Abstract
Doublecortin (DCX) and neuronal nuclear protein (NeuN) can be used as immunomarkers of neuronal progenitor cells and mature neurons, respectively. Increased DCX immunolabeling has been associated with tumor invasion in human gliomas and anaplastic canine meningiomas. These immunomarkers have not been assessed in feline gliomas. Here we characterized the DCX and NeuN immunohistochemistry (IHC) profile in 11 feline gliomas (7 oligodendrogliomas, 4 astrocytomas). Immunolabeling was classified according to intensity (weak, moderate, strong), distribution of neoplastic cell immunolabeling (1 = <30%, 2 = 30–70%, 3 = >70%), and predominant location within the neoplasm (random or at tumor margins). DCX immunolabeling was strong in 6 cases, weak in 4 cases, and moderate in 1 case. The distribution of DCX immunolabeling was characterized as 1 (4 cases), 2 (4 cases), and 3 (3 cases). DCX immunolabeling occurred predominantly in astrocytomas, which had stronger immunostaining at the tumor margins. NeuN immunolabeling was absent in all cases. Our IHC findings are similar to those reported for DCX and NeuN IHC in canine gliomas. The increased DCX immunolabeling at tumor margins is similar to labeling in invasive human gliomas and anaplastic canine meningiomas.
Keywords
Gliomas (oligodendrogliomas, astrocytomas, and ependymomas) are one of the most common primary central nervous system (CNS) neoplasms of companion animals. 8 In cats, gliomas consist predominantly of astrocytomas and oligodendrogliomas.4,8 Canine and feline gliomas are thought to arise from subependymal stem cells or from immature glial cells, which have the potential to differentiate into cells with glial and neuronal phenotype, but the histogenesis of these neoplasms remains poorly understood.6,9 Doublecortin (DCX) is essential for neuroblast migration during embryonal development of the cerebral cortex, but is also expressed by subependymal and hippocampal neural precursor cells in the adult brain.1,3 DCX has been utilized as a neuronal precursor immunomarker in human and canine gliomas.1,2,6,12 However, because of its role in neural cell migration, DCX could have a role in neoplastic cell migration and tumor invasion, 1 as evidenced by the increased DCX immunolabeling at the margins of invasive human gliomas and anaplastic canine meningiomas.1,7
Neuronal nuclear protein (NeuN), a nuclear and cytoplasmic protein expressed by mature neurons, has been used for the detection of neuronal differentiation in human gliomas. 12 Although neuronal differentiation has been described in canine and feline oligodendrogliomas using synaptophysin immunolabeling,13,14 canine gliomas are reportedly negative for NeuN. 2
Although DCX and NeuN immunohistochemistry (IHC) has been assessed in canine gliomas, these IHC stains have not been reported in feline gliomas. A better understanding of the role of DCX and NeuN in canine and feline gliomas may provide further evidence for intratumoral neuronal differentiation and, in the case of DCX, support the evidence for a useful immunomarker for the detection of more invasive tumors, which could be useful when small CNS biopsy samples are examined. 8 Here we describe the tumor neurolocalization, diagnosis, and DCX and NeuN IHC profile in 11 cases of feline glioma.
A retrospective database search of the Athens Veterinary Diagnostic Laboratory (University of Georgia, Athens, GA, USA) was performed for cases of feline glioma from 2000 to 2022. All archived glass slides were reviewed histologically, classified according to an adaptation of the Histological Classification of Tumors of the Nervous System of Domestic Animals and the World Health Organization (WHO) Classification of Tumours of the Central Nervous System,5,10,11 and subjected to IHC for Olig2 and GFAP for diagnostic confirmation, according to previously published protocols. 14 Oligodendrogliomas were classified as WHO grade 2 (low cellularity and nuclear atypia, absent or low mitotic activity) or 3 (moderate-to-high cellularity and nuclear atypia, increased mitotic activity, geographic necrosis with palisading of neoplastic cells, microvascular proliferation). Astrocytomas were classified as WHO grade 2 (low cellularity and nuclear atypia, absent or low mitotic activity), 3 (moderate-to-high cellularity and nuclear atypia, increased mitotic activity), or 4 (high cellularity and nuclear atypia, increased mitotic activity, geographic necrosis with palisading of neoplastic cells, microvascular proliferation). During histologic evaluation, the presence or absence of invasion of the glioma into the adjacent neuroparenchyma was also recorded. Cases were immunostained subsequently with DCX and NeuN (Table 1). Cytoplasmic and nuclear immunolabeling was considered a positive reaction for DCX and NeuN, respectively. Neoplastic cell immunolabeling for DCX and NeuN was recorded as positive or negative. Positive immunolabeling was classified according to intensity (weak, moderate, or strong), distribution of neoplastic cell immunolabeling (1 = <30%, 2 = 30–70%, 3 = >70%), and predominant location of labeling within the neoplasm (random or at tumor margins).
