Abstract
We evaluated the extent of hepatic fibrosis in chronic liver disease of dogs using a modification of Ishak’s staging criteria for human chronic liver disease, and examined the association of stage of fibrosis with immunophenotypic markers of transdifferentiation of hepatic sinusoidal endothelial cells and hepatic stellate cells. Formalin-fixed, paraffin-embedded, hematoxylin and eosin–stained liver biopsy specimens from 45 case dogs with chronic liver disease and 55 healthy control dogs were scored for the presence and extent of fibrosis. This stage score for fibrosis strongly correlated with upregulated von Willebrand factor (vWF) expression in lobular sinusoidal endothelial cells (Spearman correlation coefficient [SCC] = 0.57, p < 0.05). Immunoreactivity for vWF factor was identified in 68.9% of case biopsies, varying in distribution from periportal to diffuse, whereas vWF immunoreactivity was identified in only 14.5% of control specimens, and was restricted to the immediate periportal sinusoids. The majority of both case and control biopsies exhibited similar prominent lobular perisinusoidal expression of alpha–smooth muscle actin (α-SMA). A minority of specimens (17.8% of case biopsies, 1.8% of control biopsies) exhibited low perisinoidal α-SMA expression, and there was a weak negative correlation between α-SMA expression and stage of fibrosis (SCC = −0.29, p = 0.0037). These results document a method for staging the severity of fibrosis in canine liver biopsies, and show a strong association between fibrosis and increased expression of vWF in hepatic sinusoidal endothelial cells.
Keywords
Introduction
Chronic liver disease is the result of ongoing injury to hepatocytes, wherein exposure to regenerative, inflammatory, and profibrotic mediators is prolonged. Fibrosis is a defining feature of chronic liver disease and is a common pathologic change associated with a wide variety of persistent insults. Hepatic fibrosis involves changes within the extracellular matrix (ECM) of the portal tracts and terminal veins, as well as alteration of the perisinusoidal ECM in the space of Disse, and may result in the development of portal hypertension, intrahepatic acquired shunting, and impairment of normal liver function and regeneration. 6
In human and rodent models of chronic liver disease, fibrosis within the space of Disse has been identified as an important feature in the progression and prognosis of chronic liver disease, and involves 2 processes. The first process is “capillarization” of the sinusoids, in which the normally attenuated endothelial basement membrane is thickened, with increased proportions of types I and III collagen, and lesser quantities of type IV collagen, 26 and the sinusoidal endothelium upregulates expression of von Willebrand factor (vWF; detectable immunohistochemically) 17 and loses normal fenestrae (detectable ultrastructurally). 23 This process has been described in studies of alcoholic liver disease 9 and other forms of chronic hepatitis 25 in humans, and in rodents exposed to carbon tetrachloride. 16 It is suspected to occur in dogs exposed to carbon tetrachloride 27 or with chronic bile duct ligation 5 based on light microscopic appearance, Masson trichrome staining, and increased quantities of ECM deposition seen with electron microscopy, but the occurrence and importance of the altered vWF endothelial cell phenotype in sinusoids of dogs with chronic liver disease has not been established.
The second process is the proliferation and transdifferentiation of hepatic stellate cells (HSCs) into cells with a myofibroblast-type phenotype, with contractile, profibrotic, and proinflammatory activities. 15 In humans and rodents, increased alpha–smooth muscle actin (α-SMA) expression is used as a phenotypic marker of activated HSCs and portal myofibroblasts in liver disease. 14 However, within the normal canine liver, α-SMA is constitutively expressed in quiescent perisinusoidal HSCs throughout the lobular parenchyma, as well as in myofibroblast-type cells and vascular smooth muscle within portal tracts. 10 Studies of the expression of α-SMA in dogs with liver disease have yielded contradictory results; a previous study 4 described normal to decreased α-SMA immunoreactivity in putative HSCs in lobular parenchyma and in myofibroblasts and vascular smooth muscle cells in portal and septal areas. However, in a 2007 study of 13 dogs with chronic liver disease and 6 healthy control dogs, 18 the intensity and staining area for α-SMA correlated with the intensity of fibrosis, whereas regenerative parenchymal nodules exhibited an overall decrease in immunoreactive α-SMA.
In humans, the extent and severity of fibrosis in chronic liver disease can be staged using established scoring systems, which have been used extensively in correlative studies investigating factors involved in the pathogenesis of chronic liver disease. The objective of our study is to adapt a histologic scoring system developed to stage fibrosis in biopsies from humans with chronic liver disease 11 to determine if alterations in the phenotype of hepatic sinusoidal endothelial cells and perisinusoidal stellate cells are associated with development of hepatic fibrosis in naturally occurring chronic liver disease of dogs.
Materials and methods
Case selection
Pathology reports of canine liver biopsies submitted to the diagnostic pathology services of the Department of Pathobiology and the Animal Health Laboratory, University of Guelph (Guelph, Ontario, Canada; 1998–2006) and Histovet Surgical Pathology (Guelph, Ontario, Canada; 2001–2006) were reviewed. A total of 45 cases were selected based on the following inclusion criteria: 1) a compatible morphologic diagnosis of chronic liver disease (chronic hepatitis, chronic-active hepatitis, hepatic fibrosis/nodular regeneration/cirrhosis, hepatic postnecrotic collapse and nodular regeneration, lobular dissecting hepatitis) and description including the presence of one or more compatible morphologic findings (hepatic fibrosis, parenchymal collapse, single-cell necrosis/apoptosis, piecemeal necrosis, mononuclear cell infiltrates, nodular regeneration, biliary proliferation); 2) adequate biopsy size (for the purposes of this study, only surgical incisional biopsies were included, with a minimum of 10 portal tracts); and 3) adequate tissue fixation (24–48 h in formalin). All cases included were surgical biopsies. Postmortem samples were excluded because of heterogeneity of tissue preservation and variation in length of formalin fixation. Criteria for exclusion from the study included the presence of other forms of liver disease (including acute hepatic zonal or massive necrosis typical of primary acute toxic or hypoxic liver injury, acute cholangiohepatitis, extrahepatic cholestasis, hepatic abscessation, chronic passive congestion and periacinar fibrosis secondary to cardiac failure, congenital vascular anomalies, and hepatic neoplasia); however, lesions with extensive neutrophil involvement were not excluded if they met inclusion criteria for chronicity without any indication of abscessation. Needle biopsy specimens were excluded because of size limitations (many were <2 cm in length, fragmented, and included fewer than 10 portal tracts, below the minimum guidelines suggested in human biopsy studies to avoid inaccuracies of assessment19,21,22). Case dogs had an average age of 7.6 years and a median age of 7.7 years. Purebred dogs of 22 different breeds made up 89% of case dogs, the remaining 11% were of mixed breed. Intact males (47%) and intact females (44%) were nearly equally represented, as were spayed females (4%) and neutered males (4%; Table 1).
