Abstract
Gliomas are common primary central nervous system neoplasms of dogs and cats, but atypical glioma subtypes are rare. Herein we report an angiocentric astrocytoma in a 15-y-old spayed female domestic shorthaired cat that was euthanized after therapy-resistant seizures. Gross anatomic changes consisted of swelling of the rostral leptomeninges over the olfactory bulbs and rostral telencephalon. Histologically, polygonal-to-elongate atypical neoplastic cells were arranged along perivascular spaces within these areas. Neoplastic cells were positive for glial fibrillary acidic protein, S100 protein, and vimentin. Ultrastructurally, round-to-elongate neoplastic cells emitting long processes with aggregates of intermediary filaments expanded and occupied the spaces between the vascular basement membrane and the glia limitans; nuclei had marginal and central heterochromatin. Tight junctions connected the plasma membrane of neighboring cells. The cell morphology, immunohistochemistry, and ultrastructural findings were consistent with an astrocytoma; the exclusive perivascular arrangement of neoplastic cells with no parenchymal mass warranted the diagnosis of angiocentric astrocytoma.
Gliomas are neoplasms (astrocytoma, oligodendroglioma, and ependymoma) that arise from glial cells in the central nervous system (CNS).15,20 Gliomas are one of the most frequent primary CNS neoplasms of dogs and cats.20,24 Prevalence data varies according to different studies and institutions, but astrocytoma is the most frequent type of glioma reported in cats.2,3,5,9,15–18,22–24 Tumors typically occur as a solid intraparenchymal mass within the telencephalon, and less frequently brainstem or cerebellum and spinal cord.2,9,17,24 Although diagnostic confirmation via histology and immunohistochemistry (IHC) can be relatively simple for a well-differentiated astrocytoma,15,24 rare and atypical tumor subtypes, such as subependymal giant cell astrocytoma,3,24 pilocytic astrocytoma, 16 and granular cell astrocytoma 9 can make a final diagnosis more challenging. Herein we describe an astrocytoma exhibiting an angiocentric growth pattern with no distinct parenchymal mass in a cat. To our knowledge, no such glioma subtype has been described in the veterinary medical literature to date.
A 15-y-old spayed female domestic shorthaired cat was referred to a veterinary clinic after a 3-mo history of therapy-resistant seizures. Treatment with phenobarbital was unsuccessful, and clinical signs progressed. Euthanasia was elected because of the poor prognosis, and the cat was submitted for autopsy. Gross anatomic changes were restricted to the brain. The leptomeninges over both olfactory bulbs were swollen, light-gray to tan, and firm, with small hemorrhagic areas (Fig. 1). Similar changes were also present within the olfactory bulbs and neuroparenchyma at the level of the olfactory cortex, cingulate gyri, septal nuclei, and right globus pallidus (Fig. 2). Representative tissue sections (including brain and spinal cord) were collected, fixed in 10% buffered formalin, processed routinely for histology, and stained with hematoxylin and eosin. Histologically, dense aggregates of polygonal-to-elongate neoplastic cells were arranged along perivascular spaces within the rostral leptomeninges, as well as the olfactory cortex, cingulate gyri, septal nuclei, right globus pallidus and thalamus, and to a lesser degree the mesencephalon near the crus cerebri (Fig. 3). Neoplastic cells were occasionally oriented perpendicularly to the blood vessels and formed palisading arrays (Fig. 4). Neoplastic cells had a moderate amount of eosinophilic, homogeneous or vacuolated cytoplasm with indistinct margins, and round-to-elongate nuclei with dense or finely stippled chromatin and 1–2 nucleoli. There were 2 mitoses in ten 400× fields. Anisocytosis and anisokaryosis were moderate. Multiple parenchymal foci of hemorrhage with accumulations of hemosiderin-laden macrophages and peritumoral areas of gliosis with occasional necrotic foci partially filled with foamy macrophages (gitter cells) were also present, and were most likely caused by compression of the neuroparenchyma by clusters of neoplastic cells with subsequent ischemia.
