases of feline MA retrieved no results, suggesting that no descriptions of this condition have been reported in cats. Here we describe the clinical signs, image findings, neuropathology, and immunohistochemistry (IHC) of a case of encephalic MA in a 13-y-old spayed female domestic shorthaired cat that was examined because of an acute change in behavior.
During physical examination, the cat appeared anxious and was open-mouth breathing, tachycardic (280 bpm) and hyperthermic (42.1°C; 107.9°F). Facial twitching progressed to a generalized seizure, which ceased after administration of midazolam (0.3 mg/kg IV; Almaject). On neurologic exam, the cat was obtunded and nonambulatory. Postural reactions were absent in all 4 limbs. Spinal reflexes were normal. Cranial nerve evaluation revealed an absent menace response and absent response to noxious stimulation of the nasal mucosa. Based on the neurologic examination findings combined with the occurrence of a seizure, the anatomic diagnosis was consistent with a multifocal disorder affecting the prosencephalon and a focal or diffuse lesion affecting the C1–C5 spinal cord segments. CBC and chemistry profile revealed neutrophilic leukocytosis (WBC: 23.1 × 109/L, RI: 4.6–15.7 × 109 /L; neutrophils: 21.5 × 109/L, RI: 1.4–8.2 × 109/L), hypernatremia (168 mmol/L; RI: 148–153 mmol/L), hyperkalemia (9.7 mmol/L; RI: 3.4–4.7 mmol/L), hyperglycemia (10.5 mmol/L; RI: 3.5–7.4 mmol/L), hyperlactatemia (19.4 mmol/L; RI: 0.9–3.9 mmol/L), and azotemia (creatinine: 194 µmol/L, RI: 71–159 µmol/L; urea: 12.8 mmol/L, RI: 5.3–10.3 mmol/L). Venous blood gas analysis revealed respiratory acidosis with minimal metabolic compensation (pH 7.2, RI: 7.28–7.46; pCO2 7.6 kPa, RI: 2.8–5.2 kPa; HCO3 22.8 mmol/L, RI: 13.0–22.1 mmol/L). Urinalysis revealed pigmenturia and proteinuria with urine sediment containing <10 RBCs/hpf. Serum creatine kinase was 14.8 µkat/L (RI: 1.4–6.2 µkat/L). Thoracic radiographs were normal. Abdominal ultrasound revealed mild peritoneal effusion.
The cat was treated with IV fluids (30 mL/kg bolus followed by 60 mL/kg/d as a constant rate infusion), calcium gluconate (50 mg/kg IV slowly; Fresenius Kabi), dextrose solution (0.5 mL/kg IV; Hospira), and was provided supplemental oxygen (FiO2 = 40%) via an oxygen cage. After 24 h of IV fluid therapy, the electrolytes and venous blood gas analysis normalized, and the azotemia resolved. Despite the correction of biochemical abnormalities, the cat continued to have episodes of right-sided facial twitching and ptyalism, which were interpreted as focal seizures. Phenobarbital (2 mg/kg IV q12h; Westward Pharmaceuticals) was administered.
Approximately 48 h after presentation, MRI of the head was performed (3.0 T Skyra; Siemens) with an extremity coil. Multiplanar images were acquired using the following sequences: T2-weighted (T2W), T2W fluid attenuated inversion recovery (T2W FLAIR), susceptibility weighted (SWI), and T1W FLAIR. Additional T1W FLAIR images were obtained following IV administration of gadodiamide contrast medium (0.2 mL/kg; Omniscan, GE Healthcare). A right-sided, ill-defined, plaque-like extraparenchymal lesion conformed to the contour of the overlying cerebral cortex. The lesion extended from the temporal lobe ventrally to the piriform lobe of the right telencephalic hemisphere. The lesion was markedly hypointense on T2W, T2-FLAIR, and SWI sequences. On T1W images, the lesion was mildly hypointense and surrounded by focal areas of mild hyperintensity. Poorly delineated areas of contrast enhancement were present peripheral to the hypointense lesion. The predominant imaging features were consistent with hemorrhage. The right piriform lobe, internal capsule, and corona radiata were T2-hyperintense, consistent with perilesional edema (Fig. 1). The longus capitis and epaxial muscles of the cranial aspect of the neck displayed heterogeneous contrast enhancement, consistent with rhabdomyolysis.
