Abstract
Visceral leiomyosarcoma is well described in dogs, but information about non-visceral locations and prevalence is lacking. The diagnosis of leiomyosarcoma is challenging without a gold standard, and often includes the use of immunohistochemical (IHC) stains. We used defined histopathologic patterns, histochemical staining, and IHC staining for smooth muscle actin (SMA), desmin, and laminin to characterize suspected non-visceral leiomyosarcoma in dogs at a single academic institution. In a retrospective search, we identified 24 dogs with a definitive or suspected histologic diagnosis of leiomyosarcoma in a non-visceral location. Histopathology results and clinical details were obtained. Biopsy sections were reviewed by a single pathologist using standardized histologic criteria, including light microscopic appearance, immunohistochemistry (more than two-thirds of neoplastic cells labeled with SMA and desmin or laminin), and histochemical staining (minimal-to-mild matrix deposition by Masson trichrome). Of the 24 cases of possible non-visceral leiomyosarcomas, 4 were consistent with a definitive diagnosis of non-visceral leiomyosarcoma (3) or leiomyoma (1) based on the established criteria. Only the leiomyoma had more than two-thirds of neoplastic cells label with all 3 markers; all 3 leiomyosarcomas had more than two-thirds of neoplastic cells label with SMA and laminin. Our data highlight the uncommon nature of non-visceral leiomyosarcoma and the importance of IHC for their diagnosis. A definitive diagnosis could not be made based on SMA alone, and desmin was not useful in this cohort. Further studies are needed to clarify the histopathologic, IHC, and clinical features of canine non-visceral SMA-positive mesenchymal tumors.
Leiomyosarcoma is a malignant mesenchymal tumor of smooth muscle origin that has been classified inconsistently within the soft tissue sarcoma (STS) group, with examples of inclusion16,34 and specific exclusion15,17 in the veterinary literature. In dogs, leiomyosarcoma occurs most often in visceral organs including the gastrointestinal (GI) tract, genital tract, urinary tract, liver, and spleen.13,25–27,37,46,48,52 Visceral leiomyosarcoma frequently requires immunohistochemical (IHC) staining for definitive diagnosis, and subsequently it is likely that an unknown subset of GI tumors in older studies represent alternative diagnoses such as gastrointestinal stromal tumor (GIST).4,11,19,23,32,40,45 The prevalence of visceral leiomyosarcomas has allowed prognostic studies in these organ systems.27,32,45,46 More atypical locations such as the oral cavity, skin, and large peripheral blood vessels5,7,31,42,44 have been reported sporadically. Little is known about whether there is a similar need for IHC stains for diagnosis of non-visceral leiomyosarcomas or their clinical behavior. In people, the biologic behavior and outcome of sarcomas in general 6 and leiomyosarcoma in particular8,20,33 is predicted in part by anatomic location, suggesting that anatomic location should be investigated for prognostic significance in the dog.
Leiomyosarcomas can present a diagnostic challenge given that their histopathologic characteristics may mirror other mesenchymal neoplasms, such as fibrosarcoma, myofibroblastic neoplasm, and GIST, 24 and no gold standard exists for diagnosis. Although descriptive features of leiomyosarcoma are reported (infiltrative, broad, and long fascicles of densely packed plump spindle cells with abundant eosinophilic cytoplasm, granular chromatin, and elongate or “cigar-shaped” nuclei 24 ), the level of differentiation and atypia may limit the utility of microscopic characteristics alone, which highlights the diagnostic challenge that leiomyosarcoma presents. Diagnosis is especially challenging in non-visceral locations, with many other differentials considered for sites such as the dermis and subcutis. In these instances, the need for IHC staining for confirmatory diagnosis is emphasized to rule-in or rule-out other more common tumor types.