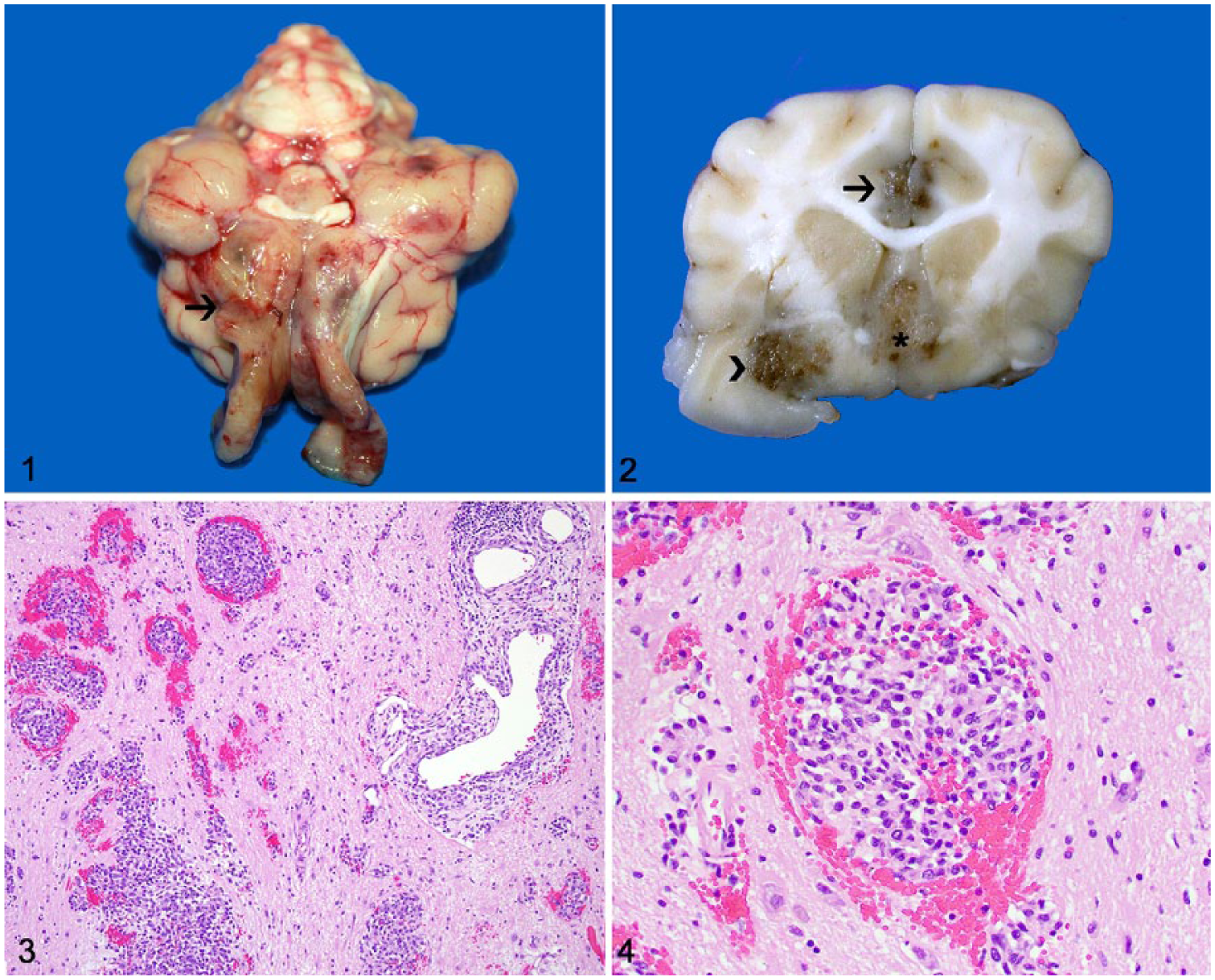
Brain of a cat with angiocentric astrocytoma.
