Abstract
Measurement of secreted interferon (IFN)-γ has proven to be a valuable tool for the detection of animals infected with mycobacterial pathogens, including Mycobacterium avium subsp. paratuberculosis. Previous reports have suggested that tuberculin skin testing can influence the performance of the IFN-γ assay. In the present study, healthy noninfected cows, and cows subclinically and clinically infected with M. paratuberculosis were administered an intradermal injection of johnin purified protein derivative (JPPD) and effects on secreted and intracellular IFN-γ were observed. Intradermal injection resulted in significant increases in secreted IFN-γ for subclinically infected cows after stimulation of peripheral blood mononuclear cells (PBMC) with concanavalin A or M. paratuberculosis antigen preparations (whole-cell sonicate and JPPD) on days 7 and 10 postinjection. Intracellular IFN-γ was increased after intradermal injection in total PBMC for all treatment groups and was higher within CD4+ and CD8+ subpopulations for infected cows compared to healthy controls throughout the study. When T-cell populations were further defined by CD45RO expression, intracellular IFN-γ was higher within CD8+/CD45RO+ lymphocytes compared to CD4+/CD45RO+ cells for subclinically and clinically infected cows but similar within these subpopulations for healthy controls. These results indicate that intradermal sensitization of cows in the subclinical stage of infection will upregulate expression of IFN-γ, enhancing the sensitivity of this assay. In addition, CD8+ lymphocytes appear to play an important role as a mediator of M. paratuberculosis infection in naturally exposed cattle.
Keywords
Introduction
Johne's disease (paratuberculosis) is a chronic progressive enteric disease of domestic and wild ruminants caused by the intracellular pathogen, Mycobacterium avium subsp. paratuberculosis. 8 Clinical disease in adult cattle is characterized by intermittent diarrhea, weight loss, and inappetence in the end stages of disease. However, clinical signs are generally preceded by a long period of subclinical infection in which the infected animal is asymptomatic. It is not known what precipitates the progression from sub-clinical to clinical paratuberculosis in cattle but it is believed that a deviation in the host immune response occurs, causing a disruption in the ability of the host to contain the disease. 25
A reciprocal relationship has been documented between the host T-cell responses and the extent of disease during mycobacterial infections. 12,14,29 Studies of human patients infected with M. tuberculosis suggest a predominance of Th1-mediated immunity in the early stages of disease, with a shift to Th2-mediated immunity in the more advanced stages. 5 This shift in host immunity has also been observed for cows infected with M. paratuberculosis, with strong IFN-γ responses noted in the early stages of disease, followed by an upregulation of IL-10 in the latter stages. 14,27 Although secretion of other Th1-driven cytokines has been studied, the enhanced expression and secretion of IFN-γ in the early stages of infection suggests that this cytokine is critical for controlling mycobacterial infections. The upregulation of IFN-γ also makes it an attractive tool for the detection of subclinical infection in cattle with paratuberculosis. 11,22,24,26 Previously, it has been shown that antigen-specific IFN-γ responses are enhanced after sensitization with mycobacteria, 2,7,30 an effect that could be capitalized on to improve the sensitivity of the assay for the detection of infected animals. The present study was designed to evaluate the effects of intradermal injection with johnin purified protein derivative (JPPD) on secreted and intracellular IFN-γ production in healthy noninfected cows and cows with subclinical and clinical paratuberculosis. In addition, the effects of in vivo sensitization on cell phenotypes present in the induration and in the peripheral blood of healthy and infected cattle were determined.
Materials and methods
Animals
Three cows for each of the following treatment groups were used in this study: noninfected healthy cows, cows naturally infected with M. paratuberculosis but asymptomatic (i.e., subclinical), and naturally infected cows with the clinical form of Johne disease. Cows used in the study ranged in age from 3 to 6 years. Infection was monitored by bacteriological culture of M. paratuberculosis from fecal samples using a standard method previously described. 23 By definition, clinical animals were shedding more than 100 colony forming units (CFU) per gram of feces and presented with weight loss and intermittent diarrhea. Subclinically infected cows were shedding less than 10 CFU/g of feces. The healthy control cows were purchased from herds with no history of paratuberculosis and were characterized by repeated negative fecal cultures. In addition, these animals were negative on any serological assays (i.e., production of antibody specific for M. paratuberculosis and IFN-γ) performed during that period. All animals in the study were pretested using the IFN-γ assay between 7 and 14 days prior to initiation of the study. IFN-γ responses were consistently negative for control cows and clinically infected cows on 2 sampling dates. Procedures outlined in this report were approved by the Institutional Animal Care and Use Committee of the National Animal Disease Center (Ames, Iowa) prior to initiation of the study.
Experimental protocol
Cows were bled on day 0 (just prior to the intradermal injection), and on days 2, 4, 7, 10, 14, and 21 after intradermal injection. Blood was collected into vacutainer tubes containing sodium heparin for IFN-γ analysis and acid-citrate-dextrose for flow cytometric phenotype analyses. Pre-injection skin thickness measurements were taken with calipers and cows were given intradermal injections of JPPD a in 0.1 ml (100 μg protein) volumes in the cervical region. After 72 hr, changes in skin thickness at the site of injection were recorded. Fine needle aspiration of the cells at the site of the injection was performed at the same time with a 26-G needle and a 3-ml syringe containing 1 ml of phosphate-buffered saline (0.15 M, pH 7.4) with 1-mM dithiothreotol b (DTT). Flow cytometric phenotype analyses were then performed on the cell aspirates.