Signalment statistics for case (n = 45) and control (n = 55) study animals.
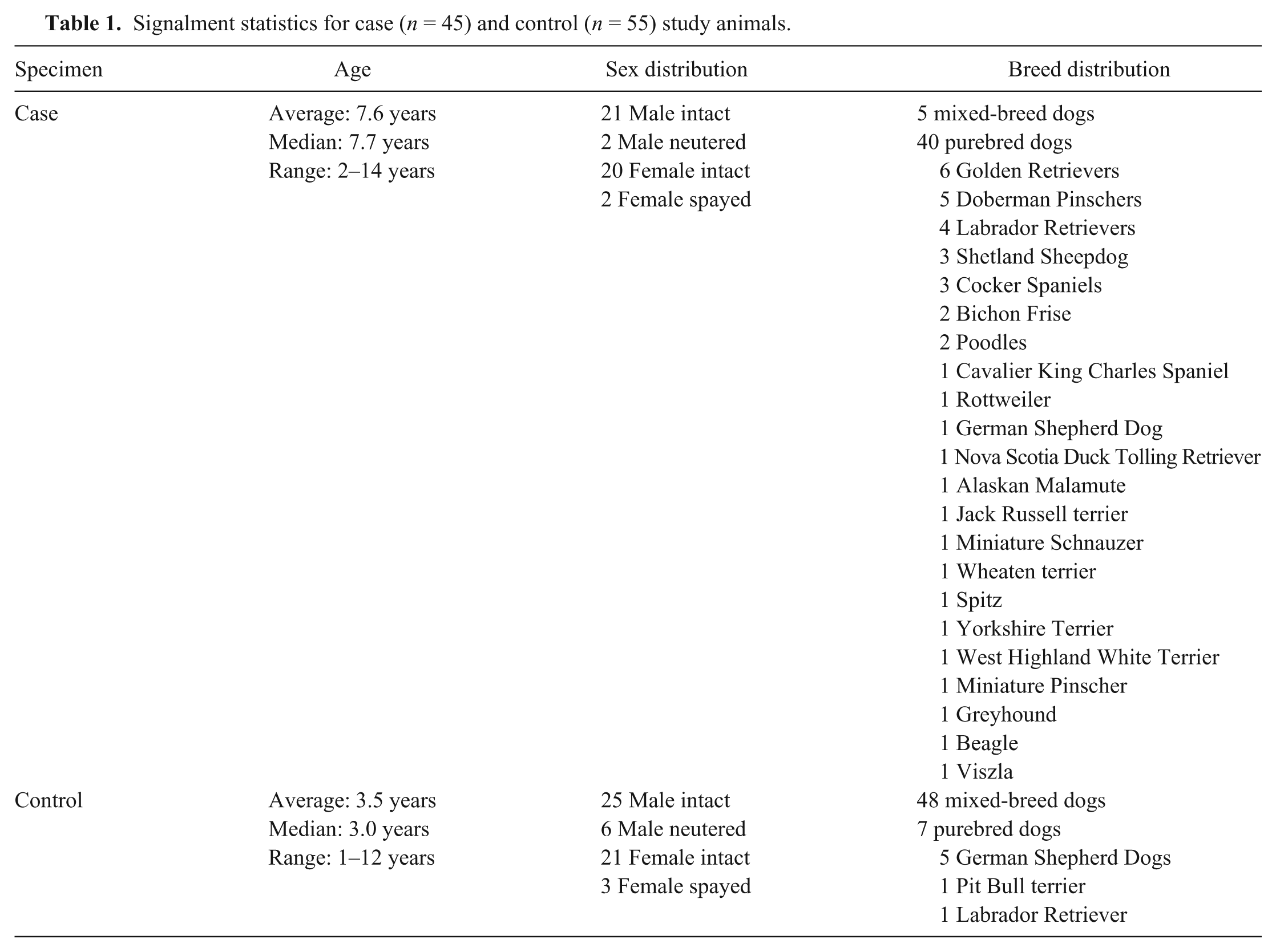
Control surgical biopsies were obtained from 55 clinically normal preconditioned dogs of various ages and breeds with normal hematology and serum biochemistry profiles (including serum alanine aminotransferase and gamma-glutamyl transferase) used for surgical teaching exercises approved by the University of Guelph Animal Care Committee and in accordance with the guidelines of the Canadian Council on Animal Care, at the Ontario Veterinary College, University of Guelph (Table 1). Intraoperative guillotine biopsies of the liver were collected, immediately fixed for 24 h in 10% neutral-buffered formalin, and routinely processed for histopathology. Control specimens were selected based on 1) absence of histologic evidence of significant primary or secondary liver disease in biopsy specimens; 2) adequate biopsy size (for the purposes of this study, only surgical incisional biopsies were included, with a minimum of 10 portal tracts); and 3) adequate tissue fixation. Control dogs had an average age of 3.5 years and a median age of 3.0 years. Purebred dogs of 3 different breeds made up 13% of control dogs, the remaining 87% being of mixed breed. Intact males (46%) and intact females (38%) again made up the majority, with fewer spayed females (6%) and neutered males (11%).
Case and control tissue samples were fixed in 10% neutral-buffered formalin for 24 h prior to routine processing and embedding in paraffin. Hematoxylin and eosin (H&E)-stained sections were prepared for the case (n = 45) and control (n = 55) biopsies using standard methods by the histotechnology laboratory of the Animal Health Laboratory, University of Guelph. Additional 6-μm serial tissue sections were mounted on charged glass slides for immunohistochemical staining.
