Abstract
Cutaneous leiomyosarcomas are malignant mesenchymal tumors of smooth muscle origin and are reported occasionally in avian species. A 14-y-old male laboratory White Carneau pigeon (Columba livia) was presented for surgical excision of a cervical soft tissue mass. Ultrasonography with color flow Doppler imaging revealed multiple cavitations of mixed echogenicity within the mass and vascularization. Histologically, the dermis and subcutis were expanded by a densely cellular multinodular mass comprised of fusiform cells forming haphazardly arranged broad streams and short interwoven bundles, often surrounding blood vessels and variably sized cavitations. Neoplastic cells were strongly immunopositive for desmin and α–smooth muscle actin, and negative for pancytokeratin, S100, and von Willebrand factor. Based on histopathology and IHC findings, the cutaneous mass was diagnosed as leiomyosarcoma (LMS). The pigeon died 312 d post-operatively. Postmortem examination revealed masses infiltrating the left and right pulmonary airways and one hepatic nodule, but no regrowth at the surgical site. Histologic and IHC evaluation of the pulmonary and hepatic masses were consistent with LMS, representing metastatic foci from the primary cutaneous LMS. Our case highlights the malignant behavior and histomorphologic features of cutaneous LMS in an avian species.
Leiomyosarcomas (LMSs) are non-encapsulated and frequently invasive mesenchymal neoplasms of smooth muscle cell origin. 12 LMSs are typically composed of intersecting bundles and fascicles containing well-differentiated neoplastic spindle-shaped cells with abundant eosinophilic cytoplasm and elongated, hyperchromatic nuclei.25,26 However, pleomorphic variants are also reported in humans and animals.1,5,12,26 LMSs often occur in middle-aged to elderly individuals and aged animals.13,18 Although common sites of primary LMS in domestic animals are the gastrointestinal and genitourinary tracts, other reported sites include the skin, subcutis, blood vessels, spleen, liver, retroperitoneum, oral cavity, brain, and heart.3,12,15,17,18 In birds, LMSs are often reported in the reproductive and gastrointestinal tracts of chickens, quail, and budgerigars.6,25 LMSs have been reported occasionally in the tracheal muscle of chickens, 21 and have been associated with avian leukosis virus subgroup J (ALV-J) infection in chickens. 21 Of 168 masses in budgerigars, 16 were LMSs, of which 15 were primary splenic tumors. 2 Cutaneous and subcutaneous LMSs have been described occasionally in birds.10,20,28,31 In dogs, cats, and ferrets, cutaneous LMSs have been observed on the head, neck, dorsal and lateral thorax, the thoracic and pelvic limbs, including the tarsus, metatarsus, elbow, hip, digit and footpad, and the perineum.12,17–19
LMSs are locally invasive, usually recur after incomplete excision, and infrequently metastasize. However, limited information is available in the literature regarding the clinical behavior of surgically excised LMSs in birds. We describe here the clinical and histomorphologic features of a cutaneous LMS with metastasis to the lung and liver in an adult White Carneau pigeon (Columba livia) used in IACUC-approved psychology studies. The pigeon was presented to the Division of Comparative Medicine, Massachusetts Institute of Technology (Cambridge, MA) for surgical excision of a cervical mass.
The 14-y-old male pigeon had no other history of illness and was single-housed in a research facility at Tufts University (Medford, MA) containing 73 other single-housed male pigeons. Immunization history consisted of a combined Newcastle disease and Salmonella vaccine administered subcutaneously in the dorsal or lateral cervical region at 2- and 6-mo-old. The pigeon had previously developed a 1.0-cm subcutaneous mass on the right lateral aspect of the mid-cervical region. The mass was removed by self-trauma before further testing or surgical intervention could occur but regrew 2 mo later.
On presentation for surgery, the pigeon was bright, alert, and responsive, with a body weight of 532 g and ideal body condition score (3 of 5). A 4.0 × 3.5 × 2.5 cm, semi-firm, multinodular, freely moveable mass was palpated on the right lateral aspect of the mid-cervical region, oral to the crop by ~2 cm, expanding the subcutaneous tissue. The mass did not impede the oral passage of a rubber catheter into the crop and was confirmed to not be continuous with the esophagus or crop. No other masses or abnormalities were found on physical examination. CBC and plasma biochemistry results, when compared to Columba livia reference intervals, were within normal limits. 32 Radiographs revealed a mid-cervical, round, soft tissue opacity that was continuous with the skin and connective tissue in the neck, with caudal displacement of the crop (Suppl. Fig. 1A, 1B). Metastases were absent on orthogonal whole-body radiographs, which aside from the cervical mass, were unremarkable. Ultrasonography showed intratumoral cavitations of mixed echogenicity. Vascularization supplying the mass was identified using color Doppler flow imaging (Suppl. Fig. 1C, 1D).