IFN-γ ELISA
Samples of whole blood (1 ml) were added to 24-well tissue culture plates and incubated either alone (no stimulation), or with concanavalin A b (ConA; 10 μg/ml), pokeweed mitogen b (PWM; 10 μg/ml), JPPD a (10 μg/ml), or a whole cell sonicate of M. paratuberculosis c (MPS; 10 μg/ml) for 18 hr at 39°Cin 5% CO2. Plates were centrifuged at 500 × g for 15 min and plasma was harvested from each well. Plasma samples were frozen at −20°C until analyzed for IFN-γ concentrations by ELISA using a commercial kit. d Results are expressed as optical density units at 450 nm. A sample was determined to be positive if the absorbance of the stimulated sample (either mitogen or antigen) was 0.100 absorbance units greater than the absorbance achieved for the nonstimulated control well for that animal. 26
Flow cytometric analysis of cell surface antigens on leukocytes
Blood samples were collected into vacutainer tubes containing acid-citrate-dextrose (ACD) as the anticoagulant e and processed as follows for 4-color flow cytometric analysis in order to differentiate immune cells. Whole blood (75 μl) was dispensed into 96-well U-bottom microtiter plates. Red blood cells were lysed with 200 μl of distilled water for 10 sec followed by restoration of isotonicity with 20 μlof10x PBS. After centrifugation at 600 × g for 5 min and decantation, 50 μl each of primary antibodies f listed in Table 1 (except the monocyte marker) were added into appropriate wells and incubated at room temperature for 15 min in the dark. Following centrifugation and decantation, 100 μl per well of secondary antibody cocktail was added, and incubated at room temperature for 15 min in the dark. Secondary antibody cocktail consisted of goat anti-mouse IgG3-FITC (fluorescein conjugate; 1:300) g ; goat anti-mouse IgG2a-PE (phycoerythrin conjugate; 1:60) g ; rat anti-mouse IgG1-PerCP (peridinin chlorophyll conjugate; 1:30) h ; and goat anti-mouse IgM-APC (allophycocyanin conjugate; 1:40). i After centrifugation and decantation, cells were suspended with 200 μl per well of FacsLyse h and kept at 4°C until analysis. Data were acquired as 5,000 events/sample using Becton-Dickinson LSR flow cytometer and CellQuest software. h
For the fine needle aspirates, cells were processed in the same manner as above without hemolysis. Analysis was limited to the expression of CD45RO, CD62L, LFA-1, CD4, CD8, and N12 (only for infected cow samples) owing to the limited number of cells.
Flow cytometric analysis of cell surface antigens and intracellular cytokines
Each heparinized blood sample was divided into 3 aliquots of 1 ml and incubated for 20 hr at 39°C in a capped polystyrene tube alone (no stimulation), with PWM (10 μg/ml final concentration), or JPPD (10 μg/ml).
Brefeldin A b (final concentration of 10 μg/ml) was added 4 hr prior to the termination of stimulation. Stimulated whole blood (100 μl) was incubated with 50 μl each of the primary monoclonal antibodies f for phenotype analysis (CD4, CD8, monocyte, and CD45RO in Table 1) for 15 min at room temperature in a microtiter plate (U-bottom, 96-well). After centrifugation at 600 × g for 5 min the supernatant was decanted, red blood cells were hemolyzed with 200 μl of hypotonic saline (10.6 mM Na2HPO4, 2.7 mM NaH2PO4) followed by restoration with 20 μl of hypertonic saline (10.6 mM Na2HPO4, 2.7 mM NaH2PO4, 462 mM NaCl). The peripheral blood mononuclear cells (PBMC) were then washed once with 200 ml/well of PBS. Cells were then incubated at room temperature in the dark for 10 min with 50 ml each of the secondary mAbs (rat anti mouse IgG1 -PerCP h [1:10]; and goat anti-mouse IgM-APC [1:125]). After incubation and 1 wash with PBS, the cells were incubated with 50 μl/well of Fixation Medium i for 15 min at room temperature in the dark. After 1 wash with PBS, 100 μl of permeabilization medium i and 10 μl each of PE-conjugated mAb for IFN-γ j (Clone CC302, 2.5 μg/ml working concentration) or isotype control h (MOPC-21, 2.5 μg/ml working concentration) were added. Cells were incubated for 15 min at room temperature in the dark. After 2 washes with PBS, cells were resuspended with 200 μl of PBS. Data were acquired for 20,000 events/sample immediately using a Becton-Dickinson LSR flow cytometer and Cell Quest software. h
Working concentrations and description of primary monoclonal antibodies used for cell analyses.

For the fine needle aspirates, cells were processed in the same manner as above without hemolysis. Analysis was limited to the expression of CD45RO, CD2L, LFA-1, CD4, CD8, and N12 (only for infected cow samples) owing to the limited number of cells.
Statistical analyses
Data were analyzed by repeated measures using PROC MIXED procedure of the Statistical Analysis System (SAS) k . Means and SEM are reported for all data. When significant effects (P <0.05) owing to treatment, day or treatment × day interactions were detected, means separation was conducted by the Tukey-Kramer option in SAS.
Results
Results of the intradermal injection with JPPD are presented in Table 2. Healthy noninfected control cows and cows with clinical M. paratuberculosis infection demonstrated only negligible increases in skin thickness (0.3-0.5 mm) after the intradermal skin test. In contrast, significant (P < 0.05) increases in swelling (9.3-15.6 mm) were noted for the subclinically infected cows in this study.
Infection status (healthy, subclinical, or clinical) or in vivo sensitization via the intradermal injection did not affect the secretion of IFN-γ in nonstimulated cultures from PBMC (data not shown). Intradermal injection with JPPD did significantly increase (P < 0.05) secretion of IFN-γ by PBMC after stimulation of cells with PWM in both subclinically and clinically infected cows by day 2 postinfection (Fig. 1A). In contrast, subclinically infected cows demonstrated the most noticeable increase in ConA-mediated IFN-γ production by PBMC during the study with significant (P < 0.05) increases observed on days 7, 10, and 21 as compared to day 0 (Fig. 1B).
Differences in antigen-specific IFN-γ production were more marked than mitogen-induced IFN-γ, with higher (P < 0.05) IFN-γ production noted for the subclinically infected cows throughout the study when compared to the healthy and clinical cows (Fig. 2A, 2B). Responses to MpS were fairly robust for subclinically infected cows prior to intradermal injection on day 0 (0.339) as compared to values for control and clinically infected cows (0.145 and 0.087). In contrast, IFN-γ secretion after stimulation of PBMC with JPPD on day 0 was not different between treatment groups. Although IFN-γ levels began to increase for subclinical cows within 2 days of intradermal injection, significant (P < 0.05) differences between pre- and postinjection IFN-γ were noted only on days 7 and 10. Interestingly, the pattern of IFN-γ secretion and effects owing to the skin test after in vitro stimulation of cells with either antigen preparation (MpS or JPPD) closely simulated the ConA-responses noted for cows in the subclinically infected treatment group.