Histologic stage scoring
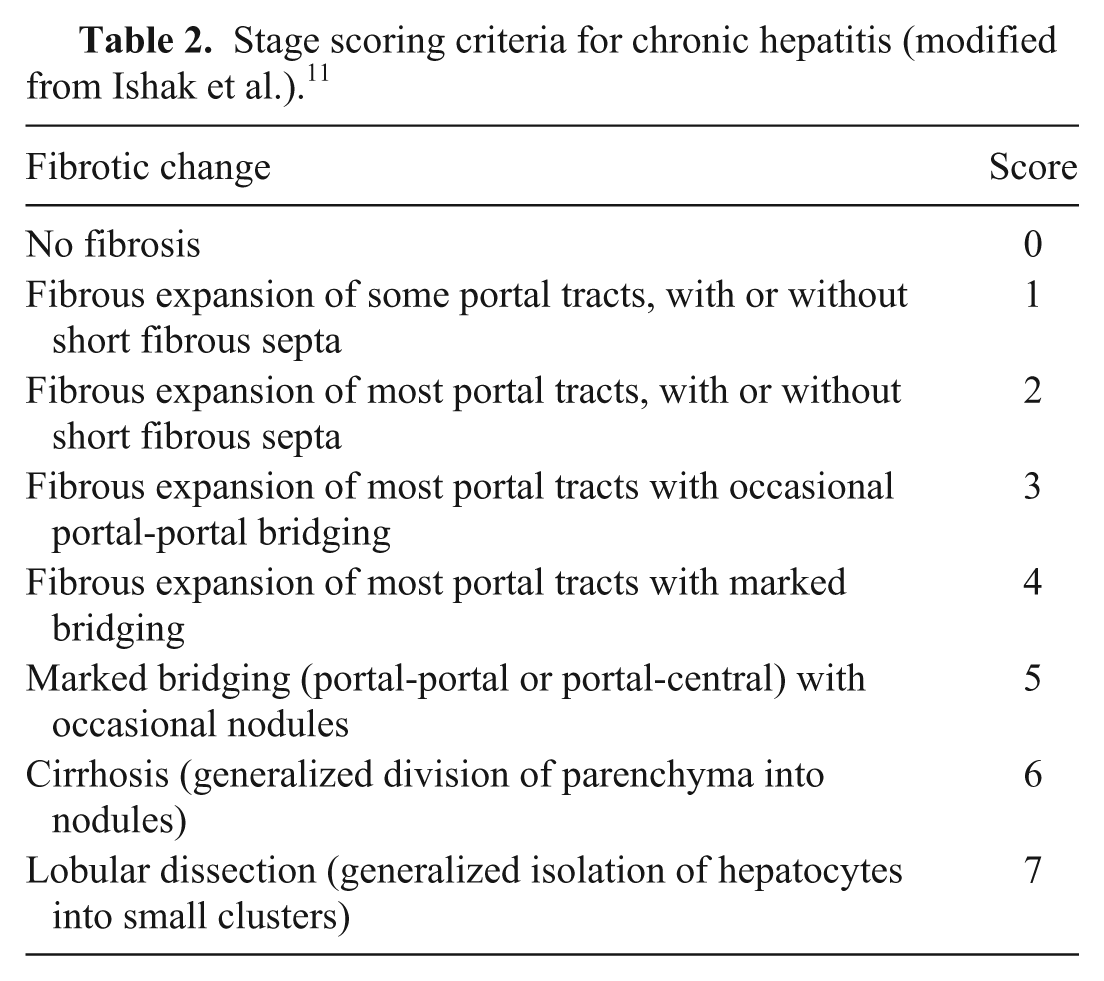
For the purpose of scoring the specimens, a modification of the scheme developed by Ishak et al. 11 was used (Table 2). This scheme was modified to include an eighth score representing lobular dissecting fibrosis, a pattern of fibrosis seen in dogs and generally regarded as having a profound negative prognosis. 24 H&E-stained histology slides from cases and controls were examined blindly and assigned individual semiquantitative scores for the presence and severity of fibrosis according to the thresholds described in the scheme. All biopsy sections were examined and scored no fewer than 3 times; twice by a single pathologist (AR Vince, for purposes of determining repeatability; repeat scores were performed within 2 weeks of initial scoring) and once by a second pathologist (MJ Stalker, for purposes of determining agreement). Prior to examination, both pathologists were given written definitions of the specific criteria evaluated and the thresholds for each score. In those circumstances in which scores for each criterion were identical between pathologists, these scores were used as a final score for purposes of comparison with other criteria. In those circumstances in which scoring varied between pathologists, the specimens were re-examined and assigned scores by consensus. For this cumulative score, interobserver agreement (κ = 0.89; 95% confidence interval [CI] = 0.82–0.95) and intraobserver repeatability (κ = 0.87; 95% CI = 0.82–0.91) were both assessed to be substantial.
Stage scoring criteria for chronic hepatitis (modified from Ishak et al.). 11
Histochemistry: collagen
Routine Masson trichrome staining for examination of collagen content and distribution was performed by the histotechnology laboratory of the Animal Health Laboratory, University of Guelph, using standard techniques. All slides were stained in a single batch with a laboratory-standard positive control slide. Masson trichrome–stained slides were scored using a similar staging scheme to that used for the H&E-stained slides (Table 2).
Immunohistochemistry
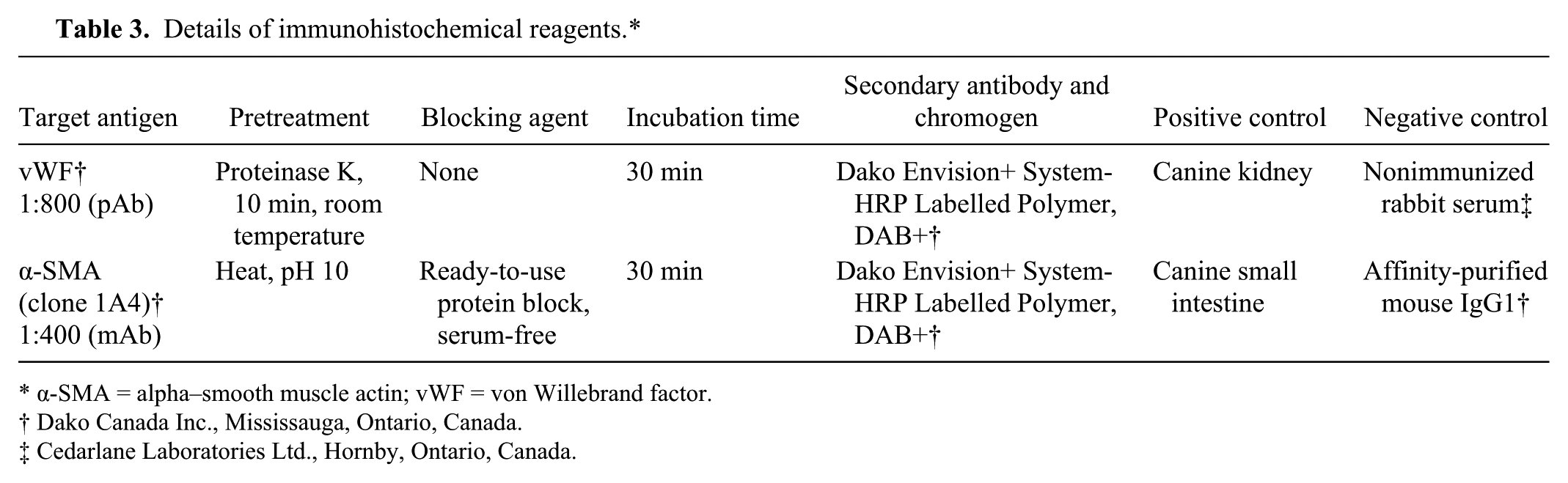
Six-micrometer serial unstained sections from all formalin-fixed, paraffin-embedded case and control livers were mounted on charged glass slides and prepared for immunohistochemical evaluation. Endogenous peroxidase activity was quenched by preincubation in 3% hydrogen peroxide. The details of each primary antibody used, antigen retrieval and protein block procedures, secondary antibodies, chromogens, and positive and negative controls are listed in Table 3. Slides were counterstained with Mayer hematoxylin. Additionally, comparable staining of vascular smooth muscle and endothelium within large hepatic vessels were used as internal controls within these specimens for α-SMA and vWF, respectively. vWF immunoreactivity was assessed according to broad patterns of expression; those with vWF staining limited to vessels in portal tracts and terminal hepatic veins were scored as 0, those expressing sinusoidal vWF staining in Rappaport zone 1 were scored as 1, and those expressing generalized sinusoidal vWF staining throughout the lobule were scored as 2. α-SMA immunoreactivity was evaluated for the extent of perisinusoidal staining present within lobules; those with staining throughout the lobules were allocated a score of 1 (high expression), whereas those with a lesser extent of expression were allocated a score of 0 (low expression).
