Abstract
A comparative serological study was conducted using the Leptospira microscopic agglutination test (MAT). Genotypes hardjoprajitno (HP), hardjo-bovis A (HA), and hardjo-bovis B (HB) were compared to determine which best detects hardjo antibody in cattle serum. A total of 2,431 cattle sera were tested. Sera were collected from 4 geographic regions of the United States. Samples were obtained without knowledge of breed, age, vaccination history, or herd health status. Of the sera collected, 60.7% (1,475) were negative at the 1:100 dilution for all three genotypes. Serological reactivity at the 1:100 dilution was identified in 956 (39.3%) of the sera tested. Considering the 956 positive sera, 941 (98.4%) reacted to HP, whereas the remaining 15 sera (1.6%) reacted to only HA and/or HB. A total of 394/941 (41.9%) HP positive sera failed to react to HA or HB. The results of this study support the conclusion that HP antigen was most sensitive in detection of hardjo antibody.
In 1973 improvements were made in the microscopic agglutination test (MAT). 2 The MAT has been, and is currently the most widely used method to detect Leptospira antibodies in animal serum. The recommended Leptospira MAT protocol of the US Animal Health Association (USAHA) was published in the 1987 USAHA Proceedings. 1 The protocol contains minor alterations of earlier MAT methods.
As Leptospira isolates were originally recovered from various sources, the MAT was used to group those with moderate antigenic similarity into serogroups. Each serogroup was loosely established based on an isolate's serological reactivity with antisera prepared against other isolates. The cross-agglutinin absorption tests 5 that utilized principles of the MAT enabled further classification of isolates into serogroups. Cross-agglutinin absorption studies of Leptospira were used to maintain culture collections and to identify field isolates. As the list of Leptospira serovars grew to over 200, it became imperative to find a representative serovar for each serogroup to be used in the MAT.
Historically in the United States, Leptospira interrogans, serovar hardjo, genotype hardjoprajitno (HP), has been the representative for the Sejroe serogroup in the MAT. The strain HP was originally isolated from a plantation worker in Sumatra, Indonesia. 10 The HP reference strain has been extensively used in the United States for detection of serovar hardjo antibodies in serum of domestic animals.
With the development of molecular techniques, specifically restriction-endonuclease analysis (REA) 6 or DNA fingerprinting, it was found that Leptospira could be more definitively characterized. A 1986 report 9 found that cattle in the United States were not infected with the hardjo genotype HP, but rather two subtypes designated hardjo-bovis A (HA), and hardjo-bovis B (HB). At the time of that report, it was thought that a third subtype, hardjo-bovis C also existed. This was later found to be in error. 8,12 The HA and HB genotypes were isolated from cattle in the United States during a nationwide survey. 7 However, the HP genotype has not been isolated from cattle in the United States. Current Leptospira bacterins licensed for use in the United States contain either HP, or a single hardjo-bovis genotype. In light of these facts, the question has been raised whether HP is the appropriate genotype to be used in the MAT for the purpose of detecting serovar hardjo antibodies.
To answer this question, three serovar hardjo genotypes were used in a comparative MAT study. Hardjo-bovis HA and HB strains were arbitrarily picked from the National Veterinary Services Laboratories (NVSL) culture collection for use in this study. The HA strain used was NVSL S 1343. This strain was originally isolated from a Texas dairy bull. The HB strain used, NVSL S 818, was isolated from a Texas beef cow. The third hardjo genotype used in the comparative study was the HP reference strain. Serovar hardjo, genotype HP is classified as a member of Leptospira interrogans based on DNA homology, whereas the hardjo-bovis serovars are classified as Leptospira borgpetersenii based on DNA homology.
DNA fingerprinting was used to confirm the identity of each genotype before initiation of the study. Cultures were propagated in liquid polysorbate 80-bovine albumin medium. a Cell suspensions were concentrated to a density of 60–70% transmittance at 600 nm. The cell pellet of a 1.5-ml cell suspension was used for the purpose of DNA extraction. DNA was extracted by use of DNAzol b per manufacturer's instructions. DNA was digested with Hha I restriction endonuclease. 9 Fragments were separated by electrophoresis in a 0.7% agarose gel for 17 hours. Following staining with ethidium bromide, the gel was photographed.
