Abstract
Salvia reflexa (lance-leaf sage)-contaminated alfalfa hay was fed to ~500 mixed-breed beef cattle. Within hours of exposure, nearly half of the cattle developed lethargy, anorexia, depression, and recumbency, followed by bellowing, colic, and death. Even though the uneaten contaminated hay was removed the first day, nearly 100 animals died within the first 48 h. Three of these cattle were examined postmortem, and tissues and hay samples were collected for microscopic and chemical analysis. Several days later, a smaller number of the clinically poisoned cattle developed neurologic disease with aberrant behavior, aggression, icterus, blindness, exhaustion, and death. A total of 165 cattle were fatally poisoned. Poisoned cattle had swollen, dark, mottled livers that had a prominent nutmeg-like lobular pattern on cut section. Histologically, there was severe centrilobular-to-panlobular hepatic necrosis with marked hepatocellular swelling, degeneration, and necrosis. The surviving cattle developed liver disease characterized by altered serum biochemical analyses and microscopic hepatocellular degeneration and necrosis. In subsequent biopsies and analysis, these lesions resolved within 6–7 mo. After confirming toxicity of the hay in cattle, goats, and mice, followed by a mouse bioassay–guided chemical fractionation process, Salvia reflexa was identified as the contaminant in the hay responsible for the hepatotoxicity. S. reflexa has not been reported previously to cause fatal hepatotoxicity in livestock in North America, to our knowledge.
Introduction
Nearly 500 mixed-breed pregnant cows were fed hay contaminated with numerous weeds including annual kochia (Kochia scoparia; syn. summer cypress, Bassia scoparia), Russian thistle (syn. Russian tumbleweed; Salsola tragus), sunflower (Helianthus spp.), red root (Ceanothus spp.), lamb’s quarter (syn. goosefoot; Chenopodium spp.), halogeton (Halogeton glomeratus), barnyard grass (Echinochloa spp.), cocklebur (Xanthium strumarium), pennycress (Thlaspi arvense), foxtail (Setaria spp.), corn stalks (Zea mays), and lance-leaf sage (Salvia reflexa). Before feeding, the hay had been sampled and analyzed for nitrates and relative feed value, which were in the acceptable ranges for beef cattle. The producer was cautioned to transition to the weedy hay slowly and to monitor animals closely for signs of toxicity. The contaminated feed was diluted 1:1 with local grass hay. It was fed for only one day, but within 12–24 h, nearly half of the cattle became anorectic, depressed, and began showing signs of malaise. Although much of the hay had been eaten, the remaining contaminated hay was removed immediately from the feedyard. Subsequently, nearly 200 severely poisoned animals developed sternal and lateral recumbency, bellowing, apparent abdominal pain, and reluctance to stand; 165 died. Most fatalities occurred within 12–72 h of exposure. Most of the fatally poisoned cattle were pregnant cows, although several replacement heifers and young steers also died. Three of the fatally poisoned cattle were examined postmortem, and severe hepatic necrosis was confirmed microscopically.
Three hepatotoxic plants that have been reported to produce severe hepatic necrosis as seen in our case include cocklebur, Wedelia glauca, and annual kochia. Cocklebur is a known hepatotoxic weed, and the toxin (carboxyatractyloside) has been characterized as a potent hepatotoxin.2,12 Pigs and calves are most commonly poisoned, although toxicosis in other livestock species and humans has been reported.7,14,19,22 Poisoning most often occurs when animals ingest seeds or when young plants are eaten in the cotyledon stage. The toxin declines as the plant matures; however, carboxyatractyloside remains in high concentration in the seeds and burrs. Contaminated hay and grain are considered poisonous. 12 W. glauca caused severe hepatotoxicity in 2013 in the Patagonia region of Argentina, with morbidity and mortality similar to our incident. 9 In that case, W. glauca–contaminated hay poisoned 342 cattle and killed 147 calves. The Wedelia toxin is also an atractyloside, and Wedelia is recognized as a toxic plant in South America and Australia. Wedelia populations have not been reported in North America.3,9 Kochia has repeatedly been associated with liver disease in livestock. Kochia was also suspected in our case given that kochia was the most common contaminant, and, in many bales, it occasionally composed >50% of the plant material. Many suspected K. scoparia poisonings suggest that it causes liver failure with subsequent photosensitization. However, kochia has not been shown experimentally to cause hepatic necrosis.1,4,5,12,18
Although not originally thought to be hepatotoxic, Salvia was identified as a contaminant weed in the hay and was suspected to be a possible cause of hepatoxicity. S. reflexa, a North American native, is commonly referred to as lance-leaf sage, wild mint, mint weed, narrow-leafed sage, blue sage, sage mint, or Rocky Mountain sage (Fig. 1). An herbaceous annual, it commonly grows 6–15 cm tall in dry rangelands and pastures. The leaves are blue-green, covered in dense fine hairs, 2–6 cm long, 4–12 mm wide, blunt at the tip, and narrowing at the base into a short stock. Flowers occur in clusters of 3 or 4 at short intervals along spikes at the ends of the branches. They have a ribbed calyx and pale-blue corolla that is ~12 mm long. The corolla has 2 lips with the lower lip twice as long as the upper. The fruits consist of 4 small brown nuttlets that develop within the base of the calyx.6,12 S. reflexa is found in a variety of habitats, but it grows most commonly in dry sandy or gravelly soils of western North America, and it has become naturalized in South America, South Africa, New Zealand, and Australia. In Australia, it was a reported cause of nitrate poisoning in cattle and sheep.10,21 In North America, S. reflexa is also listed as a nitrate-accumulating toxic plant.1,12 In New South Wales and Queensland, Australia, S. reflexa was reported to poison sheep and cattle herded through dense populations of S. reflexa.10,21 Initial feeding trials in sheep resulted in respiratory distress, dyspnea, collapse, and rapid death that were attributed to nitrate poisoning. 21 In 2012, researchers from Australia reported that S. reflexa might contain an unidentified hepatotoxin, although the toxicity, clinical signs, or microscopic lesions were not characterized. 15

Salvia reflexa grown from seeds recovered from the contaminated hay. Note the variable sized leaves with blunt tips that narrow at the base with a short stalk. The flowers are pale blue in small clusters on spikes at the end of the branches. The inset is from a slightly older plant found near the origin of the contaminated hay. The calyxes (arrowheads) are ~0.5 cm long; calyxes and leaves have the highest concentrations of diterpenoid toxins.