Details of immunohistochemical reagents.*
α-SMA = alpha–smooth muscle actin; vWF = von Willebrand factor.
Dako Canada Inc., Mississauga, Ontario, Canada.
Cedarlane Laboratories Ltd., Hornby, Ontario, Canada.
Statistical analysis
All data collected from examination of slides was analyzed using commercial software. a Numerical scores for grading and staging were considered categorical data, requiring nonparametric analysis. Repeatability was calculated as a kappa value by standard methods. 13 Agreement was determined using a weighted kappa statistic.
Correlations were made between stage score and immunohistochemical and histochemical scores by calculating a Spearman correlation coefficient (SCC). In those tests where exact probability (p) values could not be determined, exact p-values were estimated using Monte Carlo modeling, extrapolating the dataset to one million samples in order to achieve the closest approximation; Monte Carlo p-values were calculated using the StatDistrib software package b using sums of squares, mean squares, and degrees of freedom as determined by software. a P-values <0.05 were considered significant. For each criterion examined, the distribution of data points relative to case or control status was examined using the Mann–Whitney–Wilcoxon (MWW) test. P-values <0.05 were considered to indicate a significant divergence from the null hypothesis (that the distribution of data points is random with respect to case and control specimen origin) with respect to a specific criterion. Graphical representations of data were generated using commercial software. c
Results
Forty-nine of the 55 liver biopsies from the control dogs were given a stage score of 0 (89%), 4 of 55 (7%) were scored as 1, and 1 of 55 (2%) were scored each as 2 and 3. Scores from case dogs ranged from 1 to 7, the maximum attainable in this scoring system (Fig. 1). No liver specimens from case dogs were given a stage score of 0, as expected from the selection criteria.
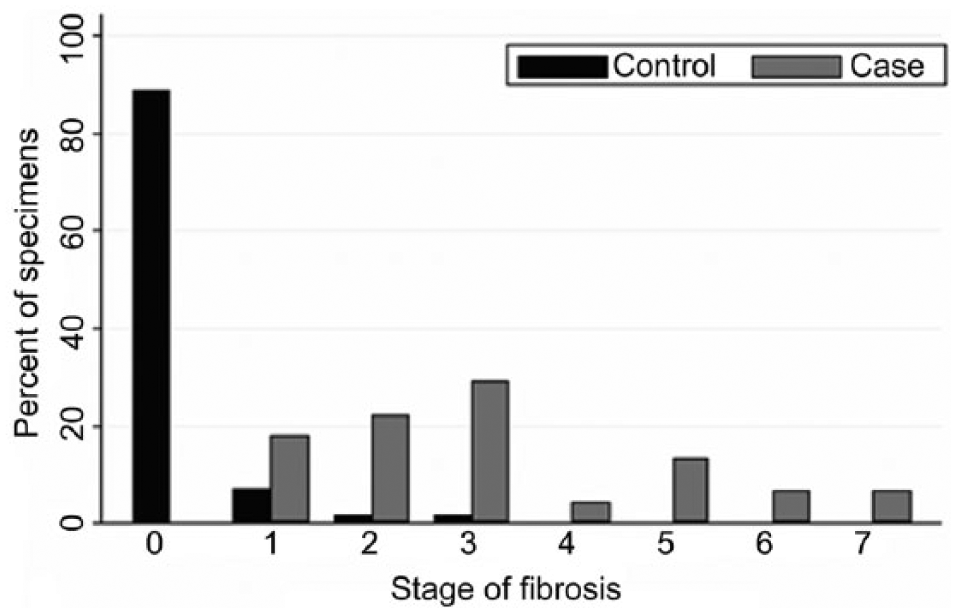
Scores for the stage of fibrosis present in case (n = 45) and control (n = 55) canine liver biopsy specimens.
Masson trichrome staining readily demonstrated collagen-rich connective tissue in liver specimens, and was particularly useful in those specimens with smaller quantities. Of the control biopsies, 42 (76%) were assigned a score of 0 for fibrosis using Masson trichrome stain; 7 (13%) were scored at 1, and 3 (6%) were scored at 2 and 3. Of the case biopsies, none were assigned a score of 0 or 1 for fibrosis using Masson trichrome stain; 6 (13%) were scored at 2, 11 (24%) at 3, 9 (20%) at both 4 and 5, 6 (13%) at 6, and 4 (9%) at 7. Within the patterns of fibrosis defined by the stage scores, other subtle patterns of fibrous connective tissue were noted in these liver biopsies: 5 (11%) case biopsies had a pattern of nodular parenchymal dissection limited to the immediate periportal parenchyma, and 2 (4%) case biopsies, and 1 (2%) control biopsy had a generalized increase in perisinusoidal collagen throughout hepatic lobules without disturbance of normal hepatic plate arrangement.
There was a significant difference in the distribution of Masson trichrome staging scores between case and control specimens, with higher staging scores for case specimens relative to controls (MWW p < 0.0001), and a strong significant positive correlation between the stage of fibrosis as determined by H&E staining and Masson trichrome staining (SCC = 0.90, p < 0.0001).