Although no gold standard criteria exist for the diagnosis of leiomyosarcoma in dogs, various immunolabels have been utilized to aid in their identification. As a mesenchymal neoplasm, leiomyosarcoma largely immunolabels with vimentin,30,41,44,45 which lacks specificity for diagnosis given that most sarcomas are vimentin positive. Immunostaining for smooth muscle actin (SMA), a cytosolic intermediate filament, is often used clinically as a standalone IHC stain for leiomyosarcoma. Importantly, other tumors such as GIST (including extraintestinal GIST), myofibroblastic neoplasms, and perivascular wall tumors, as well as benign lesions such as nodular fasciitis and granulation tissue, can also immunolabel with SMA.3,4,9,10,29,41,44 Desmin, a cytoskeletal intermediate filament, has also been considered a specific marker that demonstrates muscle differentiation and hence is used to diagnose rhabdomyosarcomas and leiomyosarcomas.2,10,41 Laminin, a large extracellular matrix protein found in the basal lamina of many cell types including blood vessels, has also been identified as a useful marker for specific variants of smooth muscle tumors. 3 The use of laminin to aid in the diagnosis of leiomyosarcoma has only been described for variants of perivascular wall tumors.3,31 Stains for other IHC targets, such as calponin and heavy-caldesmon, both of which are constituents of smooth muscle thin filament and markers used in humans, are not routinely available in clinical veterinary practice. 24 Furthermore, although SMA and desmin are considered markers for leiomyosarcoma, the cutoffs or thresholds for percent positive cells for definitive diagnosis of a smooth muscle tumor are not established.
Evidence in both the human8,20 and veterinary18,27,31,46 literature demonstrates that leiomyosarcomas may exhibit different biologic behavior based on their anatomic location of origin. In dogs, for example, esophageal leiomyosarcomas are considered to be mostly low-grade and benign, 18 whereas splenic and hepatic leiomyosarcomas are considered largely malignant.27,46 Smooth muscle tumors of the skin, reported as angioleiomyosarcoma and piloleiomyosarcoma, appear to be as benign as the esophageal lesions, and are suspected to have a better prognosis given that resection is less complex than from the esophagus. 31 Hence, there is justification to characterize the behavior of leiomyosarcomas depending on location. Although visceral leiomyosarcomas have been relatively well-characterized in the veterinary literature, there is a paucity of descriptions of those occurring in non-visceral locations. Additionally, given the proclivity for other, more common tumor types to arise in non-visceral sites, as well as the lack of a gold standard for diagnosis, clearly defined histologic parameters and IHC staining are crucial in defining this population. Our aim, therefore, was to characterize suspected non-visceral leiomyosarcomas in dogs at a single academic institution utilizing a defined histopathologic pattern, histochemical staining, and IHC staining for SMA, desmin, and laminin.
Materials and methods
Case selection
The veterinary electronic medical records database at the University of California, Davis Veterinary Medical Teaching Hospital (UCD-VMTH) was searched (1987–2019) for canine cases with a diagnosis of suspected non-visceral leiomyosarcoma made by histopathology. Any suspected leiomyosarcoma was excluded if it arose from the abdominal or retroperitoneal cavity, esophagus, rectum, or internal genitalia. Cases were included when a definitive or suspected histologic diagnosis of leiomyosarcoma was made, which included those in which a smooth muscle origin was considered and IHC staining had not definitively confirmed another sarcoma subtype. Those cases without tissue available for evaluation and IHC assessment were excluded. Information captured from the medical record included signalment, description of the tumor, histopathologic characteristics, and any previous IHC stains used. Additional clinical details were captured, including (when available) clinical signs, results of initial laboratory tests, presence or absence of metastasis, treatment provided, surgical findings, and outcome information.
Histologic evaluation
Sections of formalin-fixed, paraffin-embedded (FFPE) biopsy specimens were evaluated by a single pathologist (E Choi) using standardized criteria as described. All slides from the identified cases were evaluated. Prior to the use of IHC, tumors were evaluated microscopically for characteristics deemed to be consistent with a possible smooth muscle tumor, including spindle cells forming variably dense streams, bundles, and/or fascicles. Tumors forming whorls were not considered compatible with smooth muscle tumors and were excluded from further characterization. For the tumors with consistent cellular morphology, a representative slide was chosen to perform the IHC staining.
A FFPE tumor specimen was cut into 4-μm-thick sections for IHC staining (Table 1). Antigen retrieval was performed, and sections were blocked in 10% normal horse serum in PBS-Tween 20 (0.02%) for 20 min. The primary antibody was applied without rinsing and incubated for 1 h. All post–antigen-retrieval incubations were in a humidity chamber at room temperature. The secondary polymer was applied for 30 min to label. Labels were visualized with NovaRED for peroxidase (SK-4800; Vector), per the manufacturer’s instructions. Sections were counterstained with hematoxylin 1 (Fisher) for 10 s and bluing reagent (Fisher) for 2 min, air dried, and coverslipped. Sections of canine small intestine, skeletal muscle, and skin stained for SMA, desmin, and laminin, respectively, were used as positive controls. Blood vessels served as the internal positive control. For negative controls, primary antibodies were replaced with homologous nonimmune sera. The samples were processed in batches for each antibody, and each batch had a positive and negative control.