The objectives of our case investigation were to identify the toxic plant in the hay that poisoned these cattle; describe the clinical disease; characterize the clinical and pathologic changes; and determine the toxicity of S. reflexa and its diterpenoids.
Materials and methods
Experimental approach to the investigation
Initial samples from the poisoned cattle were collected from the ranch, from initial postmortem examinations, and from hay remaining on site. Additionally, all contaminated hay bales (220 large square bales weighing ~500 kg each) were secured, numbered, core sampled, examined visually, and contamination subjectively graded (low = <25% weeds, moderate = 25–50% weeds, and high = >50% weeds). Selected plant samples (200–500 g) were finely ground to pass through a 2-mm screen using a hammer mill grinder (95 grinder/mixer; Gehl) and analyzed for nitrates, cocklebur toxins (carboxyatractylosides), mycotoxins, pesticides, and microcystin (these analyses were performed at various laboratories [FDA/Vet-LIRN], and given that all results were negative, they are not included in this report). A few contaminated bales were cut open, and weed fragments were identified.
Pen studies in cattle
Cattle were initially used to confirm the toxicity of the contaminated hay and to identify a bale that could be used for the mouse-guided chemical extraction process. These pen studies were done under a veterinarian’s direction following protocols approved by the Utah State University, Institutional Animal Care and Use Committee (USU-IACUC, protocol 2436).
Eight surviving cows from the Colorado herd were studied more closely for disease resolution. These cows were examined several times each day, blood samples were collected weekly for biochemical analysis, and liver biopsies 16 were collected every 6 wk to determine the progression and extent of hepatic damage. Sera were analyzed biochemically using an automated wet chemistry analyzer (7180 chemistry analyzer; Hitachi) for albumin, total protein, urea, creatinine, sodium, potassium, chloride, carbon dioxide, alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), and bilirubin (BIL) using reagents and procedures recommended by the instrument supplier. Six months after these cattle were determined to have recovered from the initial exposure, they were divided into 2 pens of 4 cows each and fed 2 highly contaminated bales free choice. Although water, salt, and mineral were provided, no other feed was available. After 5 d of feeding, none of the previously exposed animals would eat the contaminated hay, and their exposure was discontinued.
The second trial included 15 naïve (not previously exposed to the toxic hay) mixed-breed, non-pregnant cows, heifers, and steers. These were divided into 3 groups of 5 cattle including at least 1 heifer, 1 steer, and 2 or 3 mature cows. The groups were fed contaminated bales with low, moderate, and high contamination. Each group of cattle was fed from the same bale for the entire 5-d study; all had free-choice access to fresh water and trace mineral salt. The cattle were examined multiple times each day, serum was collected daily, and liver samples were collected at 3 d and then 6 wk after the study. The sera were analyzed as described in the recovering cattle. Cattle in the high-contamination group that developed clinical disease (anorexia, reluctance to move, or biochemical changes suggestive of hepatic disease) were euthanized, and postmortem examinations were performed. Samples of liver, spleen, pancreas, rumen, abomasum, duodenum, jejunum, ileum, cecum, colon, kidney, urinary bladder, adrenal gland, lung, thyroid, brain, and spinal cord were collected, fixed in 10% neutral-buffered formalin, processed routinely, and sections stained with hematoxylin and eosin for microscopic evaluation. Differences in pre- and post-treatment serum biochemistry results and group results were tested using analysis of variance, and significantly different means (p ≤ 0.05) were identified using the Duncan test (SAS Statistical Software, Proc GLM SAS). A highly contaminated bale fed to the high-contamination group was identified and used in subsequent studies.
Goat studies
Spanish goats (Capra hircus) were used as a small ruminant model to confirm toxicity of the contaminated hay and the separately collected S. reflexa plant material. These goat studies were also done under the direction of a veterinarian following protocol 2463 approved by the USU-IACUC. All goats had free access to fresh water and trace mineral salt. Nine Spanish goats, 8–10 mo old, were weighed and divided into 3 groups of 3 goats each. The first group was dosed with ground highly contaminated hay from the bale shown to affect cattle in the high-contamination group. The second group was treated with dried, ground S. reflexa collected from the field in which the toxic hay was grown. Plants were 10–30 cm tall, in full bloom (the approximate stage when harvested in the contaminated hay; Fig. 1). This plant material was identified as S. reflexa by the Utah State University Herbarium, and voucher specimens were prepared and stored in the Poisonous Plant Research Laboratory herbarium (voucher 4692). The last group was fed alfalfa as a negative control. All of the plant material was ground with a laboratory grinder (Wiley mill laboratory grinder; Thomas Scientific) to pass through a 2-mm screen, then weighed; 150 g were mixed with 1 L of tap water to make a slurry. A single dose (150 g plant + 1 L of water) of the ground plant/water slurry was then administered to each goat via a stomach tube and manual pump. Blood samples were collected, and sera harvested at 0, 2, 4, 6, 8, 10,12,18, and 24 h and continuing every 8 h until the goats were euthanized 5 d post-treatment. After euthanasia, all goats were examined postmortem, and similar tissue samples were collected, processed, and examined as described in the cattle study. Serum biochemistry results were determined using similar instrumentation and methodology as described in the cattle feeding trials. Differences in pre- and post-treatment values, as well as group results, were compared using analysis of variance, and significantly different means (p ≤ 0.05) were identified using the Duncan test (SAS Statistical Software, Proc GLM SAS).
Mouse bioassay
The toxic bale was ground to pass through a 2-mm screen and extracted using various organic solvents. 8 These fractions were dosed by oral gavage to mice to identify which fractions, and finally which plant compounds, were hepatotoxic. These studies were also done under the supervision of a veterinarian following protocol 2461 approved by the USU-IACUC. One hundred and twenty weanling (14–16 g) male Swiss Webster mice (Simonsen Laboratories) were purchased and acclimated for at least 5 d before use. All had free access to fresh water and food after treatment. The mice were maintained under artificial light on a 12 h on/off light/dark cycle. Prior to treatment, mice were denied food for 8–12 h to facilitate oral dosing and provide a fasting standardized weight. The mice were weighed, tail marked for identification, and assigned to groups housed in separate cages of 3–5 mice per cage.