Number of sera from each geographic region and state of origin.
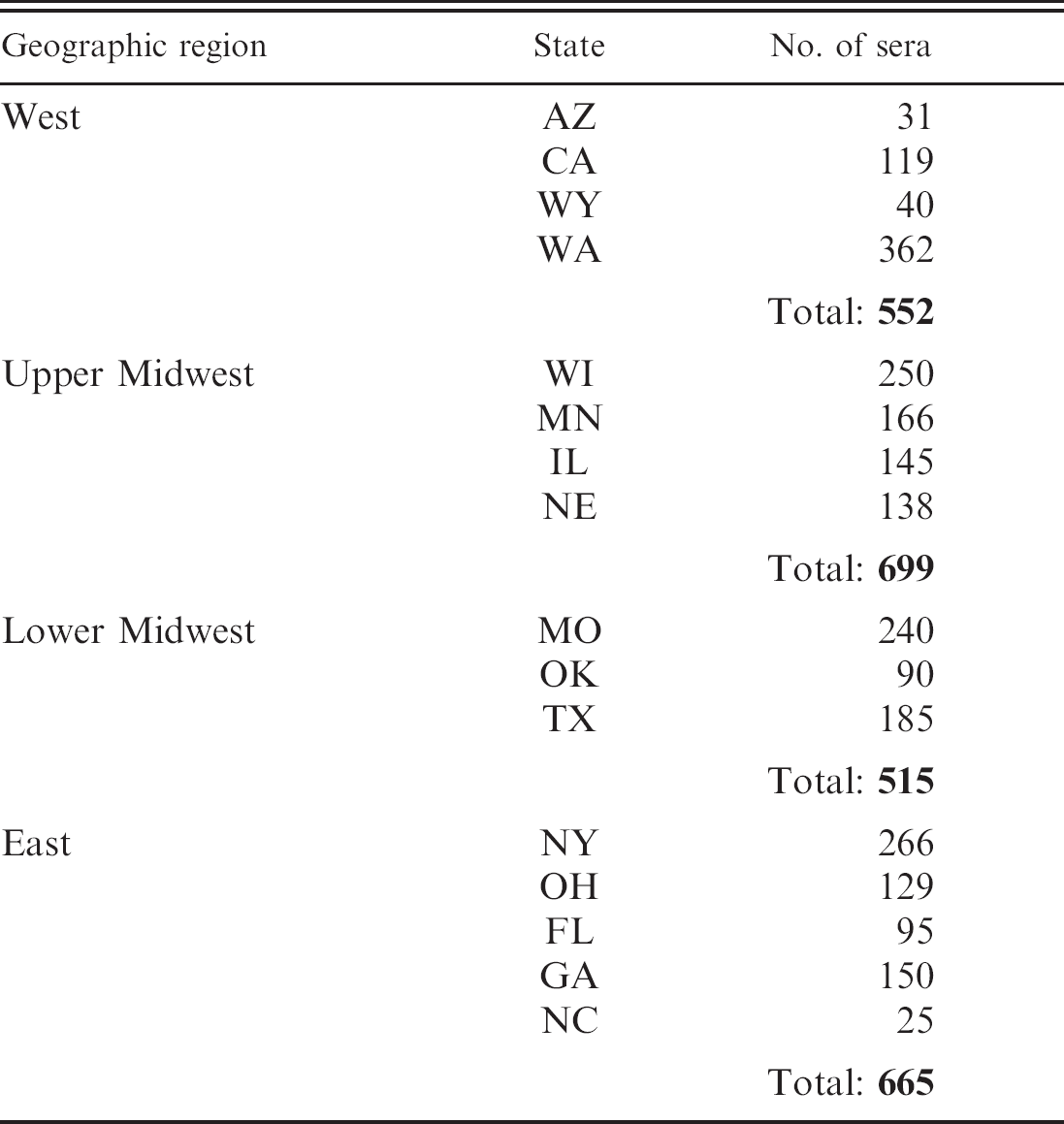
A total of 2,431 cattle sera were collected during calendar year 2004. Herd health status, vaccination history, breed, and age of the animals sampled were unknown. Serum samples were obtained from laboratories in 4 geographic regions of the United States designated as West, upper Midwest, lower Midwest, and East (Table 1). Sera were tested by using the MAT protocol previously described. 1 The method uses a 1:50 serum dilution, which results in an initial test dilution of 1:100. Each serum sample was diluted and simultaneously tested against all 3 antigens. All positive samples were titrated to an end point. End point is defined as 50% or greater agglutination of live Leptospira antigen. Reference antibody controls were run every testing day to ensure the correct antigens were being used and for quality control purposes.