In all biopsy specimens, vWF immunohistochemistry delineated the vascular endothelium lining the vessels in portal tracts, terminal veins, and vascular structures in fibrous septa. There was a significant difference in the distribution of vWF scores between case and control specimens, with higher scores for vWF immunoreactivity in case specimens relative to controls (MWW p < 0.0001), and a strong significant positive correlation between the stage of fibrosis present and the score for vWF immunoreactivity (SCC = 0.57, p < 0.0001; Fig. 2). In control biopsies, vWF immunoreactivity was restricted to these large vascular structures in 47 of 55 (86%) samples, with extension of staining to sinusoidal endothelium in the immediate periportal region in the remaining 8 of 55 (14%) control biopsies (Fig. 3). In case biopsies, 15 of 45 (33%) had staining restricted to large vascular structures, 16 of 45 (36%) had immunoreactive vWF staining extending into the immediate periportal or periseptal region (score of 3), and 14 of 45 (31%) exhibited panlobular sinusoidal endothelial vWF immunoreactivity, a pattern that was restricted to case specimens (Figs. 4, 5). All case biopsies exhibiting generalized lobular fibrous dissection (stage 7, n = 3/45, 7%) exhibited a similar lobular dissecting pattern of expression for vWF (Fig. 6).
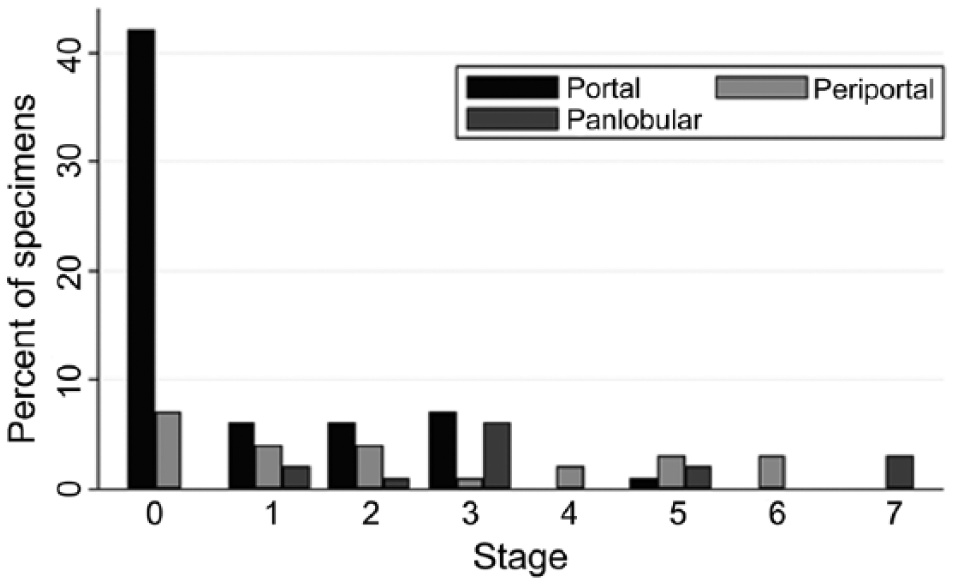
Scores for the extent of von Willebrand factor immunoreactivity (0–2) within the hepatic parenchyma in comparison with stage score (0–7) in case (n = 45) and control (n = 55) canine liver biopsy specimens.
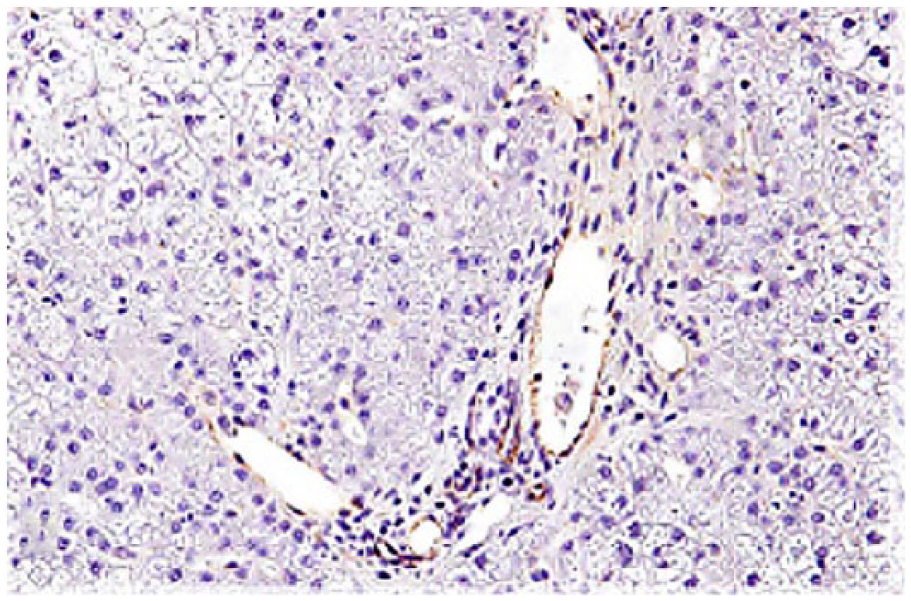
Liver of healthy control dog. von Willebrand factor immunoreactivity is restricted to the endothelium of portal veins, hepatic arteries, and terminal veins, and does not involve the endothelium of the sinusoids. von Willebrand factor immunohistochemical staining, hematoxylin counterstain.
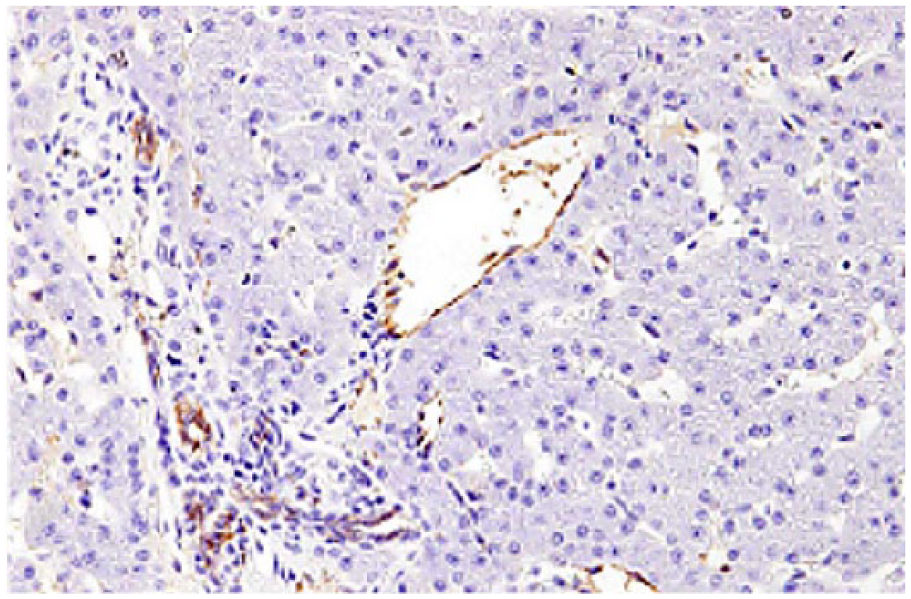
Liver of dog with chronic liver disease. von Willebrand factor immunoreactivity involves the endothelium of portal veins, hepatic arteries, and terminal veins, and extends to involve the periportal and periseptal hepatic sinusoidal endothelium. von Willebrand factor immunohistochemical staining, hematoxylin counterstain.