Details of immunohistochemical stains performed on sections of gliomas in cats.

We included 11 cases in our study (Table 2). Cases 1–6 and 9 have been published as part of a case series of feline glioma. 14 The diagnoses in the 11 cases were based on histology and IHC for Olig2 and GFAP, and consisted of 7 oligodendrogliomas (Fig. 1) and 4 astrocytomas (Fig. 2). Tumor invasion was observed in 7 cases (4 astrocytomas, 3 oligodendrogliomas). Cytoplasmic immunolabeling for DCX (Figs. 3, 4) was strong in 6 cases, weak in 4 cases, and moderate in 1 case. Strong DCX immunolabeling was observed in 3 of 4 astrocytomas and 3 of 7 oligodendrogliomas. The distribution of DCX immunolabeling was 1 in 4 cases (Fig. 5), 2 in 4 cases (Fig. 6), and 3 in 3 cases (Fig. 7). DCX immunolabeling was increased at the tumor margins in all 4 astrocytomas (Fig. 8) and in 1 oligodendroglioma. NeuN immunolabeling was absent in all cases.
Signalment, tumor location, diagnosis, and immunohistochemistry profile of gliomas in 11 domestic shorthaired cats.
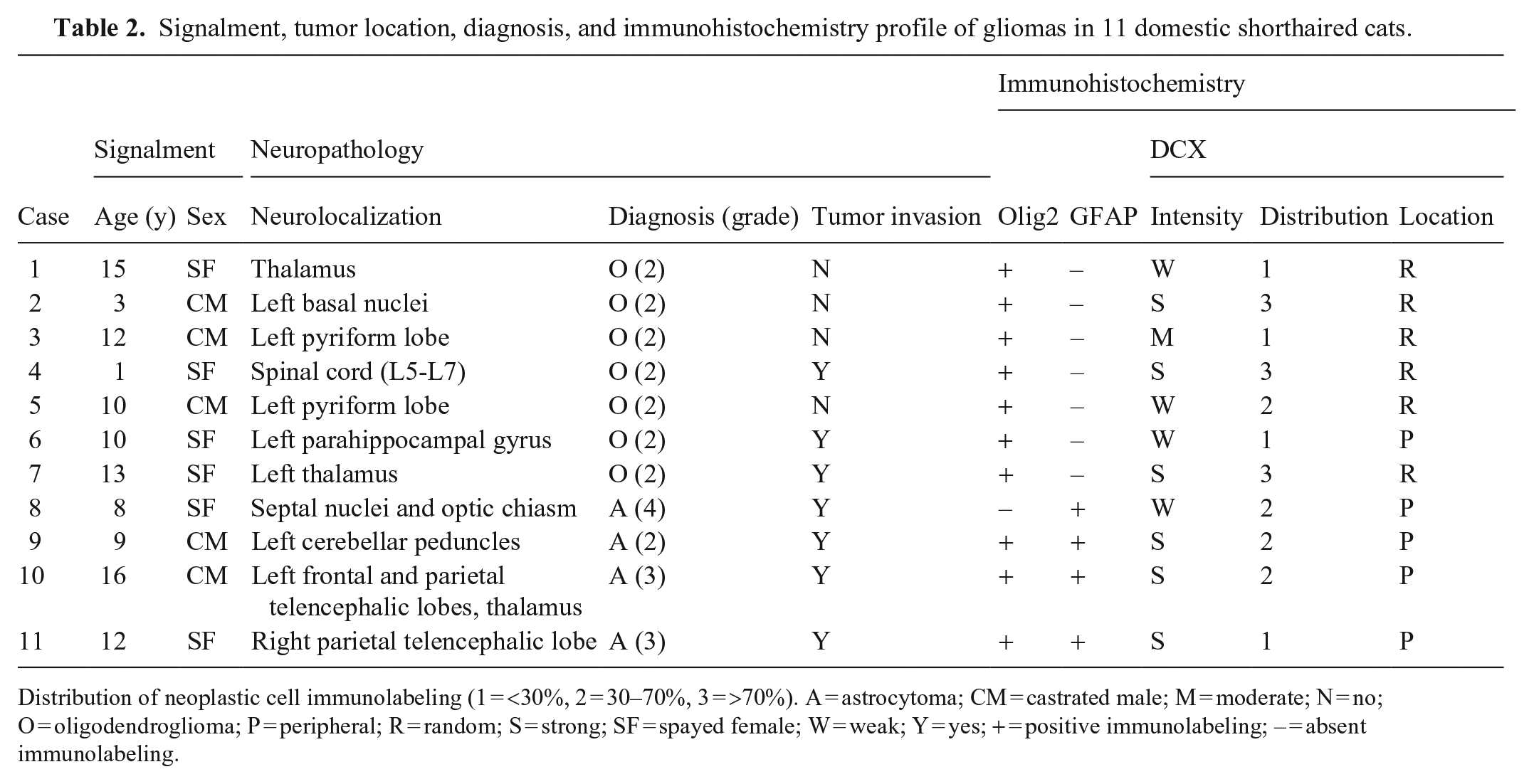