A total of 1,475 (60.7%) sera were negative at the 1:100 dilution for HA, HB, and HP antigen (Table 2). Serological reactivity was detected at the 1:100 dilution in 956 (39.3%) of the sera collected. Of the 956 serologically reactive sera, 941 (98.4%) reacted to HP (Fig. 1). The HA antigen failed to react to 51% of the 956 positive sera, whereas HB failed to react to 51.5%. In contrast, HP missed only 15 (1.6%) of the sera that reacted to HA or HB antigen (Fig. 2). Of these 15 sera, 11 were positive at 1:100 for HA and negative at 1:100 for HB. Two were positive at 1:100 for HB, but negative at 1:100 for HA, and 2 were positive at 1:100 for both HA and HB. Of the 941 HP positive sera, 394 (41.9%) reacted to only HP and failed to react to either HA or HB (Fig. 3).
Distribution of sera that tested negative at the 1:100 dilution against HA, HB, and HP.
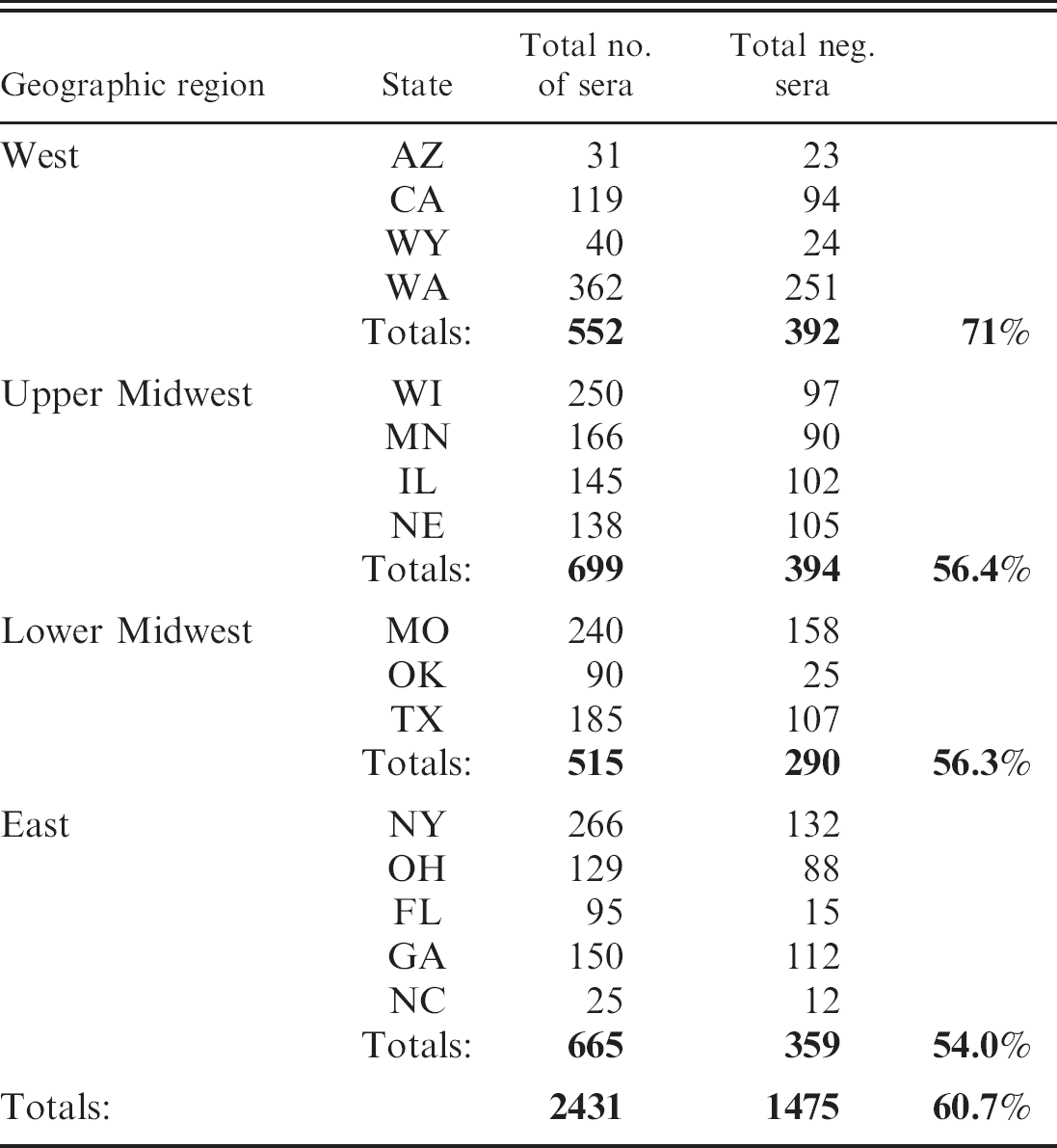
The data from this study should not be considered to reflect the seroprevalence of serovar hardjo within the United States. Because the vaccination and herd health status of all animals sampled was unknown, the data should be considered solely on the merit of comparative testing of HP, HA, and HB antigens.