The cutaneous mass, which upon dissection extended into the deep skeletal muscle layer, was surgically excised under general isoflurane anesthesia, and the pigeon had a routine recovery. Adjunctive chemotherapy or radiotherapy was not performed. On cut section, the mass was composed of semi-firm, pale-tan nodules that were separated by cavitations filled with blood clots and white, friable, necrotic material (Suppl. Fig. 2A, 2B). The mass was fixed in 10% neutral-buffered formalin and processed routinely for hematoxylin and eosin staining. Histologically, the dermis and subcutis were expanded by a well-demarcated, non-encapsulated, densely cellular, multinodular mass, which was comprised of neoplastic cells forming haphazardly arranged broad streams and short interwoven bundles and was supported by a thin fibrovascular stroma (Fig. 1A). Neoplastic cells ranged from tightly packed to loosely arranged fusiform cells with indistinct cell borders, and a small-to-moderate amount of pale eosinophilic, finely fibrillar cytoplasm. Nuclei were centrally placed, round, oval, and irregularly elongate with blunt ends, with finely stippled to vesiculate chromatin, and 1–3 basophilic nucleoli. Neoplastic cells exhibited moderate-to-marked anisocytosis and anisokaryosis, and the mitotic count was 91 per 2.37 mm 2 (equivalent to 10 FN 22/40× fields; Fig. 1B). Karyomegalic neoplastic cells were noted occasionally. Neoplastic cells extended to deep and lateral surgical margins. Within the mass were several small-to-medium sized vascular sinuses and large cavitations, which were partially filled with erythrocytes and erythroid precursors mixed with proteinaceous eosinophilic fluid, necrotic cells, karyorrhectic debris, fibrin, and fewer foamy macrophages. Vascular sinuses were lined by a single layer of flattened endothelial cells and filled with mature erythrocytes intermixed with low numbers of erythroid precursors and rare lymphocytes. Scattered within the mass were low numbers of macrophages, some of which were hemosiderin-laden, and occasional lymphocytes and heterophils. Based on the histologic features of the cutaneous mass, our primary differential diagnoses included LMS, fibrosarcoma, hemangiosarcoma, peripheral nerve sheath tumor (PNST), and rhabdomyosarcoma.
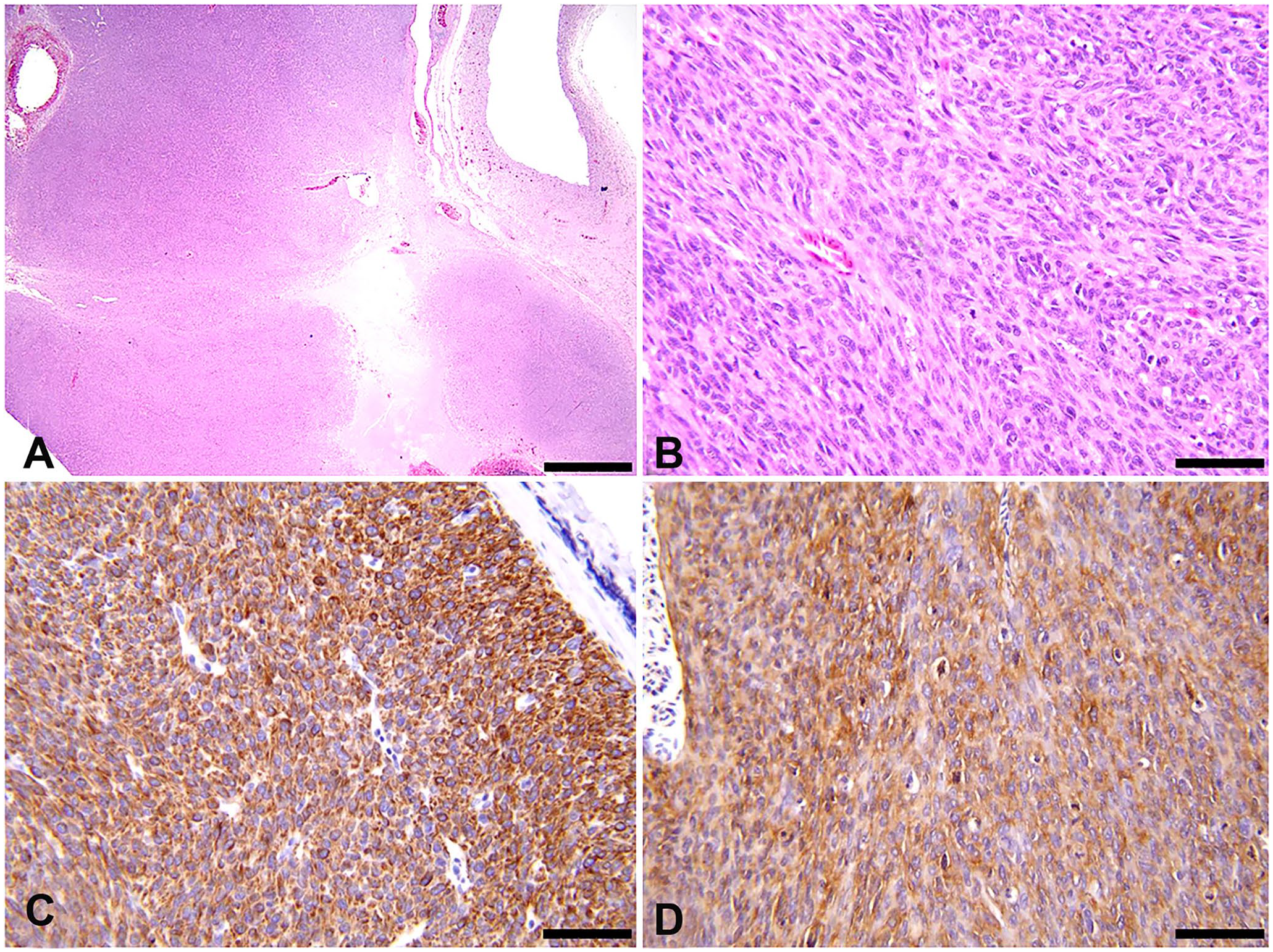
Cervical cutaneous leiomyosarcoma in a White Carneau pigeon.
