Abstract
Inflammatory mammary carcinoma (IMC) is a specific type of rare, very aggressive, and highly metastatic mammary cancer in both human beings and dogs. A 10-year-old female spayed Shih Tzu dog was diagnosed with secondary IMC. At necropsy, brain metastasis of mammary neoplastic cells was observed in tissues of the cerebrum and cerebellum. Metastases were also found in other distant organs such as heart, lung, liver, spleen, and inguinal lymph node. There is limited data about the metastasis of IMC and its pattern. The current report of IMC with brain metastases contributes to the understanding of metastatic IMC.
Canine mammary gland tumor is one of the most common neoplasias, 20 accounting for approximately 50% of all tumors in female dogs. 6 All malignant canine mammary tumors have the potential to metastasize and, in general, metastasis tends to occur via the lymphatic system to the inguinal lymph nodes or hematogenously to the lungs or to more distant body sites including the liver, spleen, heart, bone.11,15,20 Brain metastasis can be common, although the situation in canine mammary tumors is unclear.1,5
Inflammatory mammary carcinoma (IMC) is a specific type of rare, very aggressive, and highly metastatic mammary cancer in both human beings and dogs.10,17 Clinical features include the presence of pain, erythema, edema, and ulceration in the skin of the mammary gland region. These features are similar to symptoms of inflammatory diseases such as mastitis and dermatitis.18,22 Two forms of IMC have been identified clinically: primary and secondary. 18 Primary IMC occurs suddenly in dogs without lesions of mammary tumors, and secondary IMC develops with mammary tumors. 18 Dermal lymphatic infiltration (tumor emboli) is the sole histological hallmark for pathologic diagnosis of IMC.16,18
Previous studies have reported IMC with metastases in the inguinal lymph node, muscle, lung, heart, uterus, and urinary bladder17,18; however, the number of cases was limited. The present case report describes IMC with metastases to the brain in a canine that also involved other distant organs. The clinical features, histopathology, and immunohistochemistry are also reported.
A formalin-fixed mammary gland tissue sample was sent from a private clinic to the Small Animal Tumor Diagnostic Center, College of Veterinary Medicine, Konkuk University (Seoul, Korea) under suspicion of a suspected mammary gland tumor. The sample was acquired from a 10-year-old female spayed Shih Tzu dog that had received an ovariohysterectomy 1 year previous. The patient presented with a mass in the right fourth mammary gland. Grossly, the mass was firm and approximately 2 cm × 1.5 cm in size. Fine-needle aspiration revealed an abnormal number of adipose cells. A complete blood cell count was performed; the packed cell volume was 41.53% (reference [ref.] interval: 38–50%), platelet count was 182 × 103/µl (ref. interval: 180–500 × 103/µl), and white blood cell count was 13.76 × 103/µl (ref. interval: 6–17 × 103/µl).
After a right caudal mastectomy, the mammary gland tumor was classified according to the World Health Organization Histological Classification System as a solid carcinoma. 14 No chemotherapy was conducted after surgical removal of the mammary tumor. The postoperative state and appetite of the patient was relatively good for approximately 1 week, with the patient displaying no clinical signs. However, the patient then developed signs of depression, anorexia, dyspnea, and neurological signs including head tilting. In addition, pain, erythema, and edema in the right inguinal region were evident. The patient died 34 days after surgery, showing epistaxis and ulceration in the inguinal region involving a 2 cm × 2.5 cm mass and multiple nodules in the third mammary gland and inguinal region, respectively (Fig. 1A).