The chemical extracts used in the mouse bioassay and the separated fractions of those extracts are described in a companion paper. 8 Alfalfa was extracted with ethanol as a control, and that extract was given to the first group. Two additional groups were treated with extracts of the contaminated hay that were extracted with ethanol or ethyl acetate. S. reflexa plant material was extracted with ethyl acetate and that extract was given to the last group. Serum analytes were compared among all 4 groups. All extracts were prepared for treatment by mixing and suspending them in water or by deposition of the extract on dried milk, evaporating all solvents from the mixture in a vacuum oven for 16 h, and then suspending in water. The extract suspension (1.0–1.5 mL per mouse) was then given to each mouse orally using a rodent gavage needle (Popper). The mice were closely monitored for the next 48 h. Clinical signs were recorded and, when mice became lethargic or moribund, they were euthanized, blood collected, examined postmortem, and equivalent tissues (except rumen and abomasum) as described in the cow study were collected, processed, and examined microscopically. Blood samples were collected postmortem via cardiac puncture, and the harvested serum was analyzed as described in the cattle feeding trials. After 48 h, all remaining mice were euthanized and similarly examined. Differences between groups were identified using analysis of variance, and significantly different means (p ≤ 0.05) were identified using the Duncan test (SAS Statistical Software, Proc GLM SAS).
Analysis of plant parts
S. reflexa seeds were collected from contaminated hay and grown in a greenhouse (Fig. 1). Once the toxic diterpenoids were identified, 8 analysis of plant stems, leaves, roots, and seeds were done to determine where the toxin(s) occur in the plant. Also, plugs taken from each bale were analyzed to determine the level of Salvia contamination. For the analyses, a 100-mg aliquot of sample was extracted with 5 mL of dichloromethane for 1 h. The samples were filtered, and 1 mL was transferred to autosampler vials and analyzed by gas chromatography–mass spectrometry (GC-MS; 5977 MSD detector and 7890B gas chromatograph equipped with a split/splitless injector; Agilent Technologies). Samples (1 µL) were injected in the splitless mode (250°C) on to a DB-5MS capillary (30 m × 0.25 mm i.d.) column. The column oven was temperature programmed in the following sequence: 100°C (0–1 min); 100–200°C (40°C/min); 200–325°C (10°C/min). Transfer line temperature was 275°C. MS data were collected after ionization at 70eV over a range of m/z 50–650. To estimate the level of contamination by Salvia in core samples, a set of standard plant mixtures was prepared by mixing ground S. reflexa with ground K. scoparia at contamination levels of 50%, 25%, 12.5%, 6.25%, 3.12%, 1.56%, and 0.78% Salvia in Kochia. Peak area (GC-MS) for salviarin (Rt = 15.4 min) versus % Salvia was then used for calibration.
Results
Clinically poisoned animals
Fatally poisoned cattle had hepatomegaly with prominent lobular red discoloration (Fig. 2A, 2B, 2D). Histologically, there was severe centrilobular-to-panlobular hepatic necrosis characterized by marked hepatocellular swelling, degeneration, and necrosis, with focal aggregates of fibrin, cellular debris, and pools of erythrocytes. The remaining hepatocytes were individualized with loss of cord structure. The vascular structures were similarly disrupted, and there were large areas of hemorrhage replacing many of the centrilobular zones.
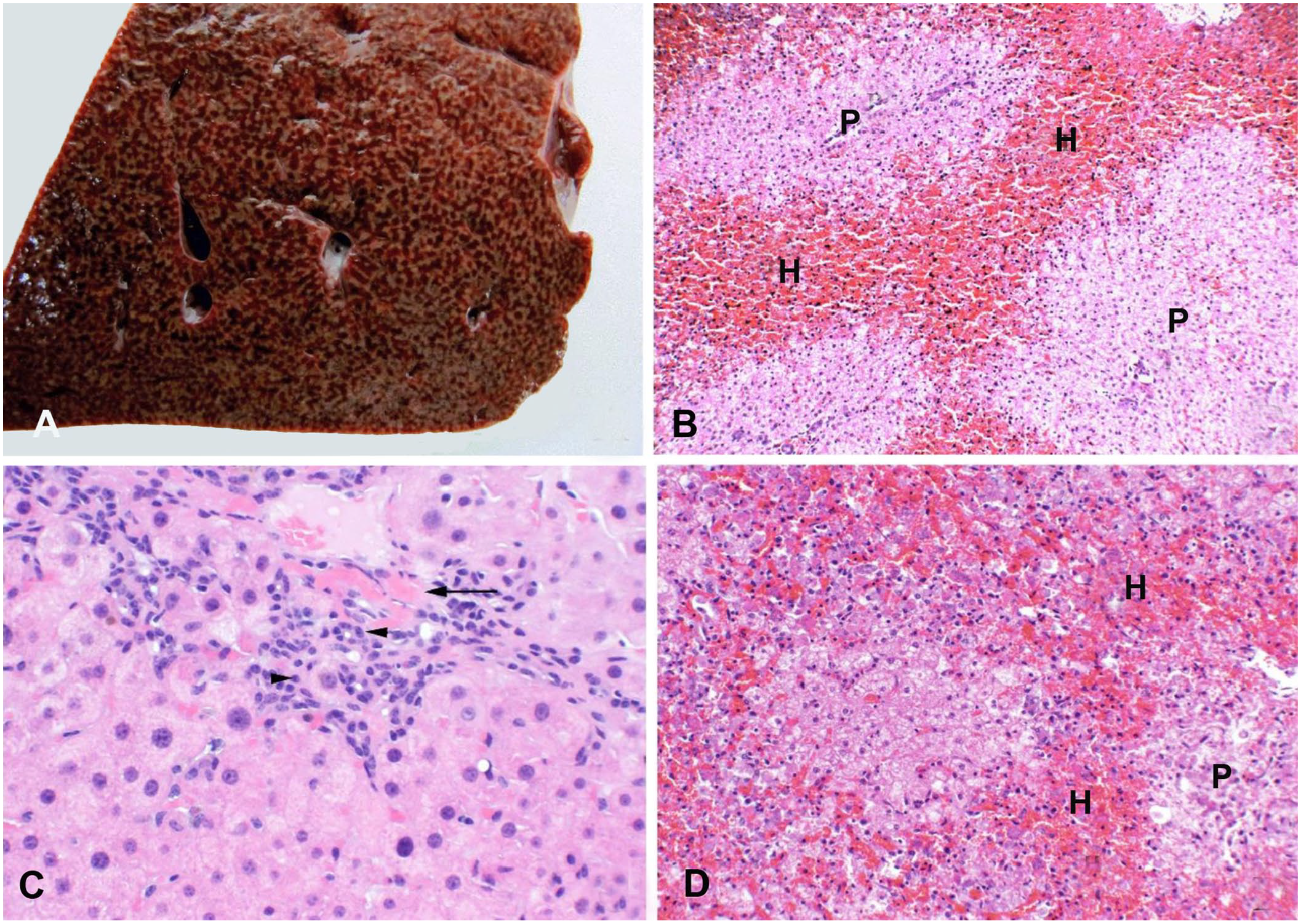
Liver from 2 cows that were fatally poisoned with Salvia reflexa–contaminated hay (1 field case, A and B; 1 experimental case, D). Panel C refers to a biopsy from an original native cow recovering from poisoning.