Details of immunohistochemical stains performed on sections of suspected non-visceral leiomyosarcoma in dogs.

SMA = smooth muscle actin.
Representative slides of each IHC stain were scored using a semiquantitative method by a single pathologist (E Choi) based on percent immunolabeled neoplastic cells (Table 2). For tumors with a SMA immunolabeling score of 3, Masson trichrome (MT) staining was performed to assess the degree of extracellular matrix (collagen) deposition between neoplastic cells. The amount of collagen was given a score of 0–3: 0 = minimal, 1 = mild, 2 = moderate, and 3 = abundant material between neoplastic cells.
Scoring system used to evaluate suspect leiomyosarcomas in dogs.

Samples that were morphologically compatible were determined to be consistent with leiomyosarcoma or leiomyoma if both SMA and desmin or both SMA and laminin scores were 3, with a minimal-to-mild amount of collagen between neoplastic cells (MT score of 0 or 1), unless otherwise defined. Good demarcation, lack of invasion, and lack of mitotic figures supported the diagnosis of a leiomyoma over a leiomyosarcoma.
Results
Suspected non-visceral leiomyosarcomas were identified from 42 dogs according to the search criteria (Fig. 1); 7 lacked available tissue samples for further evaluation, and 11 were excluded because of histologic whorls. The 24 remaining cases fulfilled the defined morphology and were included in our study. The masses were from a variety of non-visceral locations including the subcutis (n = 15), dermis (n = 3), footpad (n = 1), orbit (retrobulbar n = 1, conjunctiva n = 1, eyelid n = 1), pharynx (n = 1), and intradural cervical region (n = 1; Table 3, Suppl. Table 1).
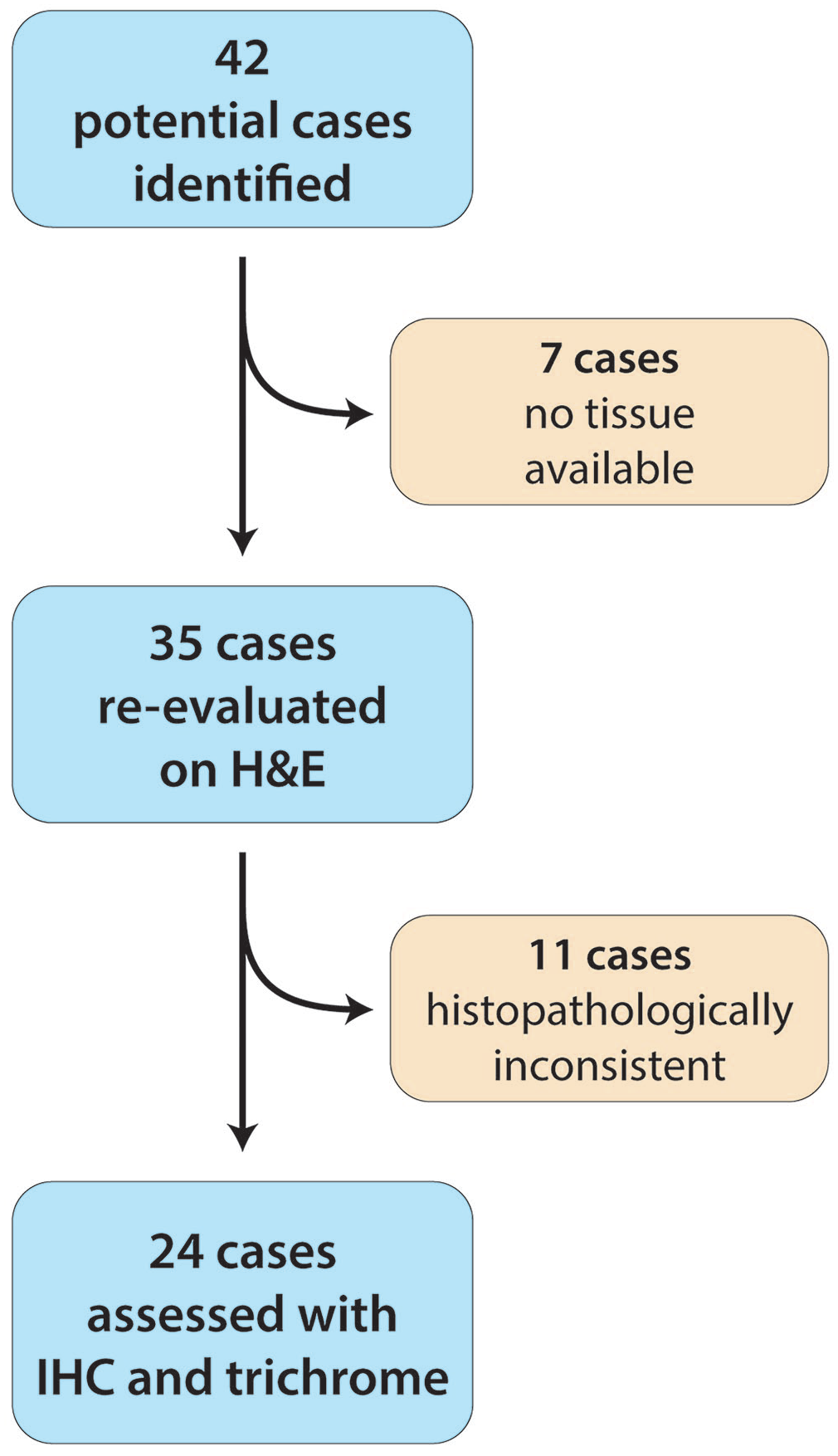
Flowchart of biopsy samples of suspected non-visceral leiomyosarcomas in dogs.