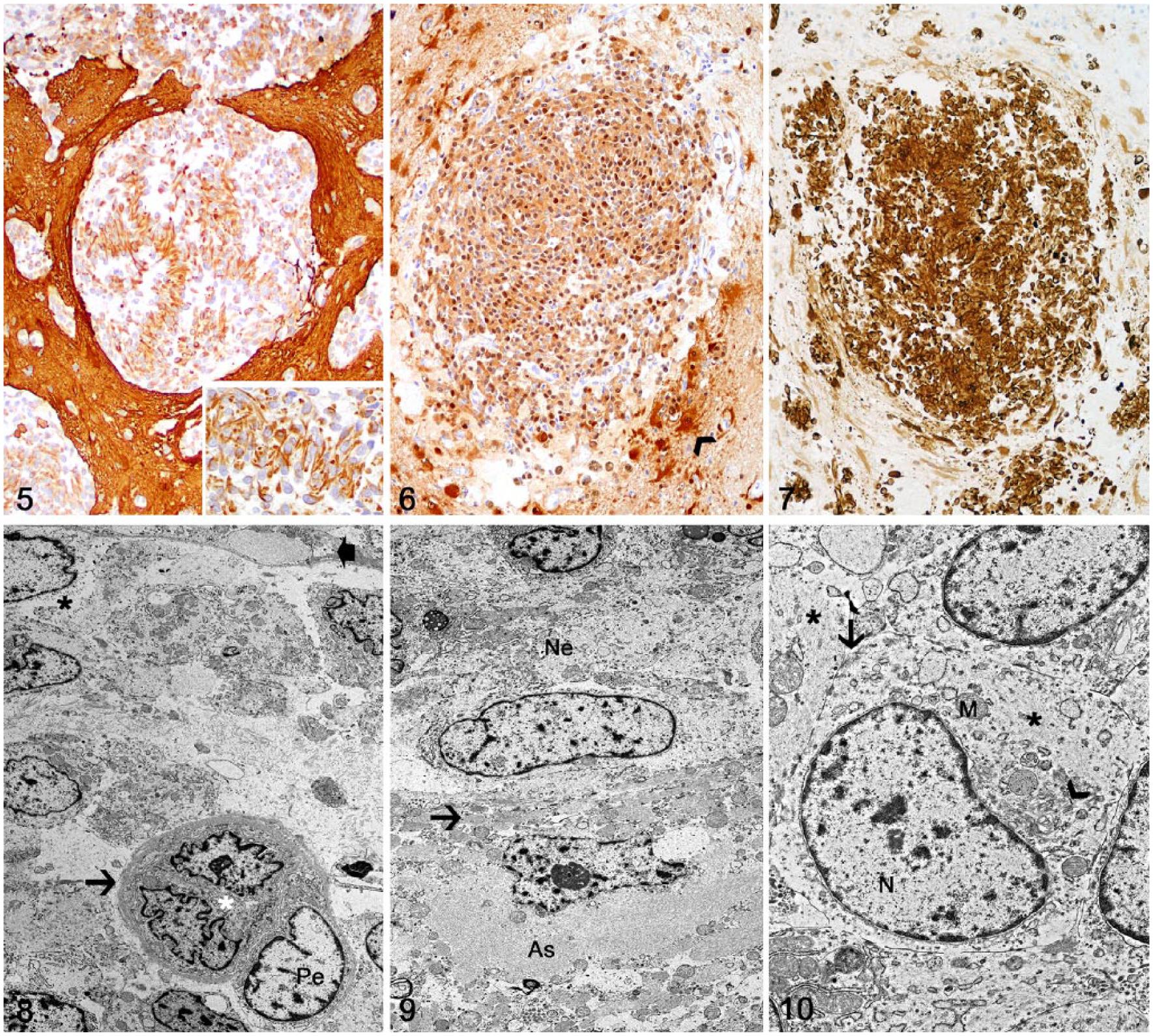
Tissue sections were subjected to an IHC panel for further tumor characterization (Table 1). Neoplastic cells had robust immunolabeling for glial fibrillary acidic protein (GFAP; Fig. 5), S100 protein (Fig. 6), and vimentin (Fig. 7). Nuclear Ki-67 immunolabeling was strong but scattered (~1% of neoplastic cells). Endothelial cells lining small capillaries surrounded by neoplastic cells had strong membranous immunolabeling for factor VIII–related antigen (FVIIIRA; Supplementary Fig. 1) and smooth muscle actin (SMA; Supplementary Fig. 2). Oligodendrocyte lineage transcription factor 2, neurofilament, neuron-specific enolase, synaptophysin, e-cadherin, CD18, CD3, CD20, ionized calcium-binding adapter molecule 1, and pan-cytokeratin (AE1/AE3) IHC were negative.