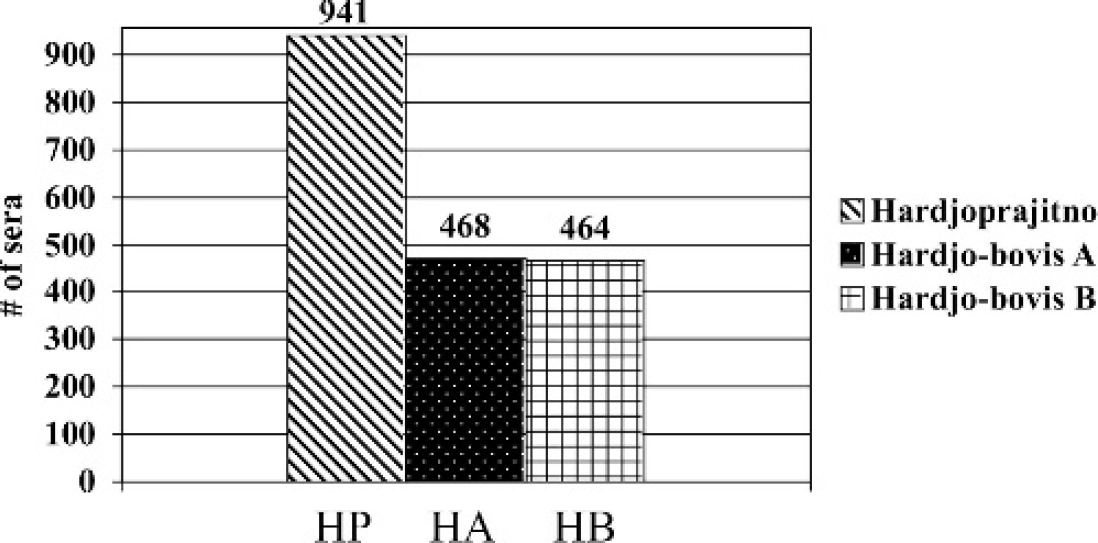
Comparison of serological reactivity using the MAT of 2,431 cattle sera. A total of 956/2,431 sera showed serological reactivity to at least 1 of the 3 serovar hardjo genotypes. Hardjoprajitno reacted to 941 (98.4%) of the positive sera. Hardjo-bovis A had serological reactions to 468/956 (49%) of all positive sera, while hardjo-bovis B had serological reactivity to 464/956 (48.5%) sera.