Clinical information and staining results for suspected non-visceral leiomyosarcomas in dogs.
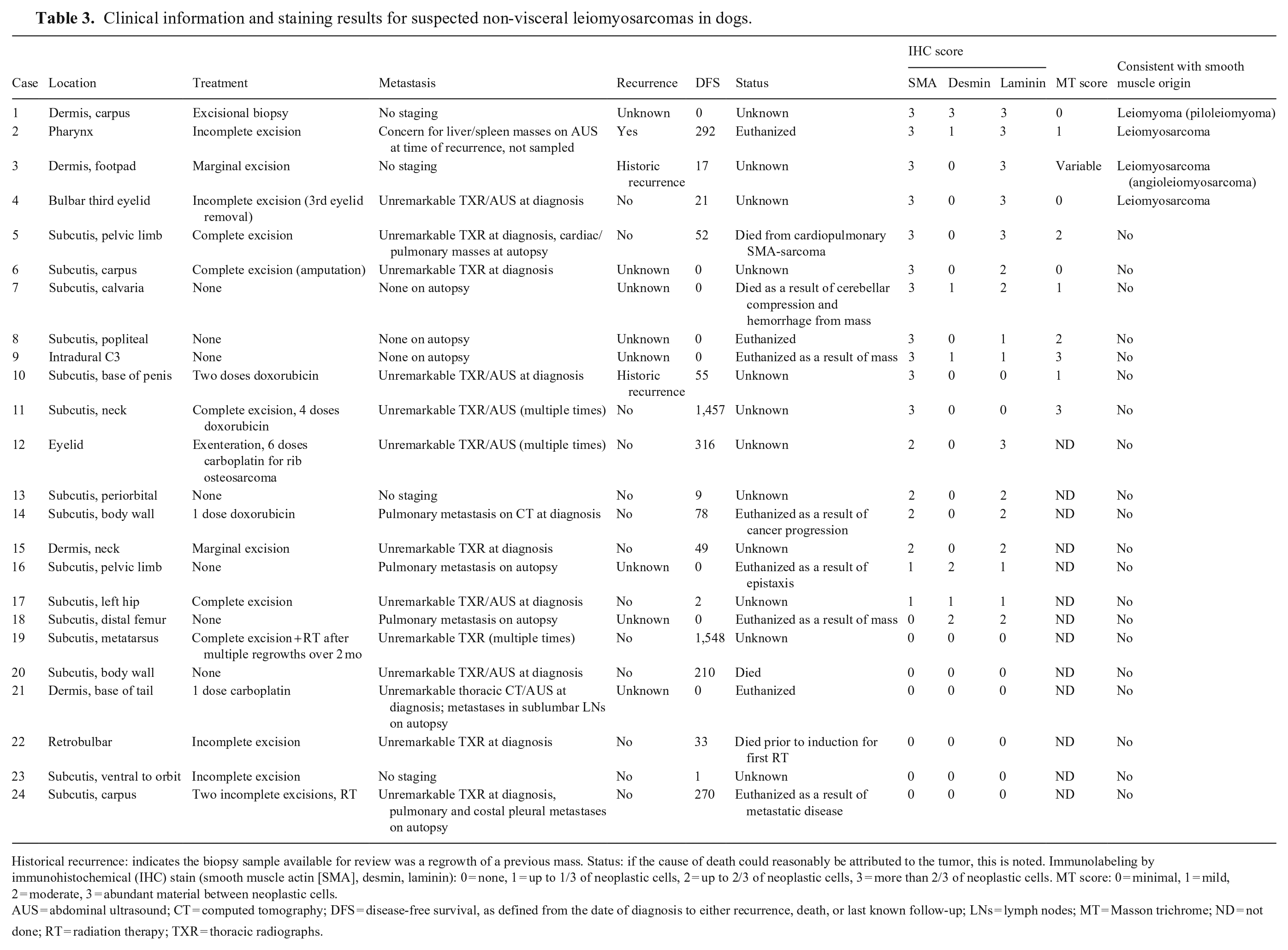
Historical recurrence: indicates the biopsy sample available for review was a regrowth of a previous mass. Status: if the cause of death could reasonably be attributed to the tumor, this is noted. Immunolabeling by immunohistochemical (IHC) stain (smooth muscle actin [SMA], desmin, laminin): 0 = none, 1 = up to 1/3 of neoplastic cells, 2 = up to 2/3 of neoplastic cells, 3 = more than 2/3 of neoplastic cells. MT score: 0 = minimal, 1 = mild, 2 = moderate, 3 = abundant material between neoplastic cells.