Immunohistochemical characterization of an angiocentric astrocytoma in a cat.
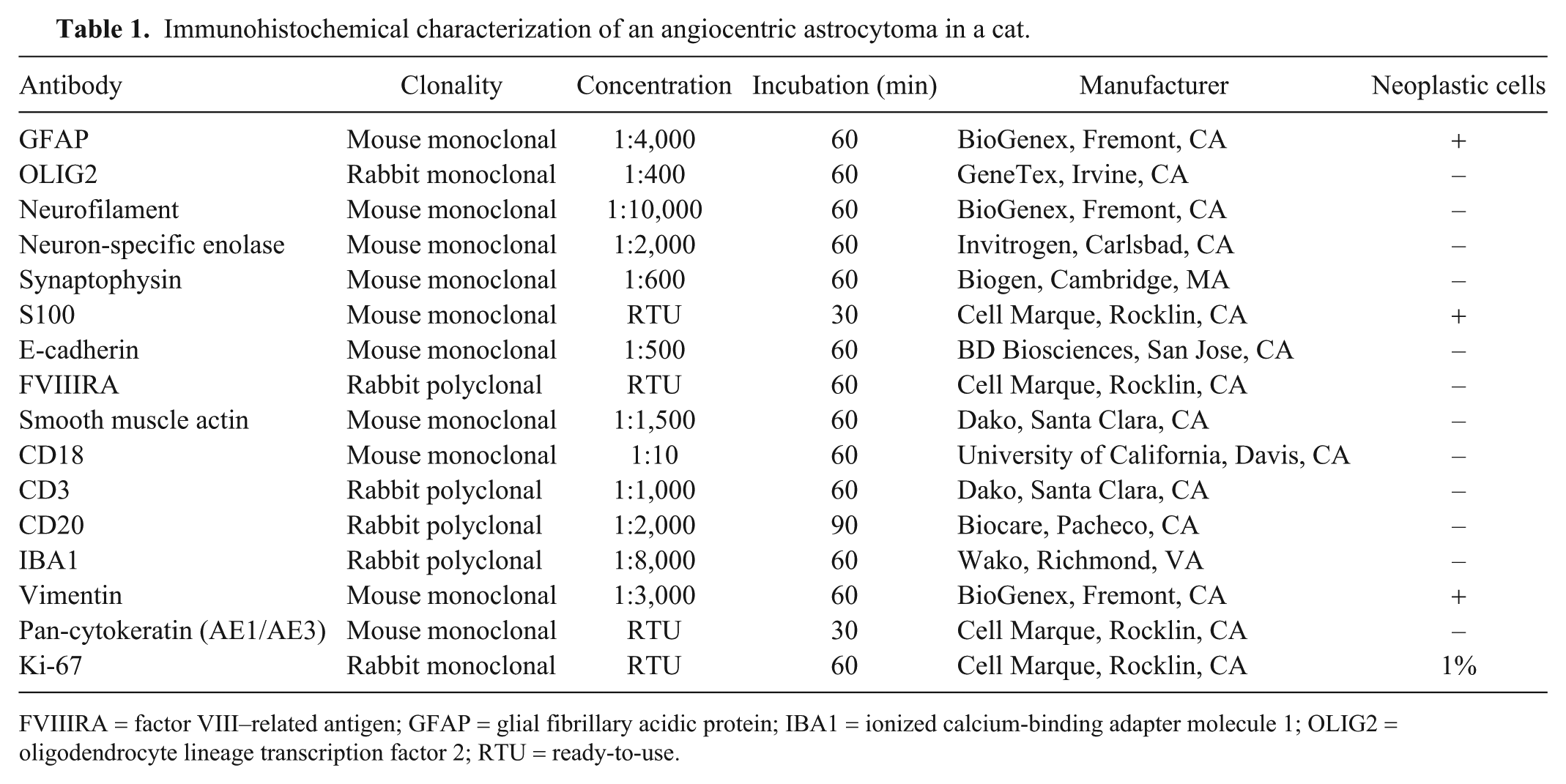
FVIIIRA = factor VIII–related antigen; GFAP = glial fibrillary acidic protein; IBA1 = ionized calcium-binding adapter molecule 1; OLIG2 = oligodendrocyte lineage transcription factor 2; RTU = ready-to-use.
Brain of a cat with angiocentric astrocytoma.
Transmission electron microscopy (TEM) was performed on selected areas of affected telencephalic cortex. Formalin-fixed tissue was post-fixed in 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer and in 1% osmium tetroxide in 0.1 M sodium cacodylate buffer (Electron Microscopy Sciences, Hatfield, PA). Samples were processed, infiltrated, and embedded (Embed 812 resin; Electron Microscopy Sciences). Thin sections (60–70 nm) were contrasted with 5% uranyl acetate and Santos lead citrate (Electron Microscopy Sciences). Leptomeningeal and perivascular proliferating cells expanded and occupied the spaces between the vascular basement membrane and the glia limitans (Fig. 8). Cells were round-to-elongate and emitted long processes. The cytosol had few organelles such as round-to-oval mitochondria, small Golgi system, and delicate rough endoplasmic reticulum. Sparse to more dense aggregates of intermediary filaments frequently displaced organelles and filled cell processes (Fig. 9). Nuclei were round-to-elongated, and had invaginations, projections, pockets and/or loops, with heterochromatin from small marginal aggregates with numerous delicate central chromatin. The nucleoli had a coarse reticular