Livers of the 8 surviving cows that had demonstrated clinical signs of poisoning (anorexia, reluctance to move, minimal jaundice, and biochemical indicators of hepatic damage) were biopsied and examined every 6 wk for 6 mo (Fig. 2C). Initially, the cows had multifocal necrotizing hepatitis characterized by focal centrilobular hepatocellular necrosis with accumulations of debris-filled macrophages and neutrophils. The adjacent hepatocytes were swollen with prominent vacuolation that was most likely lipid accumulation. The necrosis and hemorrhage were replaced with focal inflammation and hyperplastic regenerative hepatocytes. After 3–4 mo of recovery, the only lesions were minimal focal periportal fibrosis and biliary hyperplasia. Scattered throughout the fibrous connective tissue were small numbers of macrophages and lymphocytes. There were no serum biochemical changes during recovery, and there were no additional microscopic hepatic changes.
Cattle pen study
Of the 15 naïve cattle that were fed the contaminated hay, all initially ate the hay readily; however, they quickly began to sort out certain weed stems and plant fragments that they left in the bottom of the feed bunk. Three cattle in the highly contaminated bale group developed clinical disease within 3–4 d of feeding the bale. Clinical signs were characterized initially by complete feed refusal and reluctance to move. This progressed rapidly to depression, lethargy, and recumbency after which they were euthanized and examined postmortem; samples were collected, fixed, and processed for histologic evaluation. All 3 animals also had biochemical changes of liver disease that were often marked; however, there was marked variation between animals suggesting individual susceptibility. Only 3 of the 5 cattle in the high group are reported in Table 1, and all had hyperbilirubinemia and increased and highly variable activities of ALP and AST. Over the course of the feeding trial, the remaining cattle fed the low and moderately contaminated bales did not develop clinical signs of liver disease, although they lost weight and body condition; their blood serum enzyme activities did not exceed reference intervals. The cattle sorted through the hay leaving stems and fines in the bottom of the manger. The fines contained S. reflexa leaves, seeds, calyxes, and other unidentifiable plant parts from Salvia and other species. However, when later analyzed, the fines contained high concentrations of subsequently identified hepatotoxins.
Select serum biochemistry results from cattle fed hay containing different levels of weed contamination (low = <25% weed contamination; moderate = 25–50% weed contamination; and high = >50% weed contamination [3 cattle in the high group were clinically poisoned]).
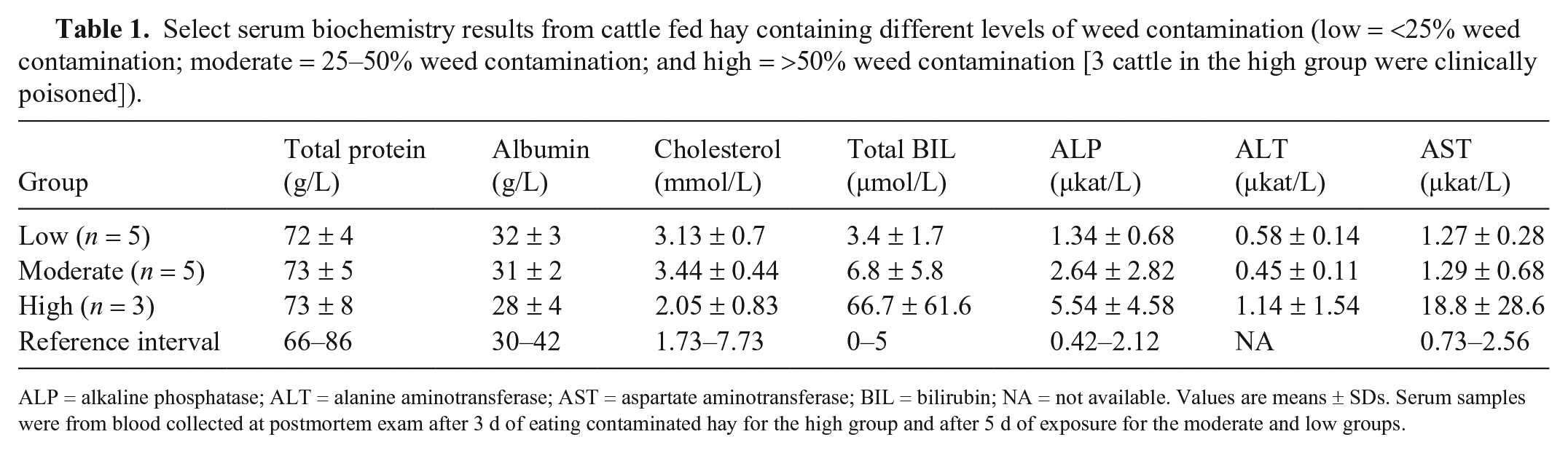
ALP = alkaline phosphatase; ALT = alanine aminotransferase; AST = aspartate aminotransferase; BIL = bilirubin; NA = not available. Values are means ± SDs. Serum samples were from blood collected at postmortem exam after 3 d of eating contaminated hay for the high group and after 5 d of exposure for the moderate and low groups.