AUS = abdominal ultrasound; CT = computed tomography; DFS = disease-free survival, as defined from the date of diagnosis to either recurrence, death, or last known follow-up; LNs = lymph nodes; MT = Masson trichrome; ND = not done; RT = radiation therapy; TXR = thoracic radiographs.
SMA, desmin, and laminin immunolabeling were identified in 16, 7, and 15 cases, respectively (Fig. 2). Cases 1–18 had immunolabeling with >2 markers; 6 cases (19–24; Table 3) were negative for all 3 markers.
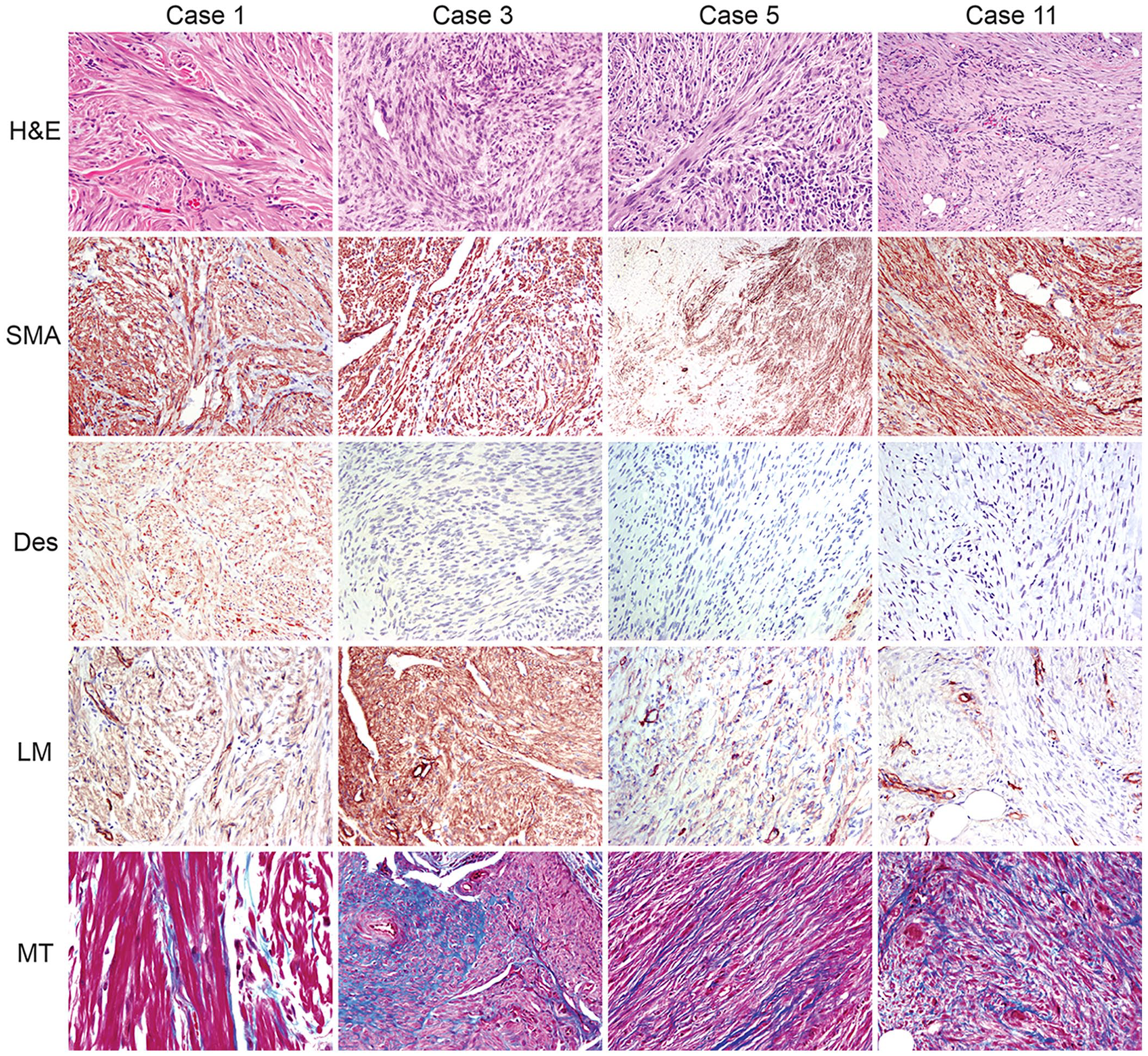
Representative images of masses considered smooth muscle tumors and other tumors.