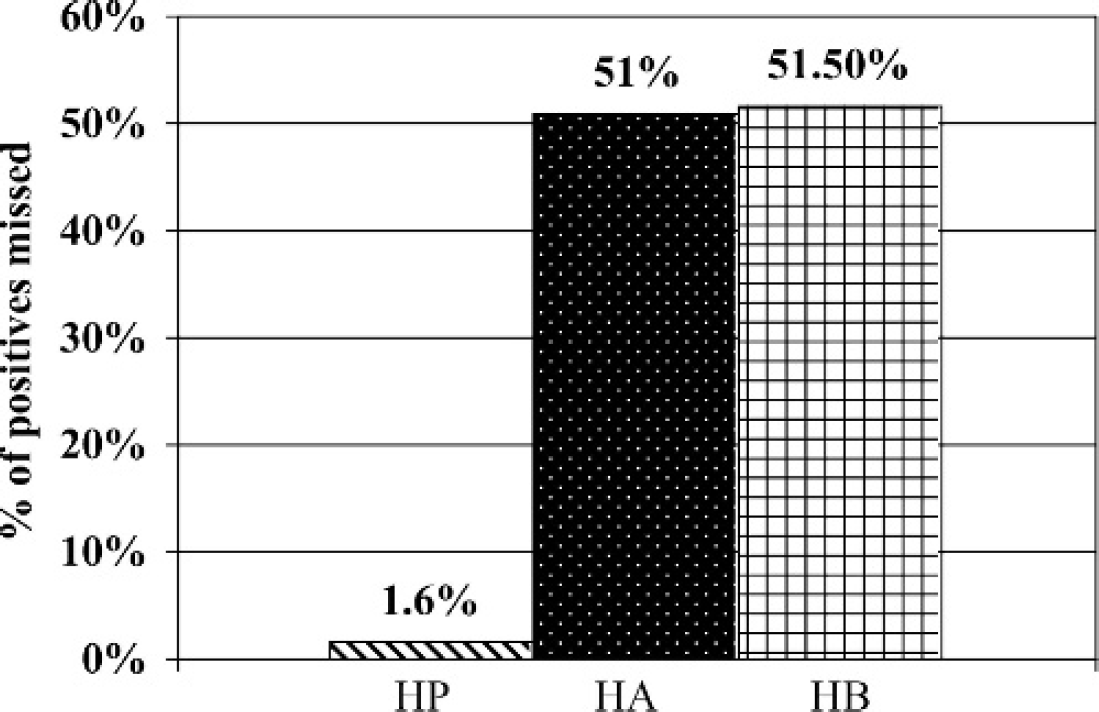
Serological reactivity was detected at the 1:100 dilution in 956 of the 2,431 cattle sera collected. The first column represents the percentage of positive sera that was not detected at the 1:100 dilution by hardjoprajitno (HP). The middle column represents the percentage of positive sera that failed to react to hardjo-bovis A (HA). The last column represents the percentage of positive sera that was not detected at the 1:100 dilution by hardjo-bovis B(HB).
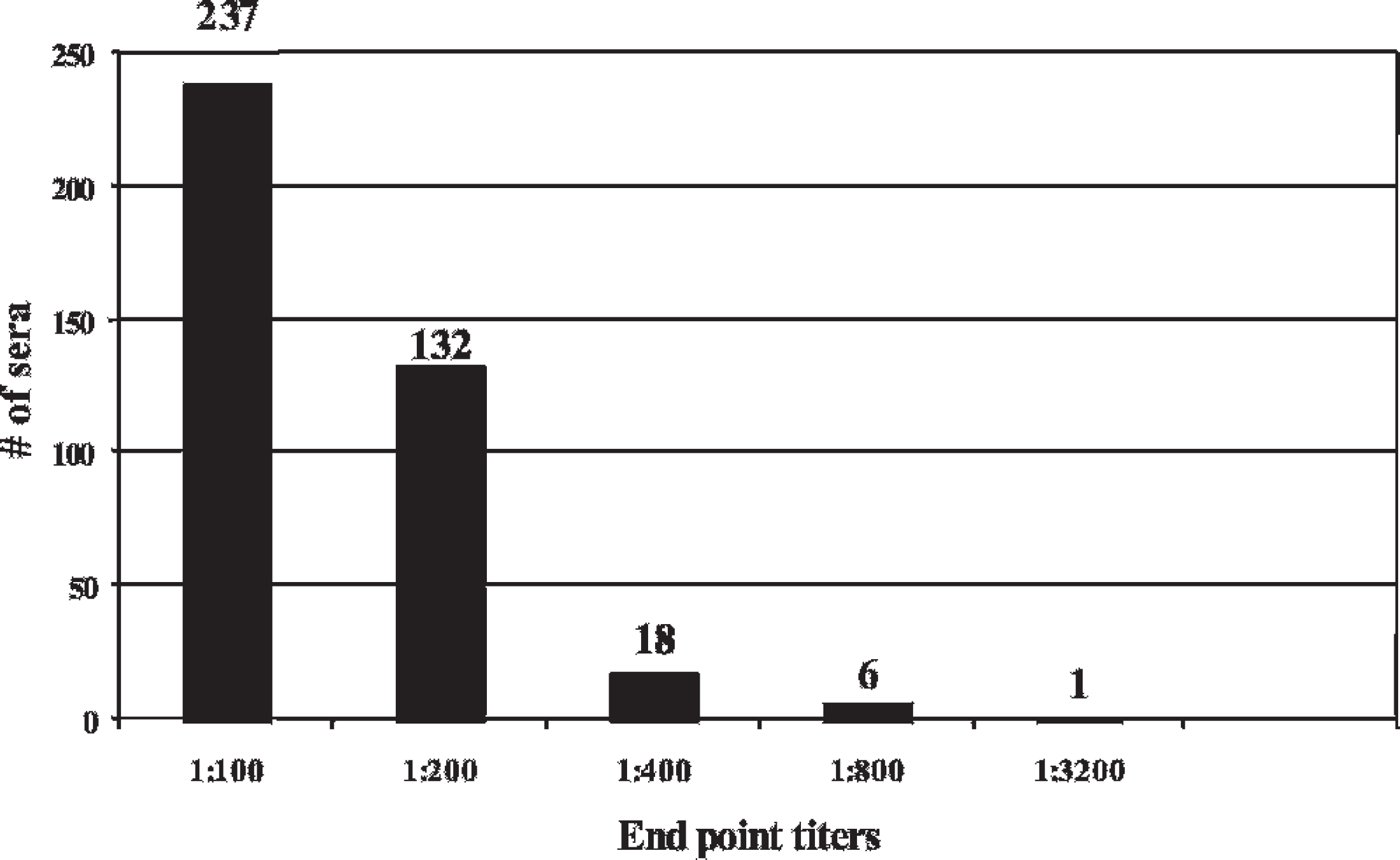
End point titers for 394 cattle sera that showed serological reactions to hardjoprajitno, but not hardjo-bovis A or hardjo-bovis B.
The results of this comparative serological data show that the HP reference antigen is more sensitive in detecting serovar hardjo antibodies in the MAT than either of the hardjo-bovis antigens used (HA S 1343 and HB S 818). Without adequate serological data, use of a hardjobovis strain in the MAT in the United States could be detrimental to the animal health industry. It was therefore concluded that the HP reference strain was the best of the three genotypes to be used in the MAT in the United States.
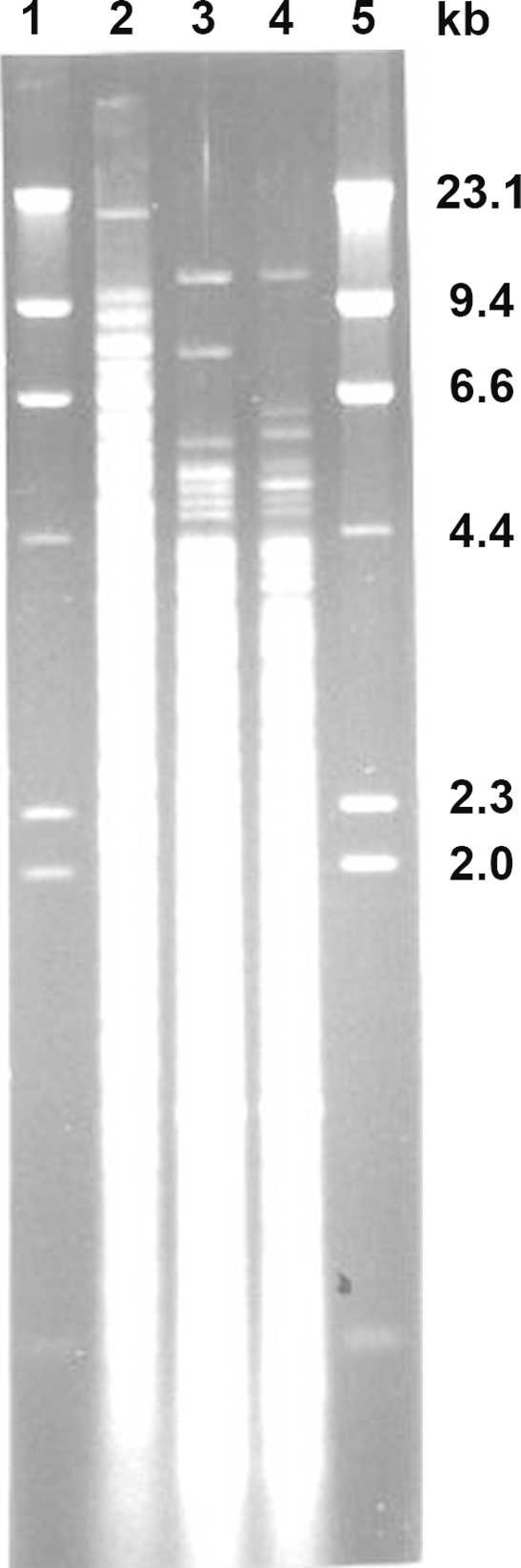
Electrophoretic patterns of chromosomal DNA of 3 serovar hardjo genotypes digested with Hha I. Lambda DNA digested with Hind III (lane 1 and 5), reference strain hardjoprajitno (lane 2), hardjo-bovis A (lane 3), and hardjo-bovis B (lane 4).