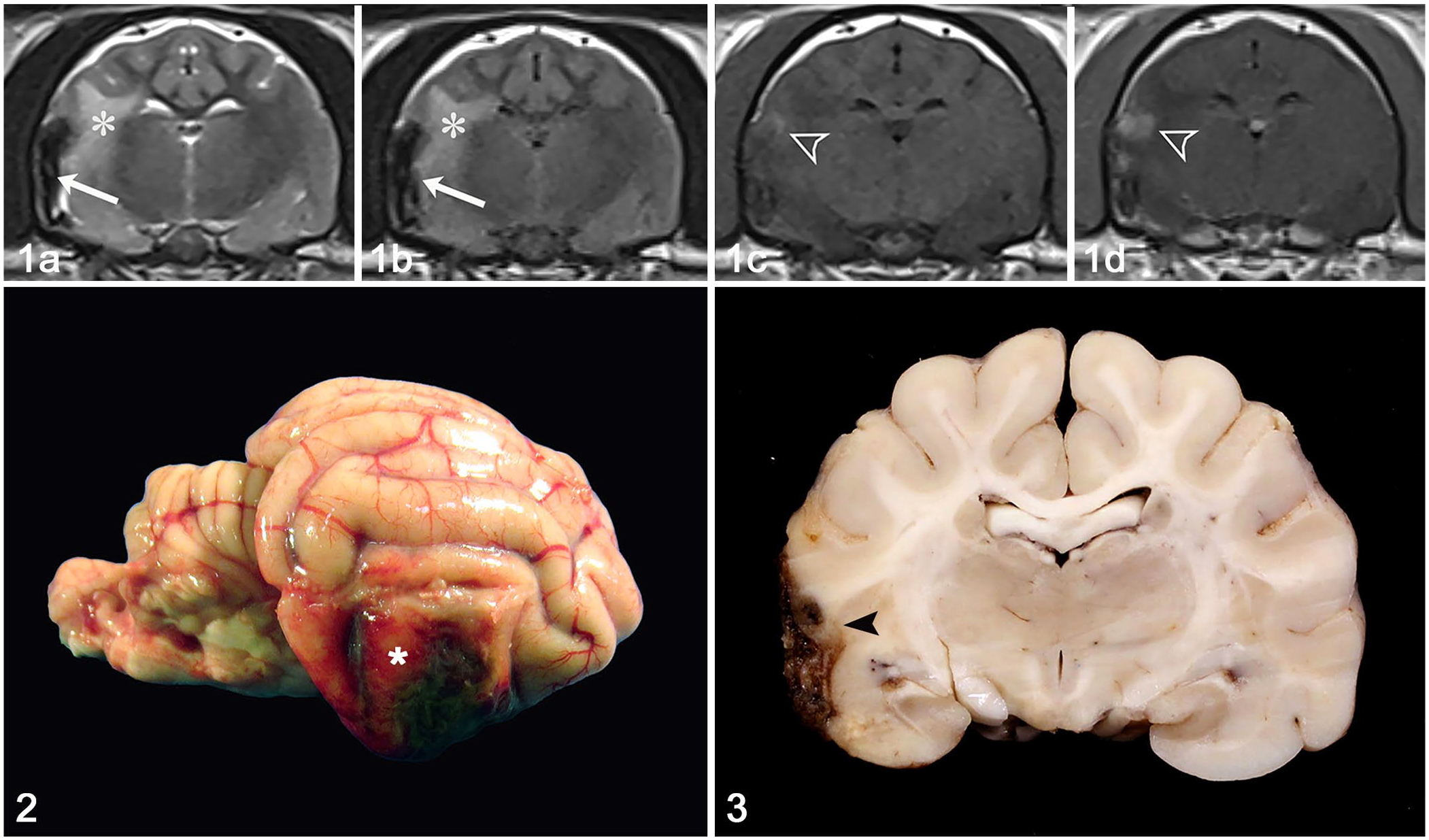
Encephalic meningioangiomatosis in a cat.