Effect of infection status of cows on induration size 72 hours after intradermal injection with johnin purified protein derivative. *
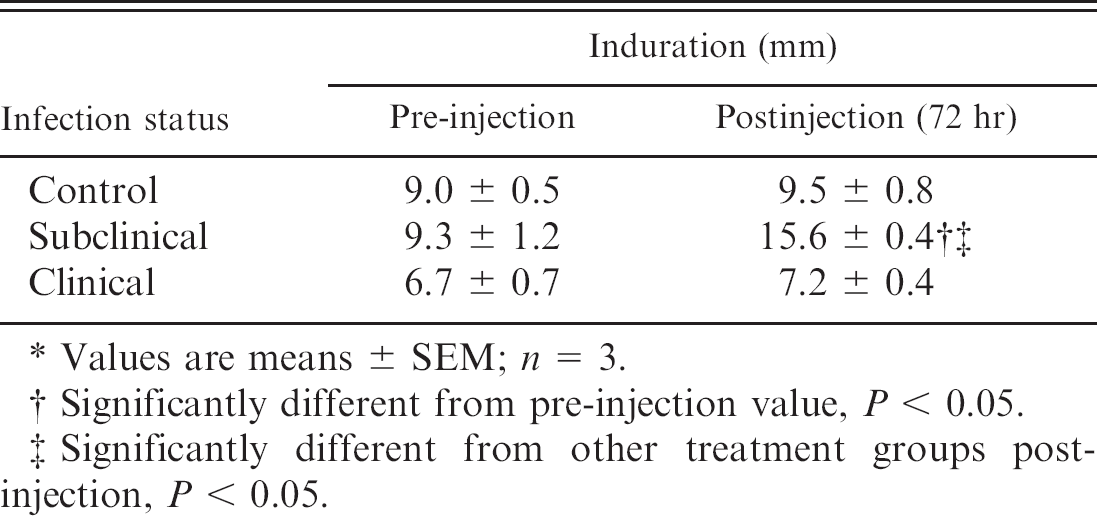
Values are means ± SEM; n = 3.
Significantly different from pre-injection value, P <0.05.
Significantly different from other treatment groups postinjection, P < 0.05.
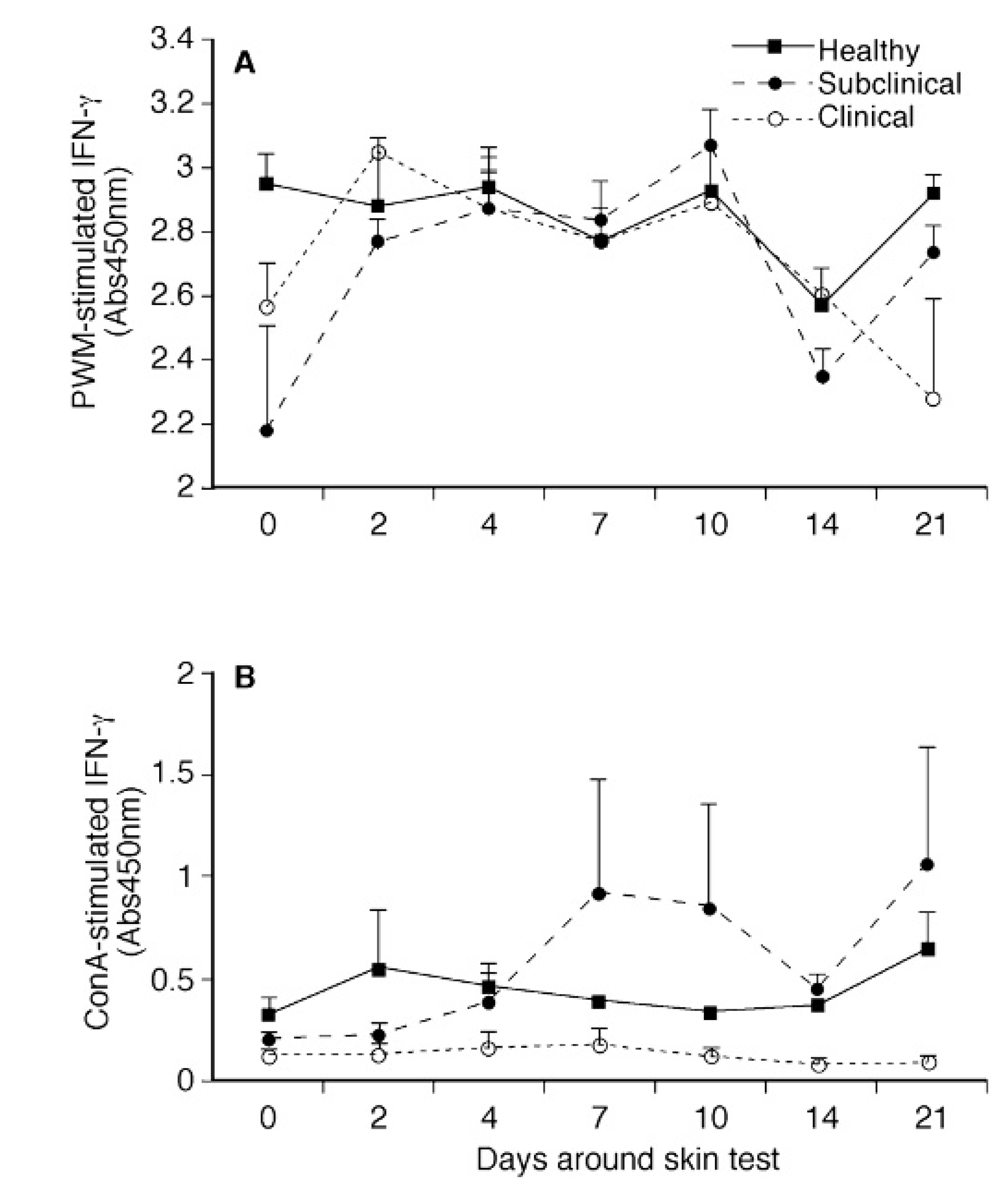
Effects of intradermal sensitization with johnin purified protein derivative on interferon (IFN)-γ production by PBMC isolated from healthy, subclinically, and clinically infected cows after in vitro stimulation with pokeweed mitogen (PWM;
Subpopulations of CD3+, CD4+, and CD8+ lymphocytes within whole blood were consistently (P >0.05) higher for infected cows compared to healthy controls throughout the study (Fig. 3A, 3B, 3C). Responses to the intradermal injection with JPPD were noted by increased numbers of CD3+ cells in each treatment group (Fig. 3A). Healthy control cows also demonstrated a significant (P < 0.05) increase in CD8+ cells post-injection that was not noted in the infected cows (Fig. 3C). The γδ TCR+ population did not differ between treatment groups but did decline by day 10 after intradermal injection, regardless of infection status (data not shown).