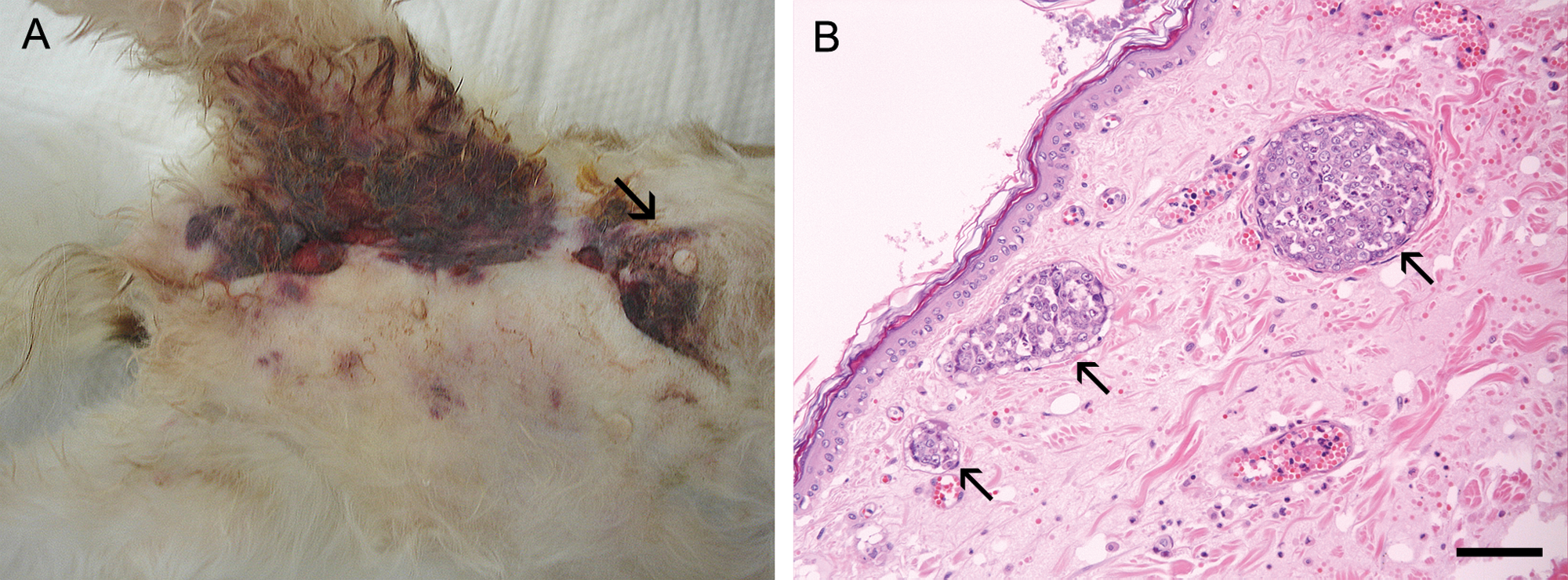
Ten-year-old female spayed Shih Tzu dog.
At necropsy, postmortem examination findings included dilation of the heart, hemorrhage in the pyloric region of the stomach, and pulmonary edema. Numerous nodules were found, which were thought to represent metastasis to the liver, lung, spleen, and inguinal lymph nodes. The latter were enlarged. Multifocal and firm nodules, 0.1–0.2 cm in size, were evident in the liver, and palpated solid nodules were observed in the lung. Numerous nodules 0.3–0.5 cm in size were present in the spleen. Although nodules were not grossly evident in the brain, a sample was examined microscopically.
All specimens collected at necropsy were fixed in 10% neutral buffered formalin and subsequently embedded in paraffin. Serial 4-μm thick sections including the abdominal mass, heart, lung, liver, spleen, kidney, urinary bladder, stomach, intestine, submandibular, inguinal, mesenteric lymph nodes, and brain were acquired from each paraffin block and stained with hematoxylin and eosin.
The metastatic tissues were evaluated immunohistochemically to confirm the origin of the tumor cells. A monoclonal mouse anti-human cytokeratin a antibody was used, given the epithelial origin of the mammary gland tumor.7,19
Sections were deparaffinized and treated with 3% hydrogen peroxide solution for 20 min. After washing with phosphate buffered saline (pH 7.4, 0.1 M), the antigens were retrieved in Tris–ethylenediamine tetra-acetic acid buffer (pH 9.0) for 10 min in a microwave oven (650 W, high power). Sections were incubated with a 1:100 dilution of primary antibody for 2 hr at room temperature after cooling and washing. The secondary polymer b was applied to each slide for 40 min. The slides were incubated with the requisite substrate after washing. The color reaction was stopped in distilled water, followed by counterstaining with Harris hematoxylin.
Microscopically, the abdominal mass was made up of invasive tumor cells in the abdominal wall muscularis. This has been described as a diffusely infiltrating neoplasm composed of large, pleomorphic cell with bizarre nuclei rich in chromatin. Tumor embolization of dermal lymphatic vessels, which are a histological hallmark of IMC, were predominant (Fig. 1B). Adenocarcinoma cell clusters indicative of metastases from mammary carcinoma were observed in not only mammary masses but also in the heart, lung, liver, spleen, muscle, and inguinal lymph nodes. In addition, some regions in cerebral cortex and cerebellar tissue were randomly evaluated, although no evidence of metastasis was grossly evident. Metastatic tumor cell clusters of different sizes were evident in various regions of the cerebral cortex (Fig. 2) and the cerebellum (data not shown).