Ultimately, 4 of 24 tumors (cases 1–4) were consistent with leiomyoma or leiomyosarcoma based on the diagnostic criterion of more than two-thirds of neoplastic cells labeled with SMA and laminin (score of 3) and low MT staining (score of 0 or 1). Two were from the dermis of the distal forelimb (carpus and metacarpal footpad), one was from the pharynx, and one from the third eyelid. The 2 dermal masses had features suggestive of anatomic origin. Case 1, the carpal mass, strongly resembled erector pili muscle and had histologic features supportive of a benign neoplasm suggestive of a piloleiomyoma. Case 3, the metacarpal footpad mass, was closely associated with numerous vascular walls and had features of malignancy supporting the diagnosis of an angioleiomyosarcoma (Fig. 2). Interestingly, this mass had variable MT staining ranging from minimal between tightly packed neoplastic cells to abundant perivascular collagen deposition. The pharyngeal (case 2) and third eyelid masses (case 4) were considered to have originated from resident smooth muscle bundles in those locations.
Seven cases (cases 5–11) received an SMA score of 3 but did not fall within our diagnostic criteria. Case 5, a subcutaneous and intramuscular mass of the thigh, scored 3 for SMA and laminin but received an MT score of 2 (Fig. 2). Additionally, 4 cases scored 3 for SMA but received a score of 2 (cases 6, 7) or a score of 1 (cases 8, 9) for laminin. Two cases (cases 10, 11) scored 3 for SMA but did not stain with laminin (Fig. 2). Of these, only 2 cases received a score of 1 for desmin (cases 7, 9). The MT score was variable in these cases and, as expected, was not correlated with the laminin score.
In 4 cases (cases 12–15), SMA labeled less than two-thirds of the neoplastic population (score of 2). In these 4 cases, 1 case scored 3 for laminin (case 12) and 3 cases scored 2 (cases 13–15). None of these cases stained with desmin. In the eyelid mass from case 12, SMA only stained neoplastic cells locked in dense fascicles, which accounted for 40% of the entire population (score of 2) and spared broad streams and bundles, supporting heterogeneity of the neoplastic cells. Interestingly, this dog developed a newly identified rib osteosarcoma within a year of the eyelid mass diagnosis, which showed similar immunolabeling of neoplastic cells but a different histologic pattern.
Two cases (cases 16, 17) were considered to have a SMA score of 1, a laminin score of 1, and a desmin score of 1 or 2. Both were subcutaneous masses, one from the pelvic limb, and the other from the left hip. One case (case 18) was negative for SMA but scored 2 for desmin and laminin. Six cases (cases 19–24) were diffusely negative (score of 0) for SMA, desmin, and laminin.