Although infected cows (subclinical and clinical) had higher (P < 0.05) CD45RO expression on PBMC compared to control cows during much of the study, expression for control cows increased from 16.3% on day 0 to 60.5% by day 21 and was higher than for either infected group (Fig. 4A, 4B, 4C). A 3-fold increase in CD45RO expression on CD8+ cells isolated from control cows was noted on day 21 compared to baseline values and may have contributed to this increase. Similar increases in CD45RO expression on CD8+ cells were noted for infected cattle by day 21 and it is likely that these increases were owing to sensitization with JPPD. A significant (P < 0.05) increase in CD45RO expression on CD4+ cells was noted for subclinically infected cows within 2 days postinjection and remained upregulated throughout the study (Fig. 4B). The percentage of total PBMC that expressed CD25 did not differ between animal groups and was not affected by the intradermal injection, however, CD25 expression within the CD4+ subpopulation was significantly (P < 0.05) higher for infected cows compared to healthy controls at all time points in the study (Fig. 5). In addition, sensitization with the JPPD did result in increased percentages of CD4+/CD25+ cells over the period of the study.
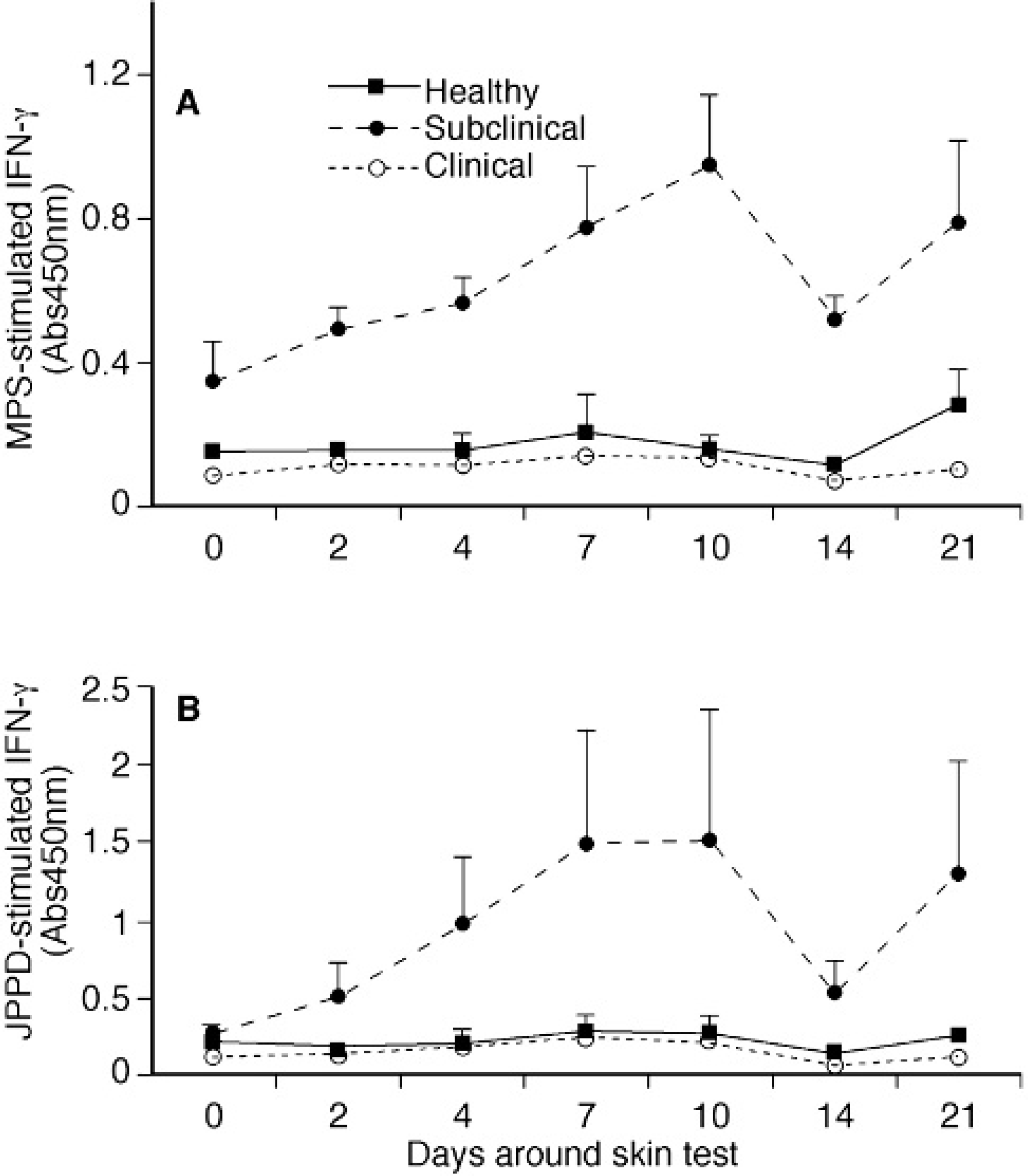
Effects of intradermal sensitization with johnin purified protein derivative on interferon (IFN)-γ production by PBMC isolated from healthy, subclinically, and clinically infected cows after in vitro stimulation with johnin purified protein derivative (JPPD;
The percentage of CD4+ and CD8+ T cells within the induration 72 hours after intradermal injection with JPPD was significantly (P < 0.05) lower for the subclinically infected cows compared to healthy and clinically infected cows (Table 3). In addition, the CD8+ subpopulation was significantly (P < 0.05) lower than the CD4+ subpopulation for subclinically infected cows, whereas, control and clinically infected cows had similar numbers of CD4+ and CD8+ cells. Expression of CD45RO and LFA on the total cell population was significantly (P < 0.05) lower for subclinically infected cows as compared to healthy and clinically infected cows (data not shown). Similar reductions in the expression of CD45RO, LFA, and CD62L markers on the CD4+ and CD8+ subpopulations were also noted for subclinically infected cows, however, these effects were significant (P < 0.05) only for the CD8+ population (Table 3). Because of the low cell recovery from the indurations of healthy and clinically infected cows, γδ5 T-cell subpopulations were not adequately represented across treatments, however, cell staining data did reflect a decrease in expression of the cell surface markers for subclinically infected cows compared to the cows with clinical disease.