It cannot be determined from the data why the HP antigen was more reactive than either of the HA or HB strains used in this study. Surface proteins or lipopolysaccharides were not evaluated. However, comparative analysis by Western blot or similar structural analysis techniques may shed some light on this topic. The difference in serological reactivity may be caused by moderate genetic differences as suggested by different DNA fingerprint profiles (Fig. 4).
Hardjo-bovis strains other than HA S 1343 or HB S 818 could yield similar results to those of HP when used in the MAT. However, the hardjo-bovis strains used in this study did not detect hardjo antibody as well as HP. Similar conclusions were found in an international MAT proficiency test in regard to using a hardjo-bovis antigen in MAT testing (Roderick Chappel, unpublished data). However, it is uncertain as to how many strains and which genotype(s) were evaluated.
Other genotypes of serovar hardjo may be isolated in the future. Although two hardjo-bovis genotypes are recognized with Hha I REA, others have suggested that further discrimination of hardjo-bovis is possible with other molecular techniques. 11 In the future it may be necessary to re-evaluate whether the most optimal serovar hardjo strain is being used.
As previously mentioned, hardjo-bovis A and B are the only Hha I genotypes of the serovar hardjo to be isolated from cattle in the United States. 7,9 A decision to change the hardjo reference serovar used in the Leptospira MAT in the United States should be data driven. The issue of whether HP can detect hardjo-bovis infected animals cannot be addressed by the data from this study. However, a prevalence study 7 of mature cattle at slaughter in the United States published in 1991 recovered 88 