Following MRI, the cat was administered dexamethasone (0.1 mg/kg IV q24h). Over the subsequent 5 d, mentation improved, and the cranial nerve deficits and focal seizures resolved. However, the cat remained nonambulatory and tetraparetic. On the 5th day following presentation, during manual expression of the bladder, the cat experienced cardiopulmonary arrest and died.
Autopsy revealed a poorly demarcated, dark-red area of hemorrhage affecting the right piriform and temporal lobes of the telencephalon. The lesion was mildly elevated, firm, and extended to the ventral portion of the right occipital lobe (Fig. 2). Serial sections of the brain after fixation in 10% neutral-buffered formalin revealed that the lesion formed a dark red plaque that expanded the leptomeninges and extended into the cerebral cortex (Fig. 3). Other autopsy findings included poorly demarcated, nodular, chalky white areas in the peripancreatic fat and a small amount of yellow fluid in the abdomen. Routine tissue samples were collected, fixed in 10% neutral-buffered formalin, processed routinely for histology, and stained with H&E.
Histologically, the lesion in the brain consisted of extensive leptomeningeal thickening by spindle cells and vascular spaces that effaced the subarachnoid space and extended perivascularly into the underlying cerebral cortex (Fig. 4). The spindle cells were arranged as streams or whorls around blood vessels (Fig. 5). Within the invaded neuroparenchyma, remnants of entrapped gray matter were observed throughout (Fig. 6). Proliferated spindle cells had a moderate amount of slender eosinophilic cytoplasm with indistinct cell margins and elongated nuclei with coarsely stippled chromatin and 1 or 2 distinct nucleoli (Fig. 7). No mitoses were observed in 2.37 mm2 (10 FN22/40× fields). Vascular spaces were partially filled with luminal erythrocytes, lined by one layer of endothelial cells, and often surrounded by extensive areas of hemorrhage or clusters of hemosiderin-laden macrophages (confirmed by Perl iron stain), especially within the leptomeninges. There were also foci of mineralization throughout. The adjacent neuroparenchyma had extensive areas of rarefaction (edema) with astrogliosis and microgliosis. Additional findings included areas of skeletal myonecrosis (supraspinatus muscles) and suppurative pancreatitis with peritonitis.