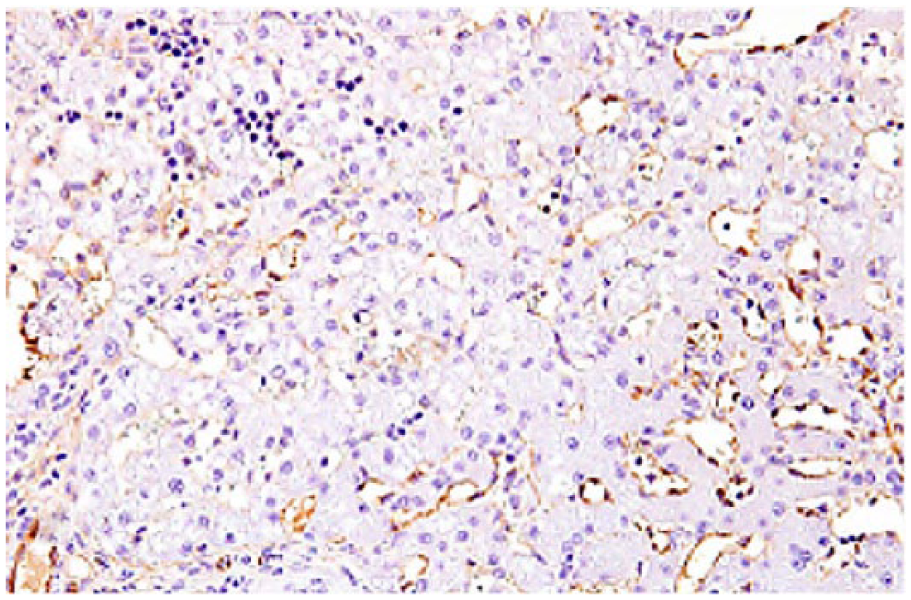
Liver of dog with chronic liver disease. Panlobular von Willebrand factor immunoreactivity within hepatic sinusoidal endothelium. von Willebrand factor immunohistochemical staining, hematoxylin counterstain.
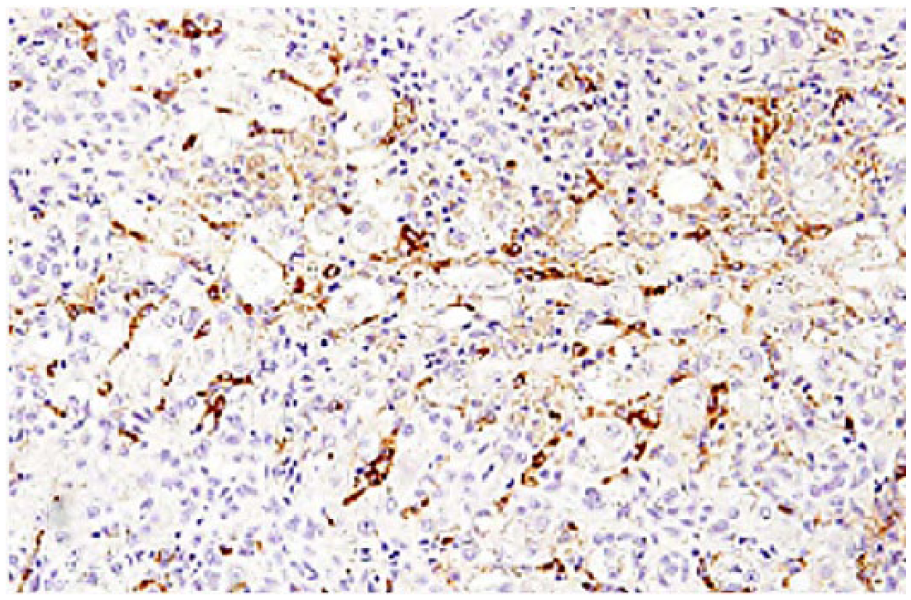
Liver of dog with lobular dissecting hepatitis. Dissection of hepatic parenchyma into small clusters of hepatocytes by von Willebrand factor immunoreactive cells and processes. von Willebrand factor immunohistochemical staining, hematoxylin counterstain.
In case and control liver biopsies, immunoreactive α-SMA was present in vascular smooth muscle and populations of stromal cells in the connective tissue surrounding portal triads, portal septa, and terminal hepatic veins. There was no statistically significant difference in distribution of α-SMA scores between cases and controls; however, there was a weak negative correlation between the stage of fibrosis present and the score for α-SMA immunoreactivity (SCC = −0.29, p = 0.0037; Fig. 7). In 54 of 55 (98%) control biopsies and in 37 of 45 (82%) case biopsies, there was also prominent lobular perisinusoidal expression of α-SMA (putatively staining perisinusoidal stellate cells and their cytoplasmic processes; Fig. 8). A minority of biopsies (1/55 control biopsies, 2%; 8/45 case biopsies, 18%) had low perisinusoidal α-SMA expression (Fig. 9). The 7 specimens with lobular dissecting hepatitis detected on H&E-stained slides also demonstrated a similar dissecting pattern of α-SMA, separating hepatocytes into small nests of cells (Fig. 10).