The immunohistochemical results revealed the epithelial origin, on the basis of the strong cytoplasmic staining in liver, lung, and spleen, in addition to brain metastases (Fig. 3). Inflammatory mammary carcinoma is the most malignant type of mammary carcinoma, exhibiting a high metastatic potential in both human beings and dogs.18,22 The carcinoma develops with rapid onset with a survival time of only 25–60 days.12,16,18 Distant metastases in organs other than the brain have been reported.17,18 In the present report, the dog died due to complications from metastases to the brain and other distant organs. Death occurred 34 days after surgical removal of the primary mammary tumor, and 27 days after the initial clinical signs of secondary IMC.
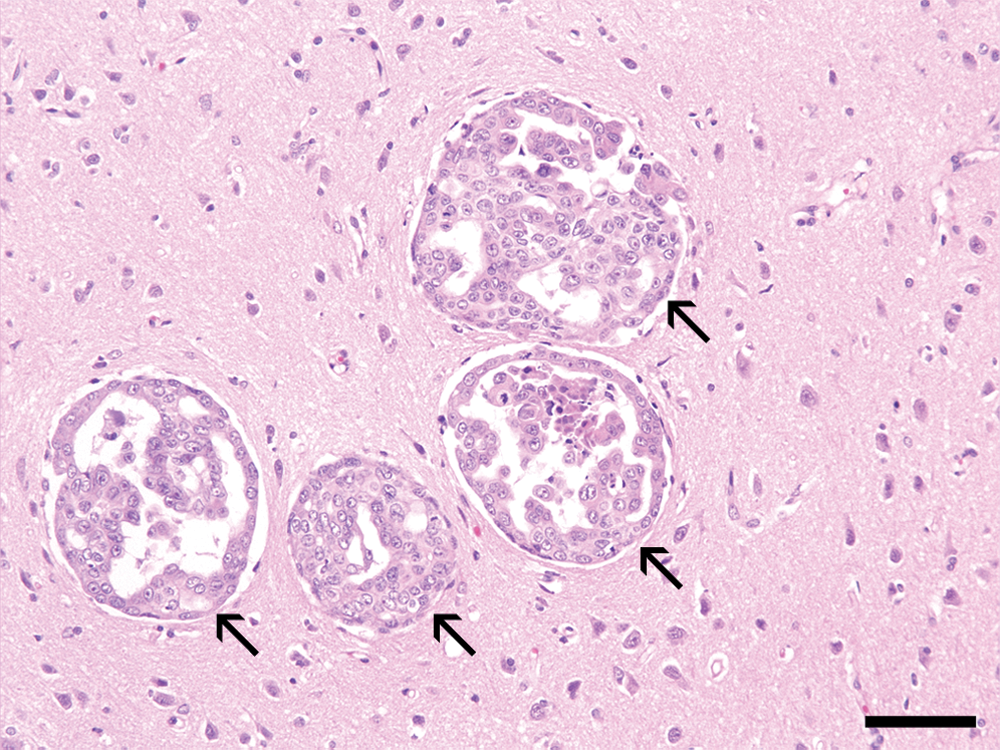
Ten-year-old female spayed Shih Tzu dog. Coloniza-tion of metastatic mammary neoplastic cells (arrows) in brain tissues. Hematoxylin and eosin. Bar = 70 µm.