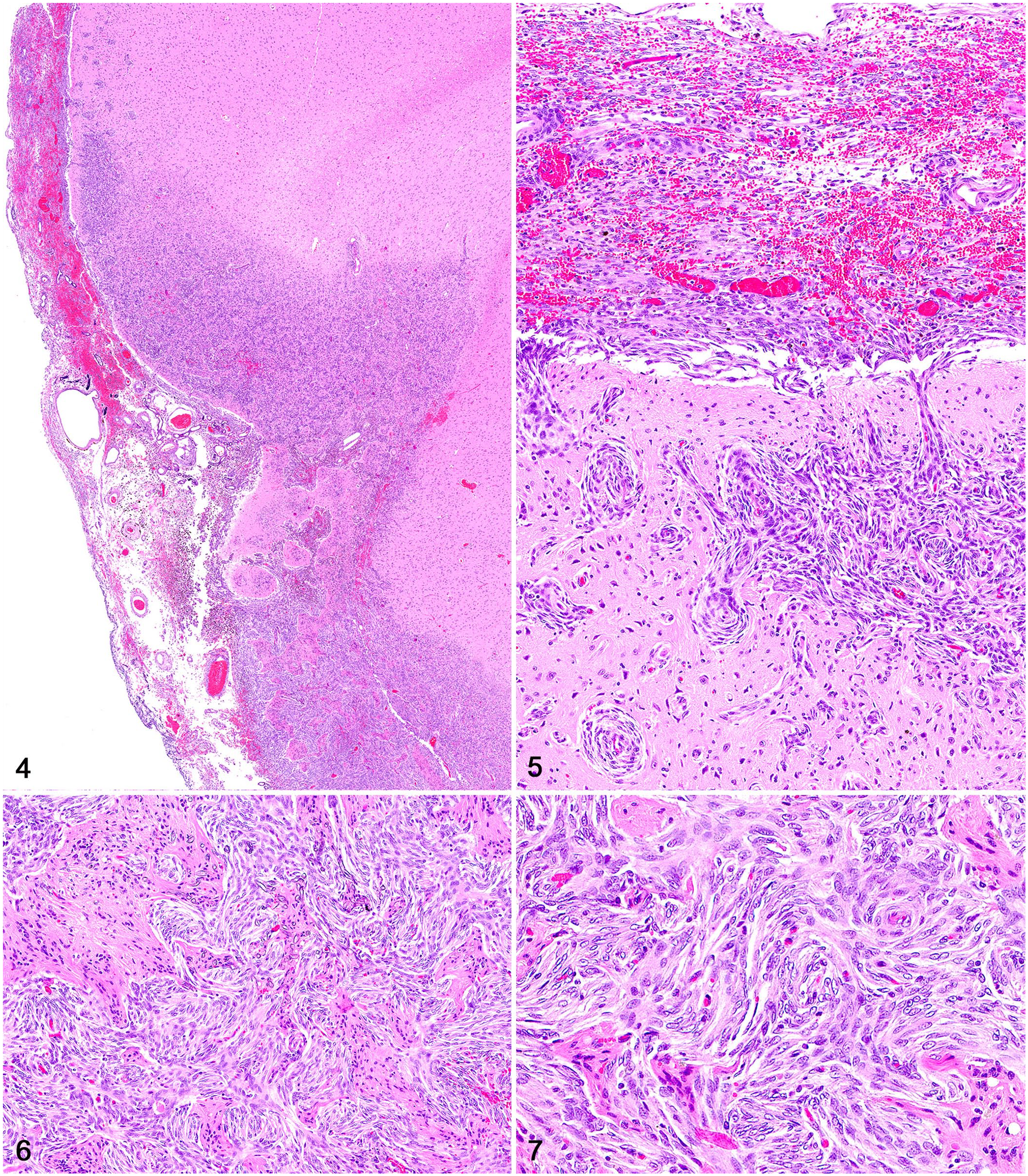
Encephalic meningioangiomatosis in a cat.
IHC for vimentin (mouse monoclonal, 1:3,000 dilution, 60 min; BioGenex), e-cadherin (mouse monoclonal, 1:500 dilution, 60 min; BD Biosciences), smooth muscle actin (SMA; mouse monoclonal, 1:1,500 dilution, 60 min; Dako), factor VIII–related antigen (FVIIIRA; rabbit polyclonal, ready-to-use, 60 min; Cell Marque), S100 (mouse monoclonal, ready-to-use, 30 min; Cell Marque), glial fibrillary acidic protein (GFAP; mouse monoclonal, 1:4,000 dilution, 60 min; BioGenex), oligodendrocyte lineage transcription factor 2 (OLIG2; rabbit monoclonal, 1:400 dilution, 60 min; GeneTex), melan A (mouse monoclonal, 1:30 dilution, 90 min; Dako), and PNL2 (mouse monoclonal, 1:100 dilution, 60 min; Cell Marque) was performed to characterize the brain lesion. Spindle cells had strong, diffuse cytoplasmic immunolabeling for vimentin (Suppl. Fig. 1) and patchy, moderate immunolabeling for e-cadherin (Suppl. Fig. 2). Strong cytoplasmic SMA immunolabeling was detected in vascular myocytes (Suppl. Fig. 3), and strong membranous immunolabeling for FVIIIRA was detected in endothelial cells (Suppl. Fig. 4) throughout the lesion. Spindle cells were negative for all other immunohistochemical stains.