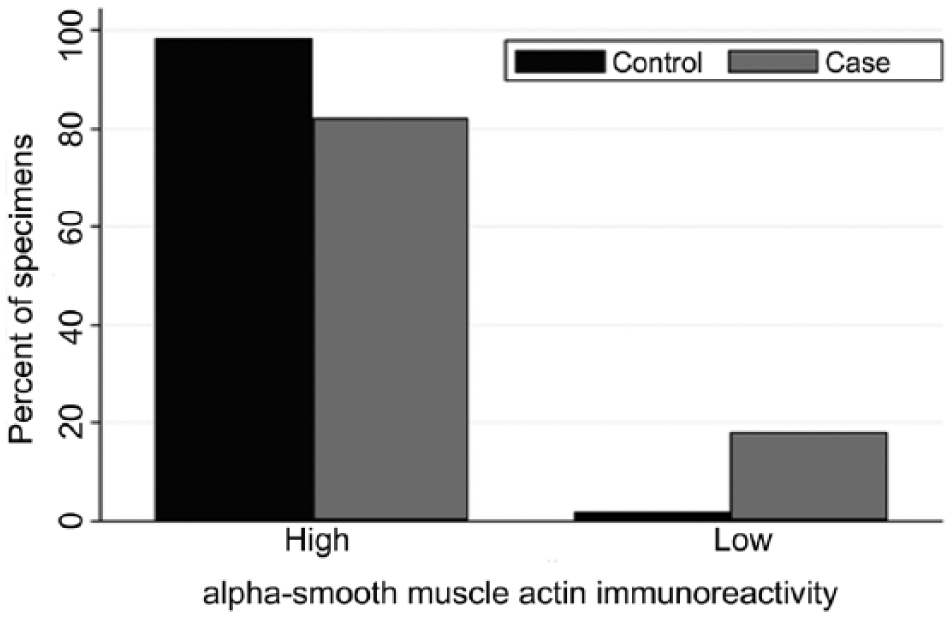
Scores for alpha–smooth muscle actin immunoreactivity (0–1) within the hepatic parenchyma in case (n = 45) and control (n = 55) canine liver biopsy specimens.
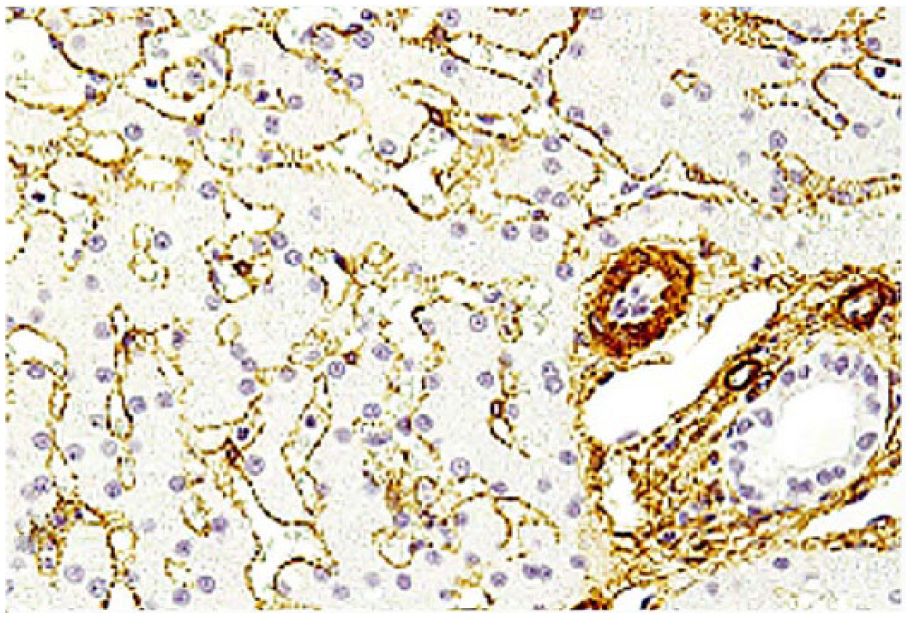
Liver of healthy control dog. Generalized and extensive perisinusoidal immunoreactivity for alpha–smooth muscle actin. Alpha–smooth muscle actin immunohistochemical staining, hematoxylin counterstain.
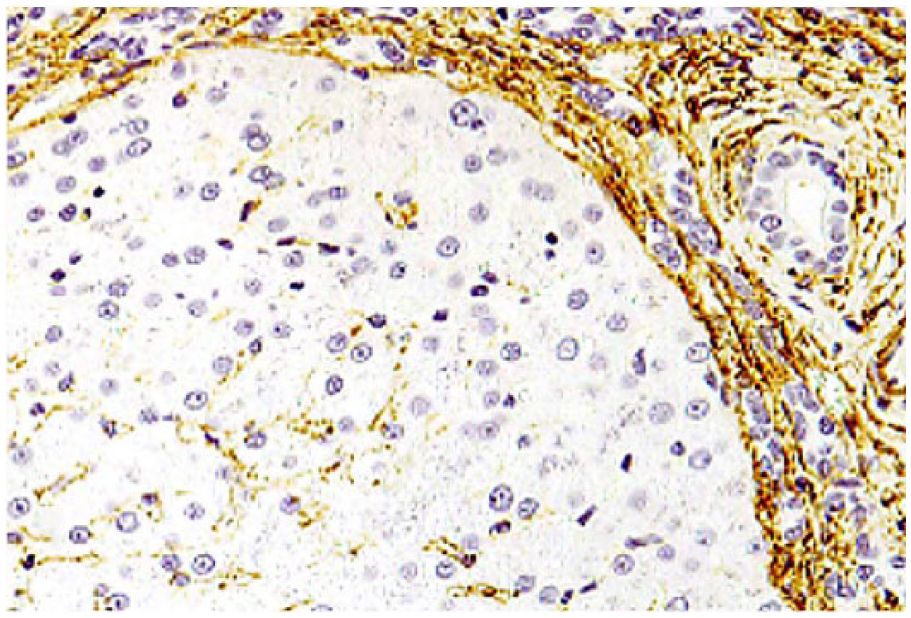
Liver of dog with chronic liver disease. Reduced perisinusoidal immunoreactivity for alpha–smooth muscle actin within an isolated nodule of hepatocytes. Alpha–smooth muscle actin immunohistochemical staining, hematoxylin counterstain.