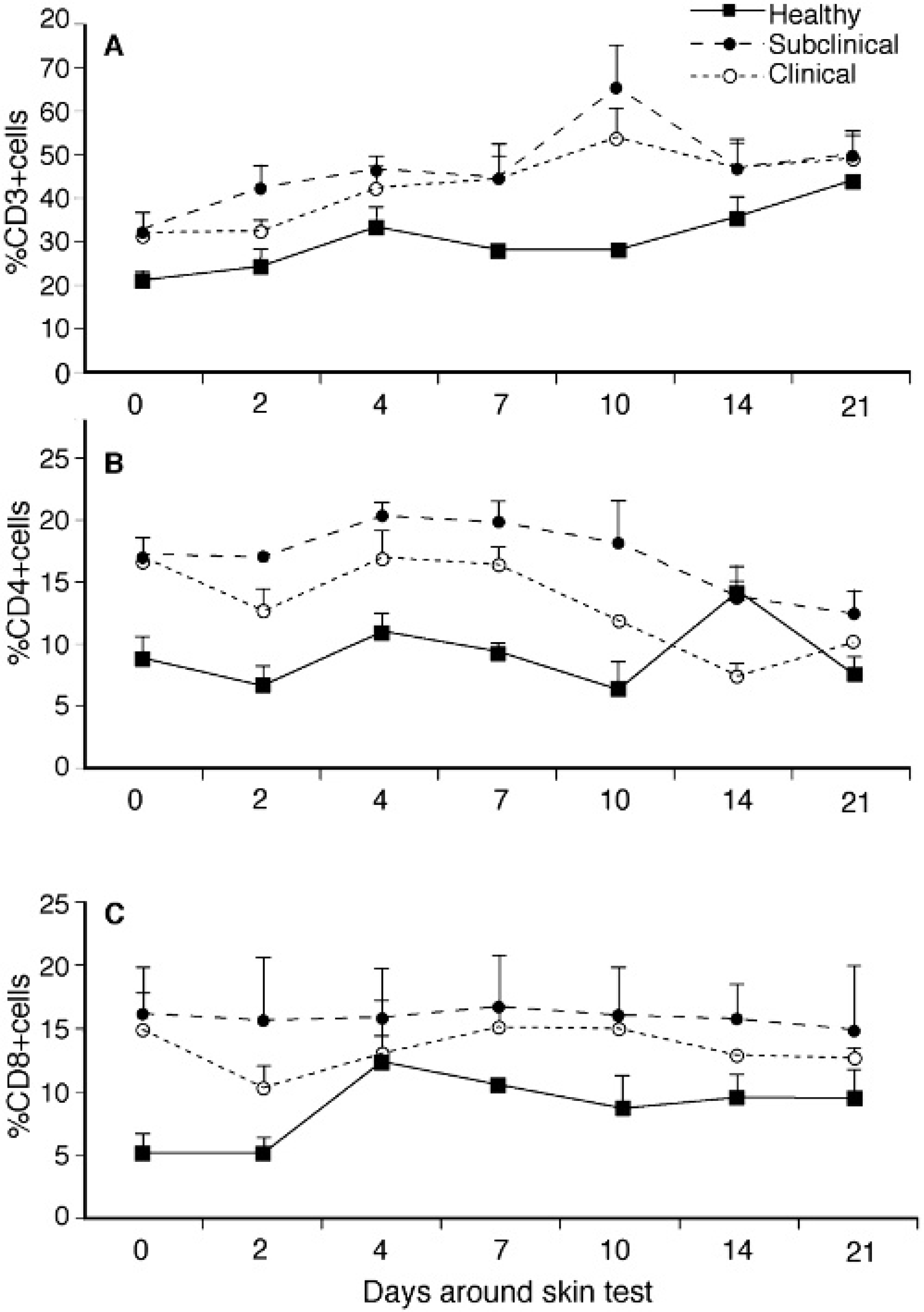
Modulation of T-cell phenotypes within PBMC isolated from healthy, subclinically, and clinically infected cows after intradermal sensitization with johnin purified protein derivative. Values are presented% of total PBMC population; means ± SEM; n = 3 (CD3+,
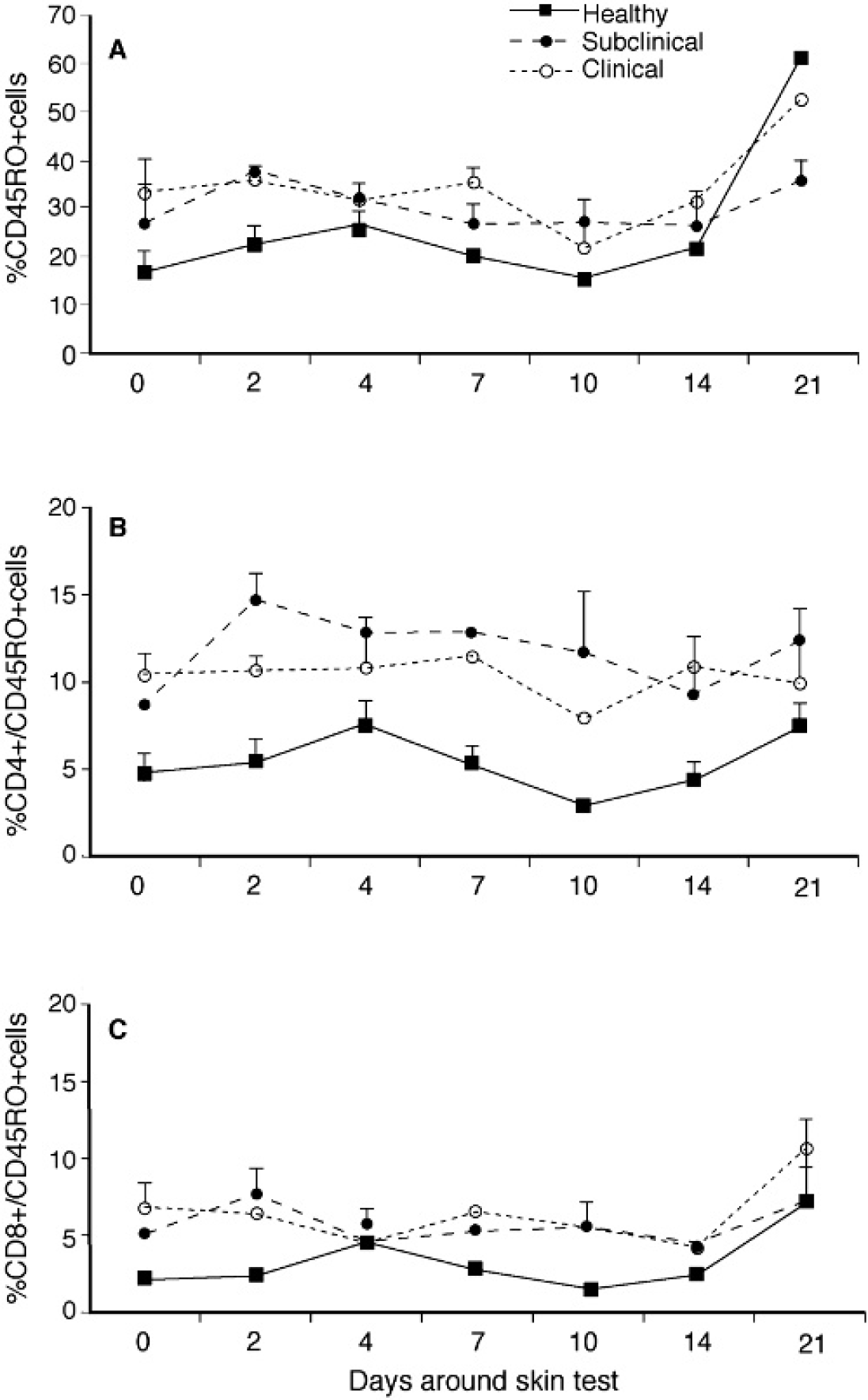
Expression of CD45RO within total PBMC population
A transient increase in intracellular IFN-γ was observed (P < 0.05) in nonstimulated cells from all cows on day 4 after the skin test (data not shown). Stimulation of cells with PWM induced 5- to 13-fold increases in intracellular IFN-γ expression compared to nonstimulated cells, with effects owing to the intradermal injection noted on days 7 and 10 of the study for the 3 treatment groups (data not shown).