Distribution of neoplastic cell immunolabeling (1 = <30%, 2 = 30–70%, 3 = >70%). A = astrocytoma; CM = castrated male; M = moderate; N = no; O = oligodendroglioma; P = peripheral; R = random; S = strong; SF = spayed female; W = weak; Y = yes; + = positive immunolabeling; – = absent immunolabeling.
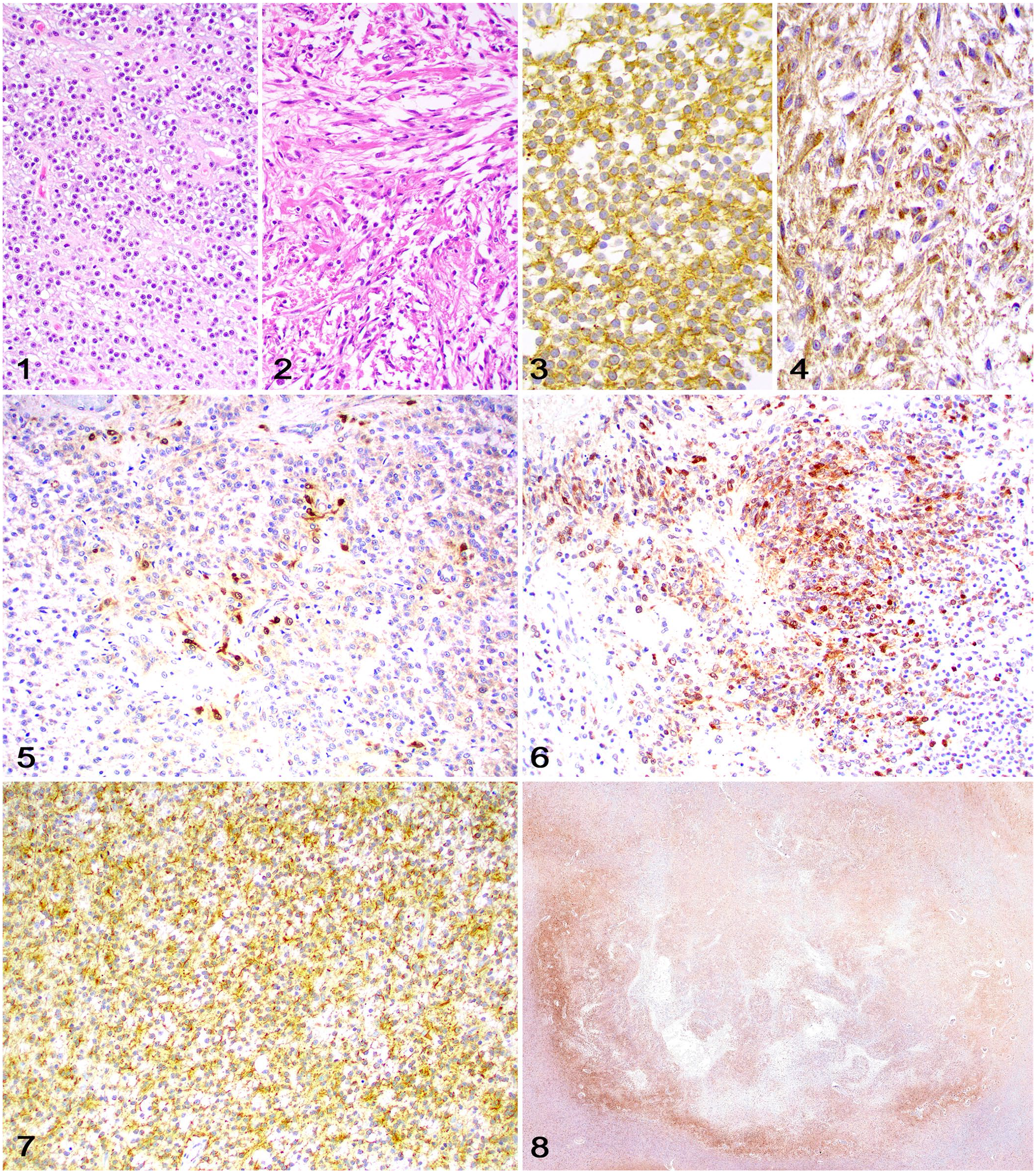
Doublecortin (DCX) immunolabeling in feline glioma.
The DCX immunolabeling in our cases was similar to findings in human and canine gliomas.1,2 Although the intensity and distribution of staining varied across cases, immunolabeling was observed in all gliomas, supporting the potential for neuronal differentiation within tumors. We observed strong DCX immunolabeling in 3 of 4 astrocytomas and 4 of 7 oligodendrogliomas. Although robust DCX immunolabeling has been reported in a group of anaplastic oligodendrogliomas in dogs, IHC features were not compared to low-grade oligodendrogliomas or to other glioma types in that investigation. 9 Therefore, the significance of the partial selectiveness for astrocytomas in our study remains undetermined and could reflect either our relatively low number of cases or the more invasive nature of astrocytomas compared to oligodendrogliomas.
DCX is preferentially expressed in more invasive human gliomas, which may have more intense immunolabeling at the tumor margins, suggesting a higher invasive capacity of DCX-positive neoplastic cells. 1 Similarly, all astrocytomas in our study had some degree of invasion and DCX immunolabeling that was distinctly more intense at the periphery of the neoplasm. In canine gliomas, DCX immunolabeling is reportedly scarce and scattered throughout the neoplasm, 2 with no stronger immunolabeling at the tumor margins, as described in anaplastic canine meningiomas. 7
NeuN immunolabeling has been a reliable tool for neuronal differentiation in human brain tumors (including most gliomas), but it has been consistently negative in canine gliomas. 2 Similarly, all feline gliomas in our study were negative for NeuN immunolabeling, indicating that other immunomarkers, such as synaptophysin, should be used for the detection of neuronal differentiation within these tumors.13,14
Footnotes
Acknowledgements
We thank Nicole Young and Jennifer Kempf (Histology Laboratory, Department of Pathology, College of Veterinary Medicine, University of Georgia) for their outstanding support with immunohistochemistry.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declare that they received no financial support for their research and/or authorship of this article.