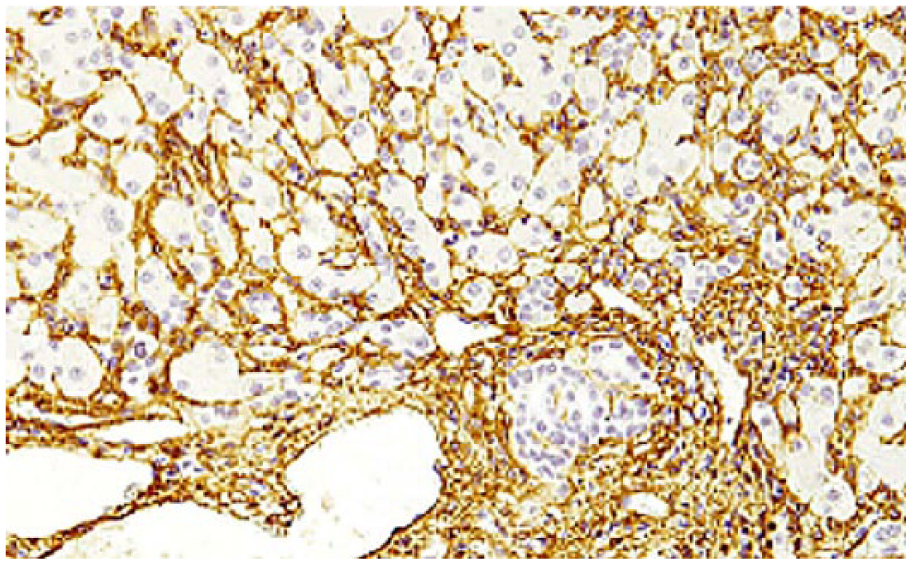
Liver of dog with lobular dissecting hepatitis. Dissection of hepatic parenchyma into small clusters by alpha–smooth muscle actin immunoreactive cells/processes. Alpha–smooth muscle actin immunohistochemical staining, hematoxylin counterstain.
Discussion
Interpretation of the presence, severity, distribution, and significance of fibrosis in canine chronic liver disease is inherently subjective, making prognostication of such disease difficult. Given the significance of fibrosis in chronic liver disease and the potential for heterogeneity in interpretation, systematic methods of interpretation using scoring schemes are used, with designated thresholds for each stratum allowing for more objective interpretation on either H&E or Masson trichrome stains. Based on agreement and repeatability statistics for these 100 specimens, this modification of Ishak’s scheme for staging fibrosis allows for the reasonably objective interpretation of fibrous connective tissue within canine liver specimens. Correlation statistics between stage of disease based on H&E-stained and Masson trichrome–stained slides further indicate that H&E stain is adequate for interpretation of fibrosis in chronic liver disease. However, Masson trichrome stain did frequently identify a subtly higher stage of fibrosis in these animals.
The process of capillarization of hepatic sinusoids involves increasing thickness, density, and altered collagen composition of the normally sparse sinusoidal basement membrane. 26 This is associated with changes in sinusoidal endothelial cell phenotype, with loss of fenestrae 23 and immunohistochemically demonstrable upregulated expression of vWF, a phenotype more typical of capillary-type endothelium elsewhere in the body. 17 This change is recognized in human liver disease, and in rodent models.9,16,17,25,26 These changes in sinusoidal endothelium impair protein and small molecule exchange between blood and hepatocytes. 1 Increased expression of vWF by sinusoidal endothelial cells throughout the hepatic lobule also occurs in canine chronic liver disease. This altered pattern of vWF expression is strongly correlated with the stage of disease present, suggesting a progressive shift in phenotype. The histologic appearance of vWF expression in the cases of lobular dissecting hepatitis is distinctive, forming an interlacing pattern of positive staining cells reiterating the dissecting pattern of fibrosis seen with H&E. This suggests that the lobular dissection may be more complicated than an atypical pattern of fibrosis, but may also include altered vascularization. Immunohistochemistry for vWF enhances our understanding of the mechanistic pathogenesis of chronic liver disease, and could prove useful for prognostication, although further prospective evaluation of this would be required to establish clinical utility.
Hepatocytes and endothelial cells produce relatively small amounts of ECM in comparison with hepatic myofibroblasts and HSCs. 1 HSCs comprise ~5–8% of the total number of cells in the normal human liver 3 and are situated beneath the sinusoidal endothelium throughout the lobular parenchyma. 7 Their long slender cytoplasmic extensions in the perisinusoidal space permit contact with large numbers of hepatocytes, endothelial cells, nerve endings, and other stellate cells. In their quiescent state, the major functions of HSCs are retinoid homeostasis, 2 and synthesis of ECM components, matrix metalloproteinases, and tissue inhibitors of metalloproteinases8,12; they may also be responsible for regulation of sinusoidal vascular resistance through contractile activity. 20 However, under pathologic conditions, HSCs can proliferate and redifferentiate into cells with a myofibroblast-type phenotype with contractile and proinflammatory activities. 15 Increased α-SMA expression is used as a phenotypic marker of activated HSCs and portal myofibroblasts in liver disease in humans and rodents. 14
Unlike the situation in human and rodent livers, α-SMA is constitutively expressed in quiescent perisinusoidal HSCs throughout the hepatic lobular parenchyma of normal healthy dogs, as well as in myofibroblast-type cells and vascular smooth muscle within portal tracts. Two previous studies of α-SMA expression in dogs with liver disease reported a negative correlation between the extent of lobular perisinusoidal α-SMA expression and severity of disease present, particularly the degree of necroinflammatory activity.4,10 However, a 2007 study 18 demonstrated a positive correlation in the intensity and staining area for α-SMA and the intensity of fibrosis present, with an overall decrease in expression within regenerative nodules.
In our study, α-SMA expression in the control dog biopsies was localized to cell bodies and slender processes adjacent to sinusoids (consistent with HSCs), as well as the smooth muscle surrounding portal, terminal, and septal vessels, and the majority of the case dog biopsies retained this staining pattern. Interestingly, specimens diagnosed with lobular dissecting hepatitis also displayed a similar lobular dissecting pattern of expression of α-SMA, in addition to vWF. This pattern of staining recapitulates the component staining of nonsinusoidal blood vessels, suggesting this pattern may be either an abnormal form of vascular remodeling or an abnormal angiogenic tissue response. There is a weak negative correlation between stage of fibrosis and α-SMA immunoreactivity; some but not all specimens with regenerative nodules demonstrated reduced α-SMA expression, and reduced expression was not restricted to those specimens with regenerative nodules. These findings suggest that α-SMA is not a reliable indicator of the state of HSC activation in canine liver. The alternative explanation, that activation of HSCs does not play a role in hepatic fibrogenesis in the diseased canine liver, is less likely; however, further investigation into the regulation and function of canine HSCs would be necessary to resolve this.
Footnotes
Authors’ contributions
AR Vince contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. MJ Stalker contributed to the acquisition and analysis of data. MA Hayes and MJ Stalker contributed to conception and design of the study. BJ Jefferson contributed to acquisition of data. All authors critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
SAS 9.1, SAS Institute Inc., Cary, NC.
b.
StatDistrib, William Sears, University of Guelph, Guelph, Ontario, Canada.
c.
Stata 9.1, StataCorp, College Station, TX.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by the Pet Trust of the Ontario Veterinary College, University of Guelph.