nucleolemma with nuclear-associated heterochromatin and lakes of nuclear matrix. Tight junctions connected the plasma membrane of neighboring cells (Fig. 10). Not infrequently, there were fibroblasts producing collagen fibrils, lymphocytes, and plasma cells. Endothelial cells were supported by a basement membrane.
The cell morphology, IHC, and ultrastructural findings in our case are consistent with an astrocytoma.2,15 A distinctive and challenging feature of this neoplasm was the perivascular arrangement of neoplastic cells and the lack of a primary parenchymal mass within the CNS. The absence of a distinct mass is a hallmark of rare glioma subtypes described in human and veterinary medicine, including diffuse leptomeningeal gliomatosis (described in dogs) and gliomatosis cerebri (described in dogs and cats).1,6,12,19 Primary diffuse leptomeningeal gliomatosis consists of widespread proliferation of heterotopic glial cells within the subarachnoid spaces with no parenchymal glioma.1,6 In contrast, subarachnoid infiltration by neoplastic glial cells in secondary diffuse leptomeningeal gliomatosis follows tumor invasion from a parenchymal glioma.1,6 In type 1 gliomatosis cerebri, widespread parenchymal infiltration of neoplastic glial cells occurs without a distinct tumor, whereas neoplastic infiltration is characterized by the formation of a mass in type 2 gliomatosis cerebri.19,21 Perivascular arrangement of neoplastic cells is not a feature of these 2 conditions; furthermore, the leptomeningeal involvement in our case was not diffuse but exclusively perivascular.