Leptospira isolates from dairy and beef cattle. Of the 88 isolates 62 (70.5%) were identified as HA and 11 (12.5%) were identified as HB. Of the hardjo-bovis culture-positive cattle, only 1 did not react positively to HP antigen in the MAT at a titer of 100 or greater. The serovar hardjo isolates from animals in that study had titers that ranged from 100 to 12,800 against HP. One could conclude from that study as well as the comparative serology data presented here, that HP is the appropriate representative to be used in the MAT and can detect hardjo-bovis infected animals.
Researchers and diagnosticians from countries other than the United States have used a hardjo-bovis strain in their MAT testing. 3,4 The data from this United States study cannot be extrapolated to Leptospira MAT testing in other countries and vice versa. The question of whether to change a representative antigen used in the Leptospira MAT within a particular country is an important issue and should be given careful consideration.
Consideration should also be given to the production and impact of vaccine products that contain antigenically different strains relative to Leptospira diagnostic serology. In light of the fact that new products containing hardjo-bovis are being used in the large animal industry, a question could be raised as to how effectively these vaccinated animals are detected with HP. However, that question can only be answered in another study.
Footnotes
a.
Liquid polysorbate 80-bovine albumin medium, National Veterinary Services Laboratory, Ames, IA.
b.
DNAzol Reagent, Invitrogen Life Technologies, Carlsbad, CA.