Similar to the lesions described in the clinically poisoned cattle, the livers from the 3 animals that were poisoned in the pen study had severe hepatocellular necrosis characterized by hepatic swelling with a prominent lobular pattern visible on the capsular surface and on cut surfaces of all liver lobules (Fig. 2A, 2B, 2D). Histologically there was extensive centrilobular to panlobular hepatic necrosis and hemorrhage, with loss of hepatic cords and lobular collapse. There was also hepatocellular vacuolar degeneration; the remaining cells were individualized with loss of cord structure and hemorrhage in the centrilobular zones. No significant histologic lesions were identified in sections of brain, thyroid, adrenal, pancreas, skeletal muscle, heart, kidney, urinary bladder, rumen, abomasum, small intestine, or colon.
In addition to confirming the contaminated hay as the cause of the clinical intoxication, a highly toxic bale was identified to be used in the mouse bioassay–guided chemical extraction and fractionation process that eventually identified the offending plant as S. reflexa and ultimately led to the identification of 4 toxic diterpenoid compounds responsible for the massive liver damage. 8
Goat confirmation study
Goats dosed with the contaminated hay or S. reflexa quickly developed depression and lethargy. Biochemically affected goats also had significant (p ≤ 0.05) increases in total BIL and ALP, and although the ALT and AST activities were variable, they also tended to be elevated (Table 2). Poisoned goats were icteric, with jaundice of mucous membranes, adipose tissue, and serum. They had red and swollen livers; however, the distinct lobular mottling seen in cattle was less prominent (Fig. 3A, 3B). The histologic changes were similar to cattle, with centrilobular hepatocellular swelling and coagulative necrosis. Other tissues were often congested, and all fatally poisoned animals had severe pulmonary congestion with edema in many alveoli and small airways. Several of the goats dosed with S. reflexa also had acute necrosis of the proximal convoluted tubules, with accumulations of debris, fibrin, and edema in the tubular remnants (Fig. 3C). These same animals also had swelling and hypereosinophilia of myocardiocytes in the large ventricular papillary muscles (Fig. 3D). The lack of inflammation and coagulation of the sarcomere proteins suggest this may be an agonal lesion secondary to the toxicosis. No significant histologic lesions were identified in sections of brain, thyroids, adrenals, pancreas, skeletal muscle, urinary bladder, rumen, abomasum, small intestine, or colon.
Select serum biochemistry results of goats treated with alfalfa (control), Salvia reflexa plant material, or weed-contaminated hay. Sera were collected from blood taken 24 h after initial dosing.
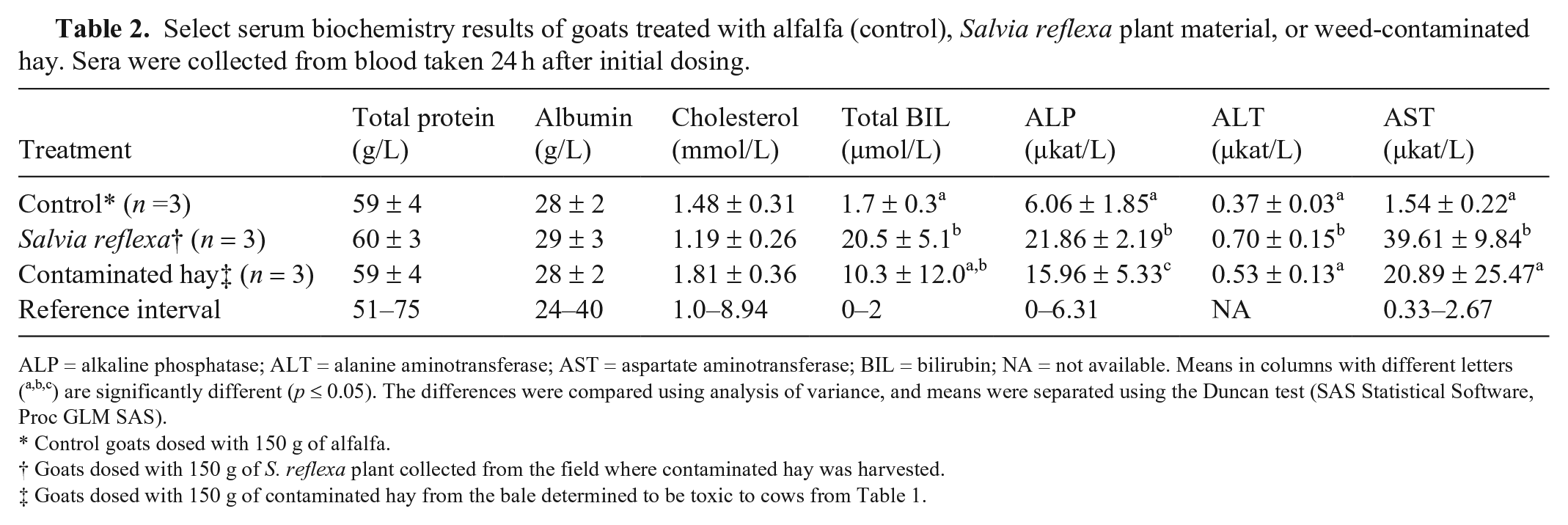
ALP = alkaline phosphatase; ALT = alanine aminotransferase; AST = aspartate aminotransferase; BIL = bilirubin; NA = not available. Means in columns with different letters (a,b,c) are significantly different (p ≤ 0.05). The differences were compared using analysis of variance, and means were separated using the Duncan test (SAS Statistical Software, Proc GLM SAS).
Control goats dosed with 150 g of alfalfa.
Goats dosed with 150 g of S. reflexa plant collected from the field where contaminated hay was harvested.
Goats dosed with 150 g of contaminated hay from the bale determined to be toxic to cows from Table 1.