To assess the cell of origin for this mesenchymal tumor, we performed a series of IHC tests on sections of the cutaneous mass. The following IHC markers were employed: alpha–smooth muscle actin (α-SMA; mouse monoclonal antibody, clone IA4, product MA5-11547; Thermo Fisher), desmin (mouse monoclonal antibody, clone D9, product MA1-0601; Thermo Fisher), S100 (mouse monoclonal antibody, clone 15E2E2, product CM128A; Biocare Medical), pancytokeratin (mouse monoclonal, AE1/AE3, product CM011B; Biocare Medical), and von Willebrand factor (VWF; rabbit polyclonal, product A0082; Agilent Technologies). Marmoset (Callithrix jacchus) tissues and normal pigeon skin were used as positive and internal controls for all IHC tests in our case. Histologic sections of the cutaneous mass were also stained with Masson trichome and phosphotungstic acid–hematoxylin (PTAH) to rule out fibrosarcomas and rhabdomyosarcomas, respectively.9,22 Neoplastic cells exhibited strong cytoplasmic staining for desmin, which was detected in 90–95% of the cutaneous mass (Fig. 1C); 60–70% of neoplastic cells were positive for α-SMA (Fig. 1D). All neoplastic cells were negative for S100, pancytokeratin, and VWF. The neoplasm was negative for collagen deposition on Masson trichome staining and did not exhibit skeletal muscle cross-striations on PTAH staining.
Based on the previous histologic and IHC findings of this cutaneous mass, our initial differential diagnosis of fibrosarcoma was ruled out, given that the tumor lacked deposition of intercellular mature collagen. The deposition of intercellular mature collagen is often present in canine fibrosarcomas. 12 Another differential diagnosis was hemangiosarcoma given the presence of vascular sinuses within the cutaneous mass. Hemangiosarcoma was ruled out because the tumor did not form interconnected vascular channels, and neoplastic cells were negative for VWF. Additionally, PNST was ruled out, given that neoplastic cells in the mass did not exhibit typical features such as Antoni type A or B patterns or nuclear palisading, and were negative for S100. Rhabdomyosarcoma was excluded because neoplastic cells were positive for α-SMA and lacked both skeletal muscle differentiation and cross-striations.9,12,18 IHC for myogenin was performed on the cutaneous mass and skeletal muscle from this pigeon; however, we were unable to validate the use of this marker on avian tissues. Based on these findings, our final diagnosis for the cutaneous mass was LMS.