The intradermal skin test did not affect the expression of intracellular IFN-γ in CD4+ and CD8+ T cells so results were averaged over the study. Intracellular IFN-γ within CD4+ and CD8+ subpopulations was significantly affected by infection status of the cows but only in the nonstimulated cells, with higher (P < 0.05) levels of IFN-γ noted for infected cows compared to the healthy controls in both T-cell subsets (data not shown). When further delineated by CD45RO expression within T-cell subsets (Fig. 6), intracellular IFN-γ was predominantly expressed within the CD8+ subset for sub-clinically and clinically infected cows. Clinically infected cows also had higher (P < 0.05) intracellular IFN-γ expression in the CD4+ subpopulation compared to subclinically infected cows and healthy controls (Fig. 6A). After stimulation of PBMC with JPPD, intracellular IFN-γ was significantly lower (P < 0.05) in CD4+/CD45RO+ cells from clinically infected cows compared to the control and sub-clinically infected cows (Fig. 6B).
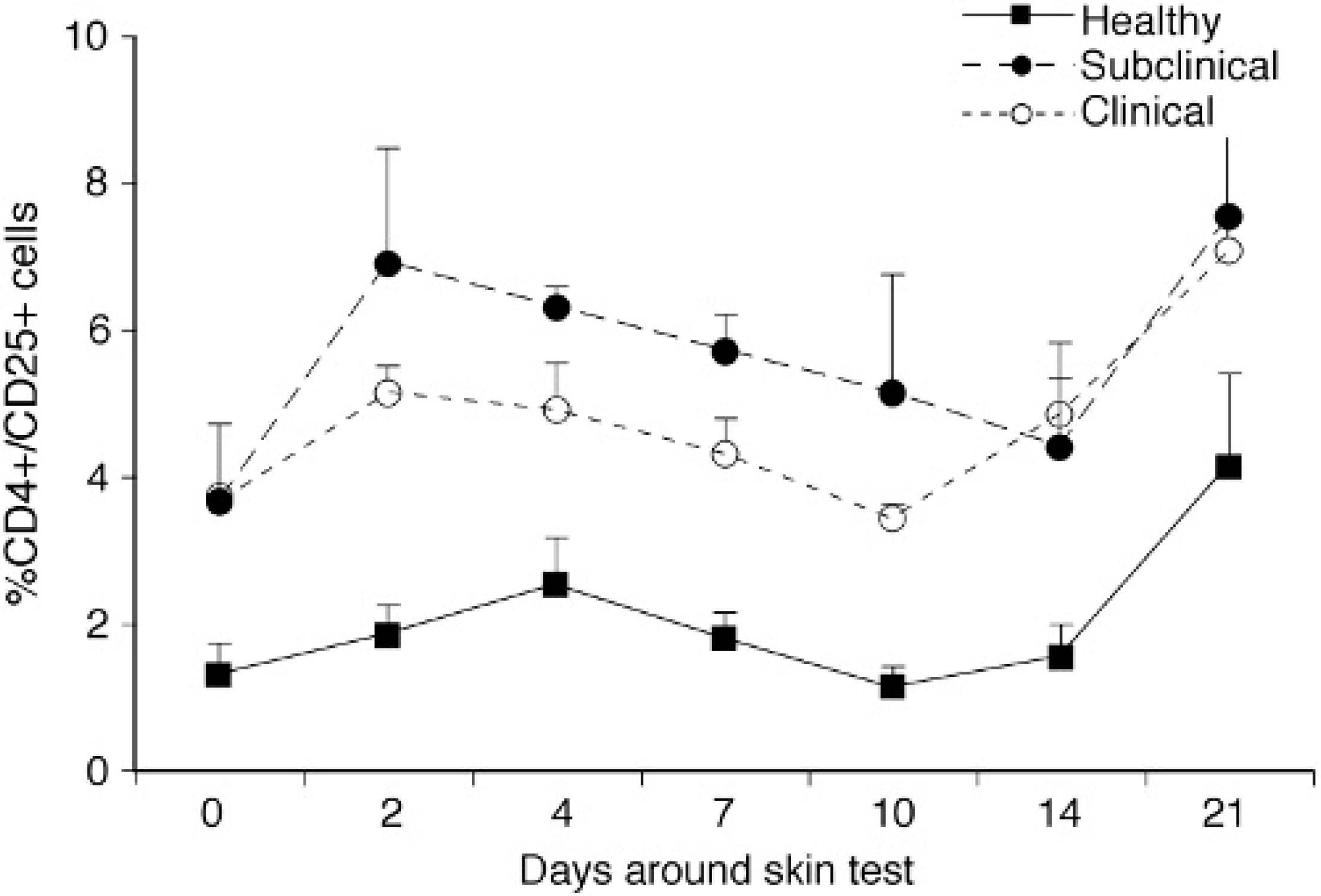
Percentage of total PBMC expressing CD4+/CD25+ for healthy, subclinically, and clinically infected cows after intradermal sensitization with johnin purified protein derivative. Values are presented as means ± SEM; n = 3.