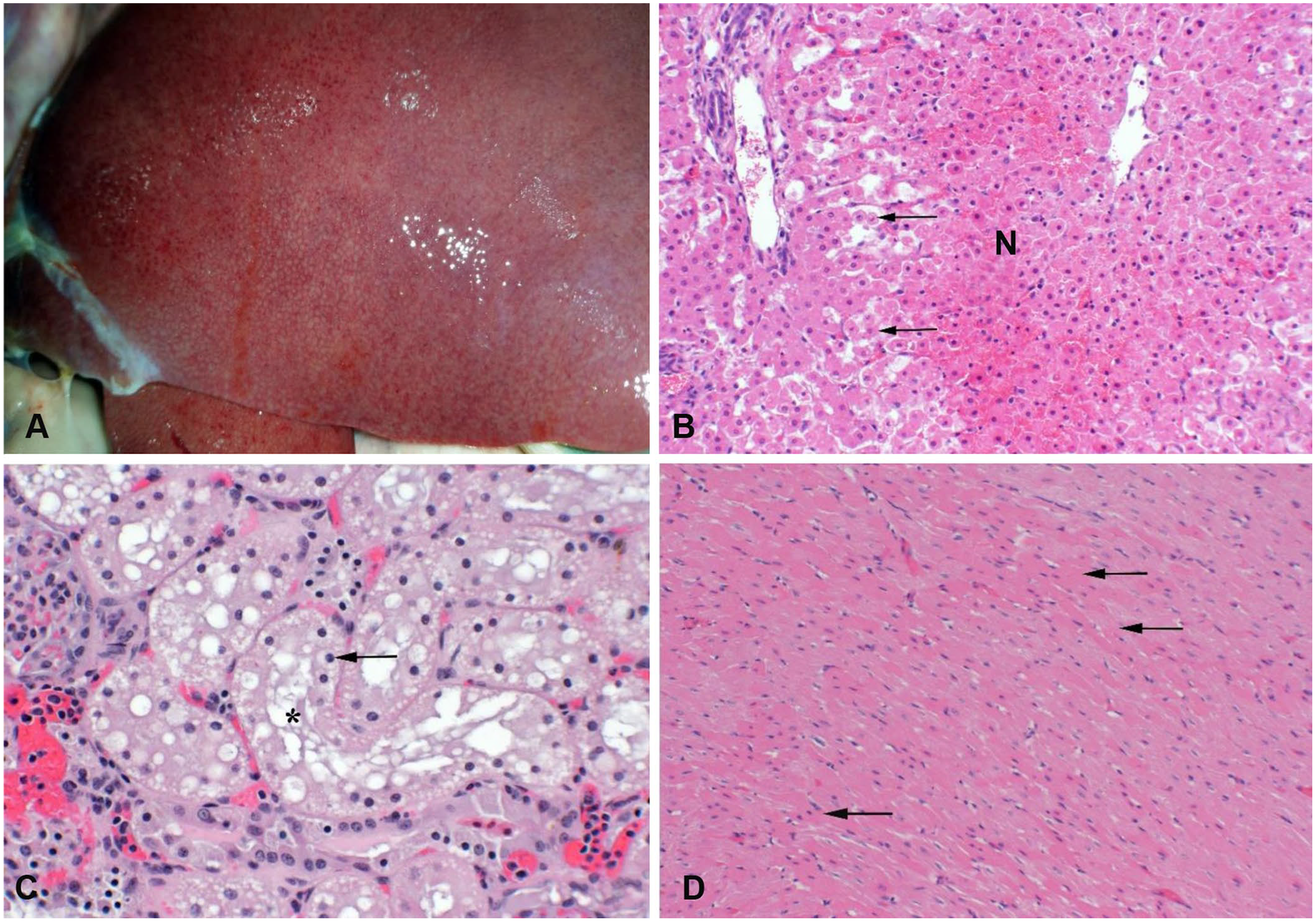
Liver of a goat dosed with ground Salvia reflexa collected near the alfalfa field that produced the hay of the original clinical poisoning. The plants were collected in the late flower phenotype, 15–30 cm tall.
Mouse bioassay
Crude extracts of the toxic hay using ethanol, methanol, ethyl acetate, or dichloromethane as solvents were all determined to be toxic given that treated mice became ill and developed hepatic necrosis. Less polar and more polar solvents were less efficient at extracting the toxic fraction, as seen by a reduced response or no response in the mouse model. In further experiments, a mixture of organic solvents would be used in conjunction with various columns to fractionate the extracts to identify: first the toxic plant or plants, and subsequently the pure compounds responsible for the liver toxicity. 8
Toxic fractions produced clinical disease in mice; serum biochemistry alterations and microscopic lesions were similar to those in cattle and goats (Table 3). Serum biochemical changes and histologic lesions were used to identify the toxic fractions and ultimately the toxic diterpenoids. Consistent changes of induced liver disease increased serum BIL and increased AST and ALP activities. These changes were significantly higher than the alfalfa control group (p ≤ 0.05). A total BIL response of 0.3 or above was considered a positive response, and the positive fraction was then advanced to the next phase. This decision was confirmed with postmortem findings of hepatomegaly, red discoloration, and diffuse mottling on the cut surface (Fig. 4). Histologically, there was acute hepatocellular coagulative necrosis. This was less severe than the S. reflexa–induced hepatic necrosis in the cattle and goats. The necrosis in mice was confined to the centrilobular or the mid-zonal regions. No significant microscopic lesions were identified in any of the other brain, heart, kidney, pancreas, stomach, small intestine, or colon sections.
Select serum biochemistries from mice orally gavaged with alfalfa extract, ethanol, or ethyl acetate extracts of a heavily contaminated bale, and Salvia reflexa extract. The sera were from blood collected postmortem ~24 h after dosing.