The pigeon died 312 d after surgery. Although a very mild, gradual decrease in body condition was noted several months prior to death, no signs of deteriorating health were observed. While in an experimental testing chamber, the bird collapsed and succumbed to aspiration of regurgitated crop contents. At postmortem examination, the pigeon weighed 475 g, and had a prominent keel and pectoral muscle atrophy. The left lung (Suppl. Fig. 2C) was effaced by a 4.0 × 3.0 × 2.5 cm firm mass that on cut section contained large red–black blood clots and soft tan nodules. The right lung contained 2 small nodules (0.3-cm diameter). Between the right lung and the dorsal rib cage was a round, tan, soft mass (1.0-cm diameter). A firm, round, raised nodule (0.7 × 0.5 × 0.5 cm) was present at the margin of the right liver lobe (Suppl. Fig. 2D). The right testis (3.5 × 2.0 × 1.5 cm) was larger than the left testis (2.0 × 1.0 × 0.5 cm; interpreted as unilateral testicular hypertrophy). Both testes were oval, soft, and pale tan. There was no mass regrowth at the surgical site.
Histologically, the pulmonary and hepatic masses (Suppl. Fig. 3A, 3D, respectively) contained neoplastic cells similar to those described in the primary cutaneous tumor and were interpreted as metastatic LMS. Rare erythrophagocytosis was present in neoplastic cells in pulmonary masses. The right lung had a focal region of mild, parabronchial heterophilic and histiocytic inflammation with type II pneumocyte hyperplasia and edema, intra-airway hemorrhage, eosinophilic proteinaceous material, polymerized fibrin, and intralesional gram-negative rod-shaped bacteria and plant material. These changes were consistent with a previous episode of aspiration pneumonia. Histopathologic findings unrelated to LMS included mild regional heterophilic inflammation in the submucosa of the crop and minimal-to-mild seminiferous tubular degeneration in both testes.
The pulmonary and hepatic masses were also evaluated by IHC for α-SMA, desmin, S100, pancytokeratin, and VWF, and by PTAH and Masson trichrome staining. Similar to the cutaneous LMS, ~90% of the neoplastic cells in the pulmonary and hepatic masses displayed strong intracytoplasmic immunoreactivity for desmin (Suppl. Fig. 3B, 3E, respectively), and ~70% of the neoplastic cells exhibited moderate-to-strong intracytoplasmic immunoreactivity for α-SMA (Suppl. Fig. 3C, 3F, respectively). Neoplastic cells from pulmonary and hepatic masses were negative for S100, pancytokeratin, and VWF. Neoplastic cells from pulmonary and hepatic masses were also negative for collagen and skeletal muscle cross-striations on Masson trichome and PTAH staining, respectively.
Cutaneous LMSs in domestic animals are typically well-differentiated and composed of elongated, plump spindle cells arranged in bundles. 12 These tumors are usually encapsulated, variably circumscribed, and exhibit an expansive growth pattern, often invading the subcutaneous tissues. 12 Neoplastic cells contain prominent eosinophilic cytoplasm or variable cytoplasmic vacuolation, blunt-ended elongated nuclei with moderate pleomorphism, and a low-to-moderate mitotic index. 12 Salient histologic features of the cutaneous LMS in our case included an invasive growth pattern in which neoplastic cells infiltrated the deep muscular layer, moderate-to-marked atypia, and high mitotic activity. In dogs and cats, cutaneous LMSs may originate from arrector pili muscles (piloleiomyosarcomas) or from vessel walls (angioleiomyosarcomas).12,18 In avian dermis, smooth muscle is present in fascicles in the deep dermis and in the tunica media layer of cutaneous vessels, and also forms attachments to the myotendinous junction of feather follicles, functioning in both thermoregulation and flight.7,14 Based on the histologic appearance of normal skin at the periphery of the LMS in our case, we suspect that this neoplasm may have originated from either smooth muscle fascicles from the deep dermis or dermal vasculature. Neoplastic cells were closely associated with the tunica adventitia of dermal vasculature present at the periphery of the mass; however, there was no evidence of neoplastic cell invasion of the tunica media in these arteries, which is often the site of origin for cutaneous angioleiomyomas and angioleiomyosarcomas in dogs.12,18