Discussion
Leiomyosarcoma, although uncommon in non-visceral organ systems, is a diagnosis that is routinely considered by the pathologist when evaluating a sarcoma because of its histologic similarities to other sarcomas, such as fibrosarcoma, peripheral nerve sheath tumors, perivascular wall tumors, and myofibroblastic tumors. However, diagnostic criteria for subtypes of sarcomas, including leiomyosarcoma, are not defined clearly. We attempted here to utilize a combination of stringently defined histopathologic and IHC characteristics to identify non-visceral leiomyosarcomas. Our data highlight the diagnostic challenge that non-visceral smooth muscle tumors present, the importance of IHC staining, and the questions that remain about the most accurate approach given the lack of a gold standard for definitive diagnosis. In our sample set of 31 suspected leiomyosarcomas with available tissue samples, 11 were excluded based on their cellular morphologic characteristics, and 11 of the 24 remaining cases were diffusely strongly positive for SMA, the IHC stain utilized most commonly for the diagnosis of smooth muscle tumors. With the diagnosis of smooth muscle origin defined in our study as positive staining with both SMA and desmin or SMA and laminin, and minimal-to-low trichrome-positive material deposition between neoplastic cells, only 4 cases fulfilled our criteria. The only case that scored 3 for all 3 IHC stains was diagnosed as a leiomyoma, a benign smooth muscle tumor likely originating from the erector pili muscle. Only 5 other tumors had joint SMA, desmin, and laminin labeling and, of these, one was considered a leiomyosarcoma. The others, given their lesser percentage of immunolabeled cells or abundant collagen deposition, did not meet our diagnostic criteria for leiomyoma or leiomyosarcoma. This finding may imply that only 2 of 24 potential smooth muscle tumors are indeed smooth muscle origin, but alternatively, it is possible that 1) laminin and especially desmin are not reliable markers to aid in the diagnosis of leiomyosarcoma in dogs, or 2) our diagnostic criteria were too stringent. Interestingly, human leiomyosarcomas may have loss of desmin immunolabeling, which is thought to be associated with loss of myogenic differentiation. 14 It is possible that neoplastic cells in these tumors may lose desmin expression, and one could theorize that this reduced expression may occur in dogs.
Laminin was chosen as the third IHC stain to compare the staining pattern in conjunction with SMA and desmin. Laminin is a major basement membrane protein and is therefore present in many different tissue and cell types, including epithelial and mesenchymal cells. Although laminin is not a specific marker for smooth muscle differentiation, the amount of laminin is suggested to decrease with myofibroblastic differentiation, and laminin is not expected to be expressed in human low-grade myofibroblastic sarcomas. 35 Indeed, in our sample set, a subset of tumors strongly expressed SMA whereas none to less than two-thirds of neoplastic cells expressed laminin. This may suggest that a subset of these tumors should be better classified as myofibroblastic tumors, and their behavior should be studied separately. In humans, myofibroblastic cells can form a variety of benign and malignant tumors, including inflammatory myofibroblastic tumor, nodular fasciitis, and myofibroblastic sarcoma.
Inflammatory myofibroblastic tumors are characterized by spindle cell proliferation with a variable inflammatory component and occur mostly in the abdominal cavity of children. 12 A subset of inflammatory myofibroblastic tumors has ALK gene arrangement. 49 Given histologic similarities, inflammatory myofibroblastic tumors have been reported in dogs,29,47 although ALK gene arrangement has not been investigated, to our knowledge. Our subset had 2 cases (case 8, 10) that could fit this diagnosis, although the diagnostic criteria are not well established in veterinary medicine.
Nodular fasciitis is a benign mass with fusion genes involving USP6 in people. 39 Nodular fasciitis has also been recognized in the dog,9,22 but gene fusion has not been investigated, to our knowledge. Histologically, nodular fasciitis is considered when the proliferating cells are loosely formed with prominent reactive blood vessels and small numbers of scattered inflammatory cells. These can grow rapidly and have mitotic figures, but the overall size of the mass tends to be small. Variable SMA, desmin, and laminin staining is expected given that these tumors are composed of mixed fibroblasts and myofibroblasts. Two cases from our set had histopathologic features suggestive of nodular fasciitis (cases 11, 13).
Low-grade myofibroblastic sarcoma (an entity distinct from ALK-positive myofibroblastic tumors 43 ) is a low-grade malignancy in people that commonly recurs. 51 High-grade myofibroblastic sarcomas are considered undifferentiated pleomorphic sarcomas, an entity that falls under the STS classification in dogs. Generally, electron microscopy is used for diagnosis because IHC will do little to distinguish among the other differentials. In our subset, low-grade myofibroblastic sarcomas were considered for most of the cases not considered leiomyoma or leiomyosarcoma.