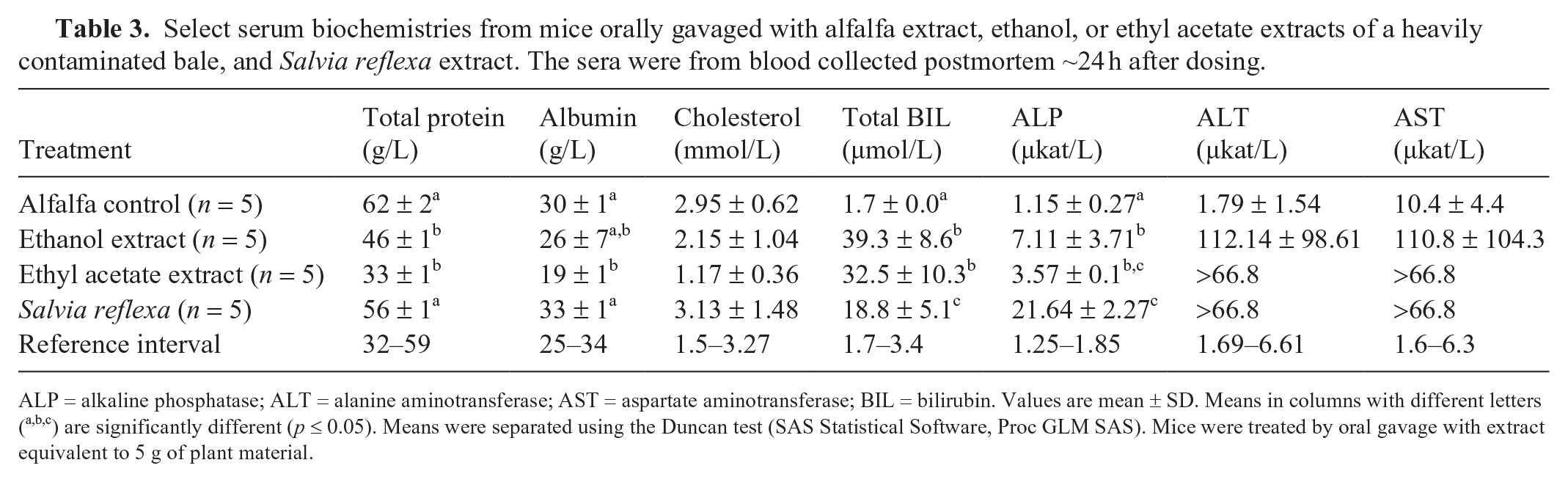
ALP = alkaline phosphatase; ALT = alanine aminotransferase; AST = aspartate aminotransferase; BIL = bilirubin. Values are mean ± SD. Means in columns with different letters (a,b,c) are significantly different (p ≤ 0.05). Means were separated using the Duncan test (SAS Statistical Software, Proc GLM SAS). Mice were treated by oral gavage with extract equivalent to 5 g of plant material.
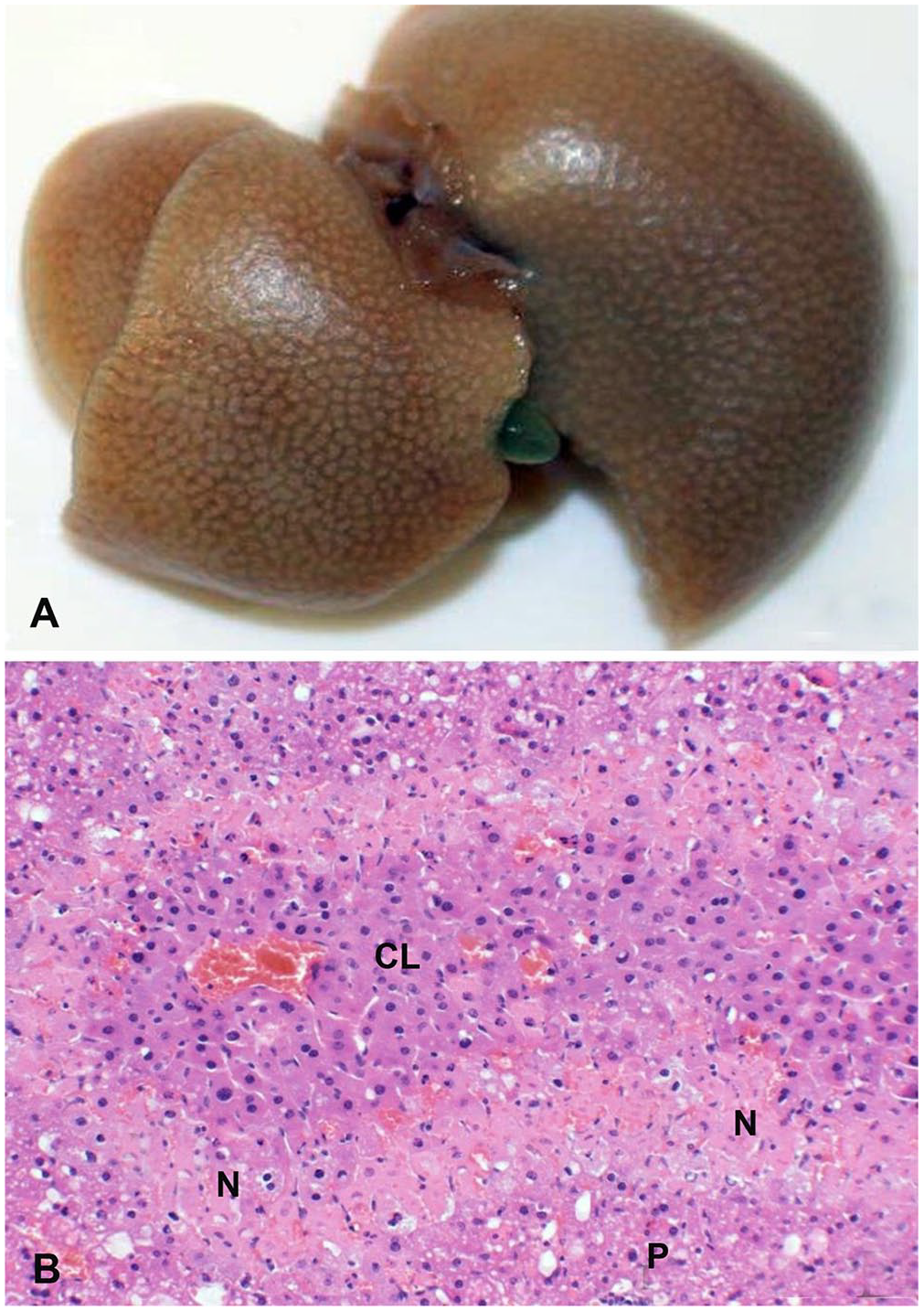
Liver from a mouse dosed with a Salvia reflexa extract via oral gavage.
During the extraction and fractionation process, one of the hepatotoxic compounds was identified as salviarin, a Salvia spp. diterpenoid. This finding confirmed the clinical indications that S. reflexa was the cause of this intoxication.