Spontaneous LMSs in birds are usually extracutaneous and affect the reproductive and gastrointestinal tracts.6,25 In commercial chickens, LMS can be associated with ALV-J infection, and has been reported in layer flocks to manifest as tumors affecting various sites such as the leg, head, liver, heart, and lung, with concurrent myeloid leukosis and hemangiomas. 29 The LMS in our case appears to have arisen spontaneously, given that its clinicopathologic presentation was not consistent with lesions associated with ALV-J infection in chickens. The pigeon did not exhibit feather abnormalities, cutaneous hemorrhages in the phalanges and feather follicles, myeloblastosis, or other tumor phenotypes.11,16,21 Spontaneous LMSs have been described in the lung of an adult White Carneau pigeon. 22 LMS has also been described in association with the rib cage of a 3-y-old female budgerigar with metastasis to the bone marrow, spleen, and liver. 24 Other spontaneous mesenchymal neoplasms described in columbiform species include fibromas and fibrosarcomas in the skin and/or skeletal muscle, lipomas, liposarcomas, leiomyomas, rhabdomyomas, rhabdomyosarcomas, and cutaneous hamartomas.6,9,23,27
Cutaneous and subcutaneous LMSs have been reported occasionally in birds and have been associated with rapid and aggressive clinical progression, including metastasis to visceral organs. For example, a poorly differentiated myxoid LMS diagnosed in the subcutaneous tissue of the tarsometatarsus of a 12-y-old female sarus crane (Antigone antigone; syn. Grus antigone) failed to respond to surgery and medical therapy and metastasized to the liver and kidney. 10 Cutaneous LMSs have been documented in the coelomic region of a 6-y-old female budgerigar, on the neck of a 4-y-old female pigeon, and on the wing of a 4-y-old female fantail pigeon.20,28,31 Histologically, these tumors were composed of bundles of fusiform cells exhibiting specific immunoreactivity to α-SMA. They also exhibited large foci of cellular atypia and tumor-associated necrosis. Mitotic counts ranged from low to high numbers in neoplastic cells.10,20,28,31 The cutaneous LMS in our White Carneau pigeon exhibited clinical and histologic features similar to those of the cutaneous LMS that metastasized to the liver, spleen, pancreas, and kidneys in a fantail pigeon. 28
LMSs in humans occur in various anatomic sites, including the retroperitoneum, abdominal organs, superficial or deep soft tissues, skin, and large blood vessels.4,8,30 Histologically, these tumors are typically well-differentiated, but epithelioid and pleomorphic variants exist, the latter of which may range from overt pleomorphism to pleomorphic regions interspersed with well-differentiated areas.4,5,13 Cutaneous LMSs in humans are usually well-differentiated, although necrosis, sclerosis, hemorrhage, hyalinization, and myxoid changes may be noted. 30 IHC markers used for the diagnosis of LMS in humans include h-caldesmon, calponin, α-SMA, desmin, and transgelin.5,26 LMSs are distinguished from other soft tissue sarcomas by enrichment in genes linked to myogenic differentiation (e.g., muscle contraction and the actin cytoskeleton), amplification of myocardin (a transcriptional coactivator involving cardiac and smooth muscle differentiation), and losses or deletions in tumor suppressor genes including phosphatase and tensin homolog (PTEN), retinoblastoma 1 (RB1), and TP53.5,13,26 This genetic profile promotes LMS formation through cell-cycle deregulation.
Our case presented unique challenges based on the atypical histologic appearance of the cutaneous LMS. Neoplastic cells were pleomorphic, had high mitotic counts, and exhibited poor differentiation of smooth muscle cells throughout the entire cutaneous mass. Additionally, the presence of cavitations and variably sized vascular sinuses within the mass were unusual features that, to our knowledge, have not been reported in cutaneous LMSs in dogs and cats. 12 The histologic features of LMS along with the IHC characterization of the neoplastic cell population supported the definitive diagnosis of cutaneous LMS in our case. LMS should be considered as a differential diagnosis when evaluating cutaneous mesenchymal tumors in pigeons and other avian species.
Supplemental Material
sj-pdf-1-vdi-10.1177_1040638721992061 – Supplemental material for Cutaneous leiomyosarcoma with visceral metastases in a White Carneau pigeon and literature review
Supplemental material, sj-pdf-1-vdi-10.1177_1040638721992061 for Cutaneous leiomyosarcoma with visceral metastases in a White Carneau pigeon and literature review by Niora J. Fabian, Michael Y. Esmail, Lauren Richey, Sureshkumar Muthupalani, Jennifer L. Haupt, Joanna Joy and Sebastian E. Carrasco in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Dr. James G. Fox of the Division of Comparative Medicine at MIT for supporting the surgery and diagnostic workup at MIT and for reviewing our manuscript. We also thank Caroline Atkinson of the Division of Comparative Medicine, as well as Nathan Li and the Tufts Animal Histology Core, for processing samples essential to our case.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the National Institutes of Health grant P30-ES002109.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