Discussion
The effectiveness of diagnostic tools for the detection of animals infected with M. paratuberculosis is greatly affected by the biology of the disease. The progression from asymptomatic subclinical infection to a clinical disease state characterized by a classical protein-losing enteropathy corresponds with a concomitant shift from Th1-driven cellular immunity to Th2-mediated humoral immunity in the host. This precludes the sole use of a serological diagnostic tool such as the ELISA test to identify animals in both disease states. Yet the ability to detect M. paratuberculosis infection in the early stages of infection is paramount to controlling the spread of this disease. Measurement of IFN-γ production in response to in vitro mycobacterial antigen stimulation has been used successfully as an indicator of infection with M. tuberculosis, M. bovis, and M. paratuberculosis. 11,20,22 In addition, studies with M. tuberculosis and M. bovis demonstrated that prior sensitization to the mycobacterial antigen by skin test or BCG vaccination enhanced the sensitivity of detection with the IFN-γ assay. 2,30 Revaccination with BCG significantly enhanced the IFN-γ response to M. tuberculosis in children (7-15 years) that were classified as low IFN-γ responders. 2 As part of the study, the intradermal skin test was performed on day 0, prior to revaccination. Although the effects of revaccination and the skin test were not independently analyzed, the study demonstrates that re-exposure to the antigen resulted in a significant improvement in IFN-γ release.
Data are expressed as a% of total cell population in the aspirate obtained from the induration.
Mean ± SEM; n = 3.
Not determined.
Superscript denotes significant differences between infection groups, P < 0.05.
n = 1.
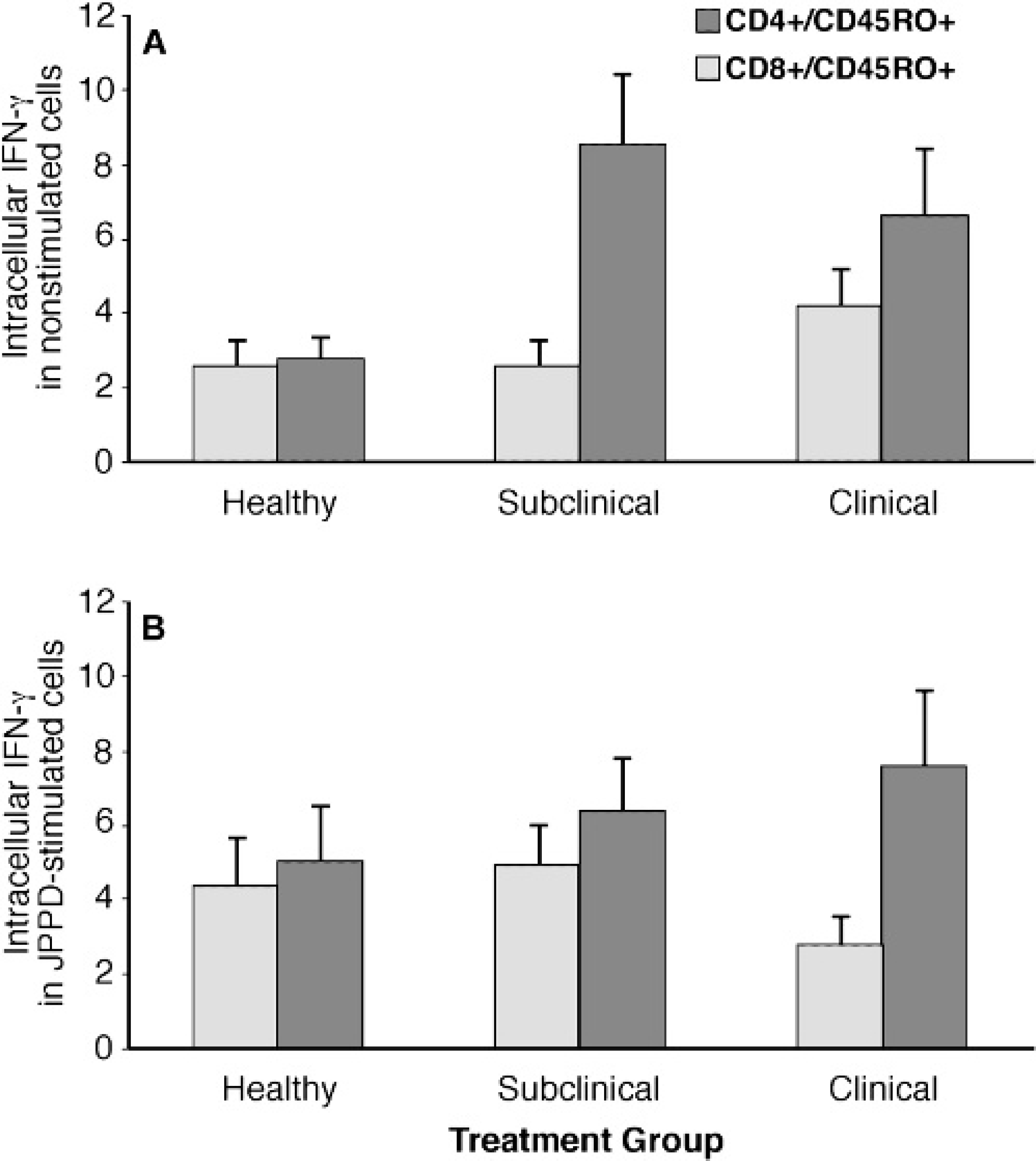
Expression of intracellular interferon-γ in CD4+/CD45RO+ and CD8+/CD45RO+ subsets isolated from healthy, subclinically, and clinically infected cows after intradermal sensitization with johnin purified protein derivative. PBMC were nonstimulated
Subclinically infected cows had positive pre-injection IFN-γ responses, averaging 0.242 and 0.295 Abs450nm for MpS and JPPD antigens, respectively, on day 0. After the intradermal injection increases in antigen-specific IFN-γ to 0.903 (206%) and 1.486 Abs450nm (514%) were achieved by day 10. The transient nature of the IFN-γ response upon re-exposure to the mycobacterial antigen has also been observed in calves experimentally infected with M. bovis and administered the caudal fold test. 19 The caudal fold test boosted the IFN-γ response within 3 days postinjection but responses had returned to baseline by day 7. Interestingly, administration of the comparative cervical test 7 and 55 days after the caudal fold test did not result in any further increase in IFN-γ production. It would appear that re-exposure to the antigen will enhance IFN-γ production but the effects are short-lived.
Although subclinical cows had a positive IFN-γ response to both antigen preparations on day 0, the IFN-γ response to JPPD in these cows did not differ from the control or clinical cows. Therefore, if one were to assess the infectivity of the subclinically infected cows based upon their IFN-γ response to the JPPD preparation on a comparative basis with the known negative animals, one would surmise that the reactions were nonspecific and erroneously suggest that these cows were negative. Since PPD preparations are more generally used in the IFN-γ assay for a wide variety of species, this is an important point. In this study, intradermal injection boosted the IFN-γ response to the JPPD above baseline values, providing a clear distinction in infectivity status between the subclinical cows and the noninfected controls. Control cows and clinically infected cows did not respond to the intradermal injection in this manner. A nonspecific increase in IFN-γ production to MpS that cannot be explained was observed on day 21 of the study in 2 of the 3 control cows.