Stringent but clearly defined diagnostic criteria were used to ultimately determine whether a tumor should be deemed smooth muscle in origin. We considered this stringency critical given that other sarcomas may express these IHC markers. For example, SMA can be expressed in other tumors, such as myofibroblastic tumors, 29 osteosarcoma, 36 rhabdomyosarcoma, 28 perivascular wall tumors, 3 and GISTs. 45 To complement IHC stains, MT was used to confirm minimal collagen production by the neoplastic cells, a feature not expected in smooth muscle tumors. Our stringent criteria may have led to fewer diagnoses, albeit with higher confidence.
Of note, case 3, angioleiomyosarcoma, was considered an exception in this case given that its close association with vasculature suggested that the anatomic origin was the vascular wall. This case had variable collagen deposition and we speculated that the areas with abundant collagen may be similar to vascular fibrosis seen in vasculopathies. Whether this tumor should be considered a smooth muscle tumor or perivascular wall tumor is debatable and further research is required.
There are several important limitations to our study, the most important of which is small sample size, which precluded statistical comparisons and limited our ability to draw conclusions regarding the clinical behavior of non-visceral leiomyosarcomas. Commercial and primary-care diagnostic laboratories may have a wider database of dermal and subcutaneous masses than tertiary institutions. Another limitation is the lack of a gold standard for comparison for leiomyosarcoma and, therefore, reliance on unvalidated and arbitrary IHC scoring criteria and morphologic characteristics for diagnostic criteria. The importance of a threshold at which staining is called positive or negative is established in the human literature 38 ; however, even in human pathology, most work has been with carcinomas of unknown origin rather than sarcomas. The movement toward using a panel of IHC stains rather than a single stain and identification of specific mutations associated with neoplastic thresholds may explain the lack of defined thresholds to identify mesenchymal tumors that may be of smooth muscle origin. Future studies should aim to better characterize the staining pattern of smooth muscle tumors in known common sites of origin, such as the GI tract and reproductive tract, to gain more reliable information on the diagnostic criteria for these tumors.
Other common limitations for IHC studies include false-negative results, which can arise from inadequate antigen preservation or expression. Especially with older, formalin-fixed samples, and samples that were maintained in formalin for longer than optimal before processing, negative immunostaining may reflect the age, storage condition, or suboptimal processing of the sample rather than being a true negative. Although well-differentiated tumors often maintain expression of several markers, these same markers may be lost in most cells in poorly differentiated tumors, or gain other traits, that together, can preclude a definitive interpretation. 10 Therefore, the absence of labeling does not always exclude a diagnosis, although true loss of expression may suggest that the behavior of the neoplastic cells would change accordingly. It is also possible that tumors that should be classified as leiomyosarcomas were not identified in our initial search given the previously mentioned limitations in diagnosis (e.g., H&E appearance overlapping with other sarcomas, lack of routine IHC use). Therefore, a set of leiomyosarcomas was likely not captured by our search terms. Other limitations are inherent to the retrospective nature of our study, including the variable availability of clinical follow-up for these cases.
Our study highlights the uncommon nature of canine non-visceral leiomyosarcoma and potential differentials that should be considered. IHC staining is useful and important; however, pathologists should recognize that there can be different tumors with overlapping staining patterns. SMA alone did not lead to a definitive diagnosis, and desmin may not be useful for the diagnosis of leiomyosarcoma because of potential loss of staining in this cohort. The loss of desmin expression should be investigated in leiomyosarcoma as a potential marker of more aggressive behavior. Although the importance of histologic and molecular subtype of sarcomas has been demonstrated in people,1,21,50 our study emphasizes the need for continued efforts in veterinary species to ensure that these subtypes are accurately classified to potentially predict behavior and guide treatment more definitively. Future studies with alternative tools such as electron microscopy and molecular characterization in addition to IHC staining are required in veterinary medicine to better document the spectrum of IHC staining variation and clinical behavior of sarcoma subtypes, including non-visceral SMA-positive mesenchymal tumors such as leiomyosarcomas and myofibroblastic tumors.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387221083570 – Supplemental material for Retrospective immunohistochemical investigation of suspected non-visceral leiomyosarcoma in dogs
Supplemental material, sj-pdf-1-vdi-10.1177_10406387221083570 for Retrospective immunohistochemical investigation of suspected non-visceral leiomyosarcoma in dogs by Rachel V. Brady, Robert B. Rebhun, Katherine A. Skorupski, Jenna H. Burton, Sami Al-Nadaf, Eunju Choi and Jennifer L. Willcox in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
Portions of this work were presented at the Veterinary Cancer Society Annual Conference, 2020, virtual meeting.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Supported by the Center for Companion Animal Health, School of Veterinary Medicine, University of California, Davis, grant/award CCAH 2019-39-R.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