Although the gross neuropathology features in our case were not specific, histologic findings were characteristic of MA and allowed for a diagnosis on routine examination.2,15 Histologically, the main differential diagnosis for MA should be meningioma.2,9 However, the meningovascular nature of the lesion with characteristic subarachnoid and neuroparenchymal arrangement of spindle cells around blood vessels is not typical of meningioma, and should suffice for a histologic diagnosis of MA. 16 Although IHC has limited diagnostic value in human and veterinary cases,10,15 the immunolabeling profile in our case was also consistent with cases of MA described previously in humans and dogs. 5 The robust immunolabeling for vimentin has been reported consistently in MA, supporting a mesenchymal origin for the proliferated spindle cells. Patchy cytoplasmic immunolabeling for e-cadherin was also observed in our case, which is supportive of a meningothelial component in the lesion, corroborating the complex histogenesis of MA. SMA immunolabeling has been detected in subsets of canine cases, suggesting myofibroblastic differentiation within the lesion. 2 In our case, immunolabeling for SMA was evident primarily in perivascular myocytes. 2 FVIIIRA was detected in endothelial cells, as reported in other cases. 2 The arrangement of spindle cells around blood vessels in our case was also somewhat similar to that reported in a case of angiocentric astrocytoma in a cat. 14 However, angiocentric astrocytoma should be primarily intraparenchymal and immunopositive for glial immunomarkers (OLIG2, GFAP), which were negative in our case.
MA consists typically of white leptomeningeal plaques that extend into the outer neuroparenchyma.2,15 A striking MRI and gross feature in our case was the extensive hemorrhage associated with the lesion, which was interpreted as chronic based on the imaging features and the intralesional hemosiderin-laden macrophages. Hemorrhage has been reported in ~1% of human MA cases, 15 and hemosiderin-laden macrophages have been reported in a dog. 18 Because too few cases have been published in veterinary medicine, it remains undetermined whether hemorrhage could be a common feature of MA.
Although considered a benign lesion, the prognosis for MA in dogs is poor because of the local infiltration of spindle cells into the neuroparenchyma, as observed in our case, and the challenges associated with surgical resection of the leptomeningeal plaque. 2 The local invasion into the neuroparenchyma can be destructive and induce gliosis, neuroaxonal degeneration and necrosis, and gray or white matter necrosis. 10 In our case, extensive astrogliosis and microgliosis was present within the surrounding neuroparenchyma.
Other relevant findings in our case included hyperthermia, acute pancreatitis with peritonitis, and rhabdomyolysis with pigmenturia. In humans, acute pancreatitis has been associated with seizures and subarachnoid hemorrhage.7,11 Seizures may cause either increased intraduodenal pressure and duodenal content reflux into the pancreatic duct, or hypoxia and ischemia with acinar cell injury and consequent pancreatitis. 11
Rhabdomyolysis and subsequent myoglobinuria has also been reported as a complication of extreme hyperthermia. 3 In our case, red pigmenturia, in the absence of hematuria, hemolysis, and hemoglobinemia, was consistent with myoglobinuria. It is likely that the extreme hyperthermia at presentation resulted in rhabdomyolysis.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387221110912 – Supplemental material for Encephalic meningioangiomatosis in a cat
Supplemental material, sj-pdf-1-vdi-10.1177_10406387221110912 for Encephalic meningioangiomatosis in a cat by Megan P. Corbett, Brianna L. Kopec, Marc Kent and Daniel R. Rissi in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