In the present study, intradermal injection with JPPD resulted in a significant increase on days 7 and 10 postinjection in secreted IFN-γ after in vitro stimulation of PBMC isolated from subclinically infected cows with either MpS or JPPD antigen preparations. Little or no effect of the sensitization was observed in healthy cows or cows in the clinical stage of disease. The lack of response in control animals would indicate that they did not have appropriate antigenic recall mechanisms since they were not previously exposed to M. paratuberculosis. However, IFN-γ secretion was likely attenuated in clinically infected cows through increased expression of IL-10, a mechanism that has previously been described to downregulate IFN-γ responses in cows infected with M. paratuberculosis. 15 Interestingly, sensitization with JPPD did not markedly affect the amount of PWM-mediated IFN-γ production for the 3 treatment groups during the study but it did increase IFN-γ secretion in response to ConA in subclinically infected cows on the same days in which effects were noted on antigen-specific IFN-γ. This would suggest that the T-cell population responding to the antigen preparations corresponded more closely with the T-cell population that reacted to ConA stimulation. Unfortunately, further characterization of this population was not performed in the present study so it was not possible to distinguish whether CD4+, CD8+, γδ subpopulations, or a combination of them were responsible for the increased IFN-γ production after the skin test was performed.
It is highly likely that each of these subpopulations contributed to the secretion of IFN-γ, however, it has been documented that CD4+ cells are the major T-cell population responsible for IFN-γ secretion in cattle infected with M. paratuberculosis. 3
Infected cows had increased frequency of CD4+ and CD8+ T cells in total PBMC compared to healthy controls with equivalent percentages of both within each treatment group. However, further delineation of the CD4+ subset demonstrated increased expression of CD25 and CD45RO, suggesting that these cells were highly activated and that prior exposure to M. paratuberculosis via natural infection fostered an effector-memory phenotype within the CD4+ subpopulation. Similar antigen-recall effects have been observed after antigenic stimulation of PBMC isolated from calves that were experimentally inoculated with M. paratuberculosis. 17,28 In both studies the predominant phenotype present within PBMC cultures after in vitro stimulation with antigens was CD4+ T cells expressing CD45RO and CD26 markers. These results are important since the CD4+ subpopulation is a major source of IFN-γ and the enhanced expression and secretion of IFN-γ in the early stages of infection suggest that this cytokine is critical for controlling mycobacterial infections. 6 In addition, it has recently been shown that CD8+ T cells also participate in memory responses to M. tuberculosis upon re-exposure of mice. Both CD4+ and CD8+ cells were equally responsive in IFN-γ production after challenge inoculation of mice with live M. tuberculosis. 21
Intracellular staining for IFN-γ within total PBMC populations was enhanced by intradermal injection depending upon the treatment group and the in vitro stimulant. When segregated into CD4+ and CD8+ subsets neither subpopulation consistently dominated over the other in expression of intracellular IFN-γ. These data are supported by a study that evaluated patterns of intracellular cytokine expression in CD4 and CD8 cells from patients infected with Mycobacterium leprae 1 but contrast with data from 2 recent studies reporting that the CD4+ subset was responsible for the majority of intracellular IFN-γ noted in antigen-stimulated PBMC obtained from either vaccinated goats or calves experimentally infected with M. paratuberculosis. 9,28 In fact, in the present study separation of the CD4+ and CD8+ subsets by CD45RO expression demonstrated that CD8+/CD45RO+ cells had higher expression of intracellular IFN-γ than CD4+/CD45RO+ cells. These data are supported by a study that evaluated IFN-γ production by both CD4+ and CD8+ lymphocytes in cattle following vaccination with BCG. 10 Stimulation of PBMC with BCG-infected dendritic cells induced higher levels of secreted and intracellular IFN-γ for CD8+ cells than CD4+ cells. In addition, human subjects immunized with BCG had high numbers of CD8+ cells that produced IFN-γ in response to BCG-infected dendritic cells. 13 Characterization of the cell phenotypes within the induration in the present study showed that reduced expression of CD45RO, LFA-1, and CD62L in the total cell population paralleled similar reductions within the CD4+ and CD8+ subpopulations yet effects were more dramatic for CD8+ cells. These data clearly indicate that CD8+ lymphocytes play a critical yet uncharacterized role in host immune responses to M. paratuberculosis, perhaps equivalent to that of CD4+-mediated protection. It also seems likely that primed dendritic cells are mediating, at least in part, CD4+ and CD8+- generated host defenses to M. paratuberculosis infection. Although not measured, the reduction in the percentage of CD4+ and CD8+ cells present in the induration of subclinically infected cows may be owing to an increase not only in monocyte/macrophages as is typical in delayed-type hypersensitivity reactions but also in the number of dendritic cells. In the present study, cells obtained from cows had multiple exposures to antigen including natural infection, intradermal sensitization, and in vitro stimulation of PBMC. Multiple exposures to antigen in an experimental situation may prompt a switch from CD4+ to CD8+ phenotypes similar to that noted in the natural disease progression. 4,18
In summary, intradermal sensitization of subclinically infected cows dramatically increased IFN-γ secretion by cultured PBMC after antigenic stimulation on days 7 and 10 postinjection, suggesting that the sensitivity of this assay can be increased for diagnostic purposes. Based upon the measurement of intracellular IFN-γ, it is also apparent that CD8+ T cells mediate host responses after exposure to M. paratuberculosis antigens but it is unclear as to their specific role. The present study was constrained by the number of animals per treatment group, owing to the limited availability of clinically infected cows at that time. Therefore, although statistical significance was achieved within the study with the small group size, interpretation of the data should be done with caution. Further studies need to be conducted to elucidate the roles of T-cell subsets during the progression of natural infection and their contribution to protection or exacerbation of disease.
Acknowledgements
The authors wish to thank Trudy L. Tatum and Bruce Pesch for their excellent technical assistance and Donnie Robinson (in memoriam) and Tim Gogerty for their care of the experimental animals.
Footnotes
a.
National Veterinary Services Laboratory, Ames, IA.
b.
Sigma Chemical Corp., St. Louis, MO.
c.
National Animal Disease Center, Ames, IA.
d.
Bovigam, BioCor, Omaha, NE.
e.
BD Vacutainer Systems, Franklin Lakes, NJ.
f.
VMRD, Pullman, WA.
g.
Southern Biotechnology, Birmingham, AL.
h.
BD Biosciences, San Diego, CA.
i.
Caltag, Burlingame, CA.
j.
Serotec, Raleigh, NC.
k.
SAS, Cary, NC.