The clinical signs, neuroanatomic distribution, and perivascular growth pattern in our case is similar to that described for angiocentric glioma in human beings.7,8,14,25 Angiocentric glioma is a rare World Health Organization grade I glioma subtype that was first described in the mid-2000s.7,8,14,25 Given its newly recognized status and rare occurrence, incidence numbers are not yet available; however, angiocentric gliomas are slow-growing tumors that affect primarily children and young adults with therapy-resistant partial seizures. 7 Similar to our case, tumors occur mainly within the superficial telencephalic cortex and subcortical areas, and typically extend along the leptomeninges and perivascular spaces within the underlying neuroparenchyma.7,13 Neoplastic cells are monomorphic and bipolar, and arranged perpendicularly around blood vessels.7,13 Although only a small proportion of neoplastic cells in our case exhibited a clear perpendicular perivascular arrangement as described in angiocentric glioma, their exclusive perivascular growth and the absence of a parenchymal mass warrants the use of the “angiocentric” modifier as a means to distinguish the current astrocytoma from other classical astrocytomas or less common astrocytoma subtypes. On the other hand, our diagnosis should not denote that our case represents the counterpart of human angiocentric glioma, given that it is difficult to draw such conclusions based on a single case.
The histogenesis and mechanism for the perivascular growth of angiocentric glioma in human beings remain undetermined; cases typically exhibit IHC and ultrastructural evidence of ependymal differentiation, such as positive podoplanin IHC, an immunomarker that suggests ependymal differentiation in human neuropathology, and intercellular microlumina with microvilli, respectively.10,13 In our case, neoplastic cells exhibited strong immunolabeling for GFAP, S100 protein, and vimentin, similar to astrocytomas in dogs and cats4,15 and to angiocentric gliomas in humans. 8 Although these immunomarkers are not specific for astrocytes, the results of our IHC panel, together with tumor morphology, are highly supportive of an astrocytoma. 15 In addition, the ultrastructural findings, such as long cytoplasmic processes with inconspicuous bundles of intermediary filaments, low number of small organelles, presence of tight junctions, and vascular tropism were consistent with astrocytic origin. In addition, we detected no ultrastructural evidence of motile cilia and microvilli, typical of ependymal differentiation, or interdigitating membrane and anchoring junctions, typical of meningothelial differentiation.
The location of the current neoplasm warrants its differentiation from other neoplasms that can occur at the same site, including nasal and paranasal tumors and olfactory neuroblastoma. 11 The lack of tissue changes in the nasal cavity, paranasal sinuses, and cribriform plate support the diagnosis of a primary brain neoplasm rather than secondary invasion into the brain from a tumor arising from adjacent tissues; in addition, the lack of expression of epithelial and leukocytic immunomarkers rules out epithelial and round cell neoplasia (especially lymphoma), the most common tumor categories arising from these sites. 11 The lack of a parenchymal mass and formation of characteristic pseudorosettes, as well as the absent immunolabeling with cytokeratin and neuronal markers, rules out an olfactory neuroblastoma. 11
Supplemental Material
DS1_JVDI_10.1177_1040638719847239 – Supplemental material for Angiocentric astrocytoma in a cat
Supplemental material, DS1_JVDI_10.1177_1040638719847239 for Angiocentric astrocytoma in a cat by Daniel R. Rissi, Brittany J. McHale and Anibal G. Armién in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Nicole Young (Histology Laboratory, Department of Pathology, University of Georgia, College of Veterinary Medicine) for the outstanding support with immunohistochemistry.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