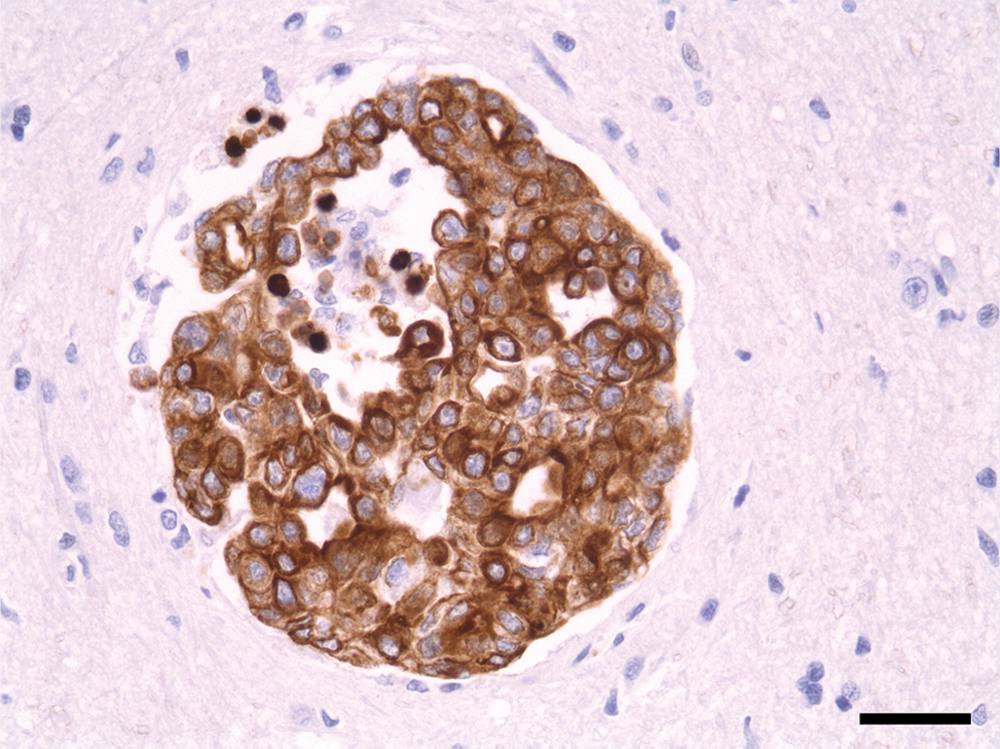
Ten-year-old female spayed Shih Tzu dog. Metastatic epithelial tumor cells showing strong immunolabeling against cytokeratin in cerebrum. Peroxidase immunohistochemistry with hematoxylin counterstain. Bar = 36 µm.
Inflammatory mammary carcinomas frequently develop in sexually intact female dogs and cats.17,18 However, the patient in this current case was a spayed female who received an ovariohysterectomy as an adult. The relationship between ovariohysterectomy of adult dogs and development of IMC is not known, 18 although the development of IMC associated with a phase of the ovarian cycle has been described, 18 and the timing of ovariectomy in adult dogs is associated with the prognosis of mammary carcinomas. 21
Inflammatory mammary carcinoma may be misdiagnosed as mastitis and dermatitis due to the presence of a cutaneous inflammatory lesion.18,22 The present case presented not only a skin lesion but also the neurologic sign of head tilting after surgical removal of the mammary tumor. Therefore, IMC should be additionally differentiated from other neurologic diseases. Metastatic tumor colonization of mammary carcinoma displaying potent immunolabeling with antibody to cytokeratin was evident in the cerebrum and cerebellum in the current case.
Studies within the last decade have not reported neurologic signs or brain metastasis in dogs with IMC.4,13,16,18 Interestingly, in a 2010 study, difference in the metastatic pattern was observed between canine IMC and noninflammatory malignant mammary tumors (NIMMTs); the incidence of metastasis to the urinary bladder and reproductive organs was higher in dogs with IMC than in dogs with NIMMTs. 4 Brain metastasis was not observed in dogs with IMC but was observed in 3 dogs with NIMMTs, although no statistical significance was apparent. The present case does not support these prior results (i.e., the absence of evidence of metastasis into the urinary bladder and reproductive organs, due to the previous ovariohysterectomy). However, the current case could provide more information for unique metastatic pathogenesis of canine IMC. 4
In human beings, breast cancer is one of the most common sources of brain metastases, which an incidence of 15–25%. 23 Overall prognosis of patients with metastatic brain tumor from human breast cancer is poor.3,8 The ability of canine mammary tumors to metastasize to the brain has also been well documented.2,9 In a previous retrospective study, the evidence of metastasis to the brain was found in 12% of 54 postmortem samples of female dogs with mammary tumors. 5 However, the data did not reveal the true prevalence and survival time, and it is not known whether the mammary tumors included IMC.
Additional information about metastatic IMC is needed. Because canine IMC is very rare, the true incidence is not exact, and the number of distant organs examined is limited upon necropsy. The current report shows secondary IMC in a spayed female dog with brain metastases including other distant organs such as the heart, lung, liver, spleen, muscle, and inguinal lymph node, and provides further valuable information about metastatic IMC.
Footnotes
Acknowledgements
The authors thank Ms. R. H. Jang for her excellent technical assistance and the private animal clinic for providing the canine samples.
a.
Clone MNF116, Dako Denmark A/S, Glostrup, Denmark.
b.
REAL™ Envision kit, Dako Denmark A/S, Glostrup, Denmark.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This study was supported in part by a grant from the Konkuk University Research Foundation 2011.