Toxins in plant parts and bales
Once the hepatotoxic diterpenoids (salviarin and related diterpenoids) were identified, various plant parts from greenhouse-raised S. reflexa were analyzed. Seeds contained little toxin, but the calyx holding the seeds contained the highest concentrations, followed by the flowers, leaves, stems, and roots. An interesting observation was that the ripe seeds easily drop from the plant but the calyx clings to the stems and remained in the hay with the stems and leaves (Fig. 5; the calyxes and leaves were the most toxic parts of the plant). Furthermore, chemical analysis was then used to analyze all plug samples collected from the 220 bales to determine how many bales contained S. reflexa contamination. It was determined that 3.2% of the plug samples were highly contaminated (≥50% contamination); 21% were moderately contaminated (25–50% contamination); and 64% were minimally contaminated (<25% contamination). Only 17 of the 220 bales had no detectible Salvia contamination. Given that only the highly toxic and weedy bales produced intoxication, this suggests that several highly toxic bales had been fed initially in this clinical intoxication.

Photograph of an opened section of the heavily contaminated bale used to identify Salvia reflexa as the toxic component. Note the numerous Salvia stems, leaves, and seed pods (calyxes). The insert is a closer view of the calyxes (arrowheads) and leaf fragments (arrow), which are the most toxic parts of the plant.
Discussion
The experimental disease that we induced with S. reflexa in cattle, goats, and mice was nearly identical to the disease in the field case. Therefore, we concluded that S. reflexa and its hepatotoxic diterpenoids caused the death of these cattle. In retrospect, the factors that led to that fatal event were related to many variables including animal appetite, cold weather, highly contaminated bales that were fed initially, cattle nutritional status including late-term pregnancy, and competition on the feed grounds. Certainly, all of these events contributed to so many of these cattle eating too much too quickly that resulted in fatal hepatocellular necrosis.
The mouse bioassay indicated that Salvia diterpenoids are potent hepatotoxins. In contrast to the massive hepatocellular necrosis seen in cattle, S. reflexa–induced hepatic necrosis in mice was less severe with variable distribution. Such lesions are nonspecific and could have been produced by a variety of hepatotoxins. Certainly, dose and duration contribute to individual and species differences in response. However, the severe hepatic necrosis in cattle suggests that these diterpenoids from Salvia are potent metabolic toxins similar to cocklebur carboxyatractylosides, other atractylosides, or curcumin. Given that these latter toxins are inhibitors of mitochondrial oxidative phosphorylation, analyzing Salvia-exposed tissues for similar mechanisms may be a starting point in determining the mechanism of S. reflexa–induced hepatotoxicity.13,14
There were some minor differences in the microscopic findings in goats. The goats developed nephrosis and myocardial lesions that were not identified in the cattle or mice. Both the myocardium and the renal tubular epithelia are metabolically active, are dependent on oxidative phosphorylation, and therefore susceptible to damage by metabolic toxins. Mice also had unique serum protein changes, including hypoalbuminemia. Additional research is needed to better characterize these extrahepatic lesions in mice and goats and determine if they are toxin related or if they are secondary to hepatic failure.
Severely poisoned animals that survived several days had hyperbilirubinemia with jaundice of many mucosal membranes, adipose tissues, and serous membranes. Histologically, these recovering animals had mild hepatocellular degeneration with minimal necrosis. Later in recovery, these lesions became chronic with focal fibrosis and chronic active inflammation. During recovery, the original centrilobular necrosis resolved as damaged hepatocytes were replaced with large swollen hepatocytes that often formed hyperplastic nodules. Later, after months of recovery, the remaining lesions were minimal fibrosis and inflammation adjacent to portal tracts. Such reactions are common when the liver responds to infectious, toxic, or immunologic damage. Regardless of the cause, cell activation and changes in expression of various fibrotic biomarkers result in fibrosis that is most common in portal areas.11,20 In any case, these lesions were not progressive given that the previously exposed but non-fatally poisoned cattle recovered. Nearly all of the biopsies taken 6 mo after the initial exposure were normal microscopically. The lack of microscopic changes as well as lack of biochemical changes suggest that these cattle have fully recovered and were not likely to suffer a relapse. Such a recovery is in contrast to hepatic disease in some animals following dehydropyrrolizidine alkaloid poisoning; poisoned animals may appear clinically normal, but the hepatic lesions continue to progress to produce fatal hepatic failure. 17 Sublethal liver lesions in cattle recovering from S. reflexa intoxication are nonspecific and would be difficult to differentiate from other hepatic diseases.
Finally, S. reflexa toxicity seems to be largely dependent on the initial dose. Both the clinical cases and our experimental studies indicate that, to develop disease, animals must ingest or be dosed with enough plant or toxin to produce disease within the first few days. Lesser doses do not produce disease, the animals become tolerant, develop aversion to eating the plant, and are unlikely to develop any clinical signs or microscopic lesions. Additional work is needed to determine the mechanism of this tolerance and to determine if it might be used as a treatment to alter or minimize S. reflexa toxicity.
Our findings indicate that S. reflexa was the cause of this fatal poisoning. More work is needed to characterize S. reflexa poisoning and to determine when plant populations are most toxic and likely to poison livestock. S. reflexa is a relatively small annual weed and difficult to identify in pastures or hay. Certainly, hay and cattle producers, extension specialists, veterinarians, and diagnosticians should be aware of this plant and its toxic potential if it is found in their areas. In addition to the risk of poisoning from contaminated hay, other studies suggest caution should also be used when trailing or pasturing hungry animals through S. reflexa–infested pastures or ranges.10,21
Footnotes
Acknowledgements
We thank Sarah M. Nemser, David Rotstein, Olgica Ceric, and Renate Reimschuessel at the FDA Veterinary Laboratory Investigation and Response Network (Vet-LIRN) for guidance and coordination among laboratories during the initial investigation. We also thank Terrie Wierenga, Scott Larsen, Joseph Jacobsen, Ed Knoppel, Katie Lott, Kermit Price, and Charles Hales from the Poisonous Plant Research Laboratory for their technical assistance. We recognize the assistance of technicians and laboratory personnel at the Western Slope Branch Veterinary Diagnostic Laboratory, Grand Junction, CO, and the Colorado State University Veterinary Diagnostic Laboratory at Fort Collins, CO. Finally, we recognize the full cooperation and transparency of the cattle producer and hay producer in this investigation without whom we would likely not have solved this case.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
