Abstract
Erysipelothrix rhusiopathiae infection and septicemia occurred in a 5-d old Boer goat found dead on a farm in western Pennsylvania. On autopsy, there was moderate, focally extensive hemorrhage along the remnants of the urachus and umbilical arteries and the apex of the urinary bladder. Microscopic examination of immunohistochemical stained tissues revealed abundant intracellular and extracellular E. rhusiopathiae antigen–positive bacilli in all tissues stained, including lung, heart, liver, skeletal muscle, kidney, and thymus. Bacteria isolated from liver and urachus were identified as E. rhusiopathiae by MALDI-TOF mass spectrometry and further confirmed by a PCR assay. An epidemiologic investigation was conducted via an on-farm questionnaire after the owners noted a 70% mortality rate from the 2019 kidding season. The epidemiologic investigation showed that E. rhusiopathiae, an opportunistic zoonotic organism, was introduced to the farm through a breach in biosecurity and was likely perpetuated among the resident poultry species.
The first case of erysipelas in a goat was reported in 1962 and was attributed to exposure of the goat to massive amounts of Erysipelothrix rhusiopathiae originating from an adjacent swine meat-packing plant. 11 Subsequent experiments showed that goats are relatively resistant to erysipelas, but under rare conditions, can contract the disease when exposed to 12 × 1011 Erysipelothrix organisms. 11 E. rhusiopathiae is a slender, non-motile, non–spore-forming, facultatively anaerobic, gram-positive bacillus that can survive in various environmental conditions, including dead animal tissue, decaying organic matter, water, and up to 2 wk in mud.7,9 Erysipelas is an economically important disease of various species including swine, turkeys, chickens, ducks, emus, and sheep. 9 This bacterium is distributed worldwide and is considered an opportunistic zoonotic organism. 9 E. rhusiopathiae causes erythematous, edematous skin lesions, primarily distributed on the hands and fingers of the infected human. 7
A 5-d-old, male Boer goat was submitted to the Pennsylvania Veterinary Laboratory (PVL; Harrisburg, PA, USA) in November 2019 for autopsy. The owner of the farm had reported a 70% mortality rate of kid goats in the 2019 kidding season (first week of November 2019 to mid-December). Among affected goat kids, 14 of 20 (70%) had died within the first week of life (0–7 d); 6 of 20 (30%) had survived past one month of age. All goat kids appeared healthy after birth; deaths occurred within a week with no apparent clinical signs (Fig. 1).
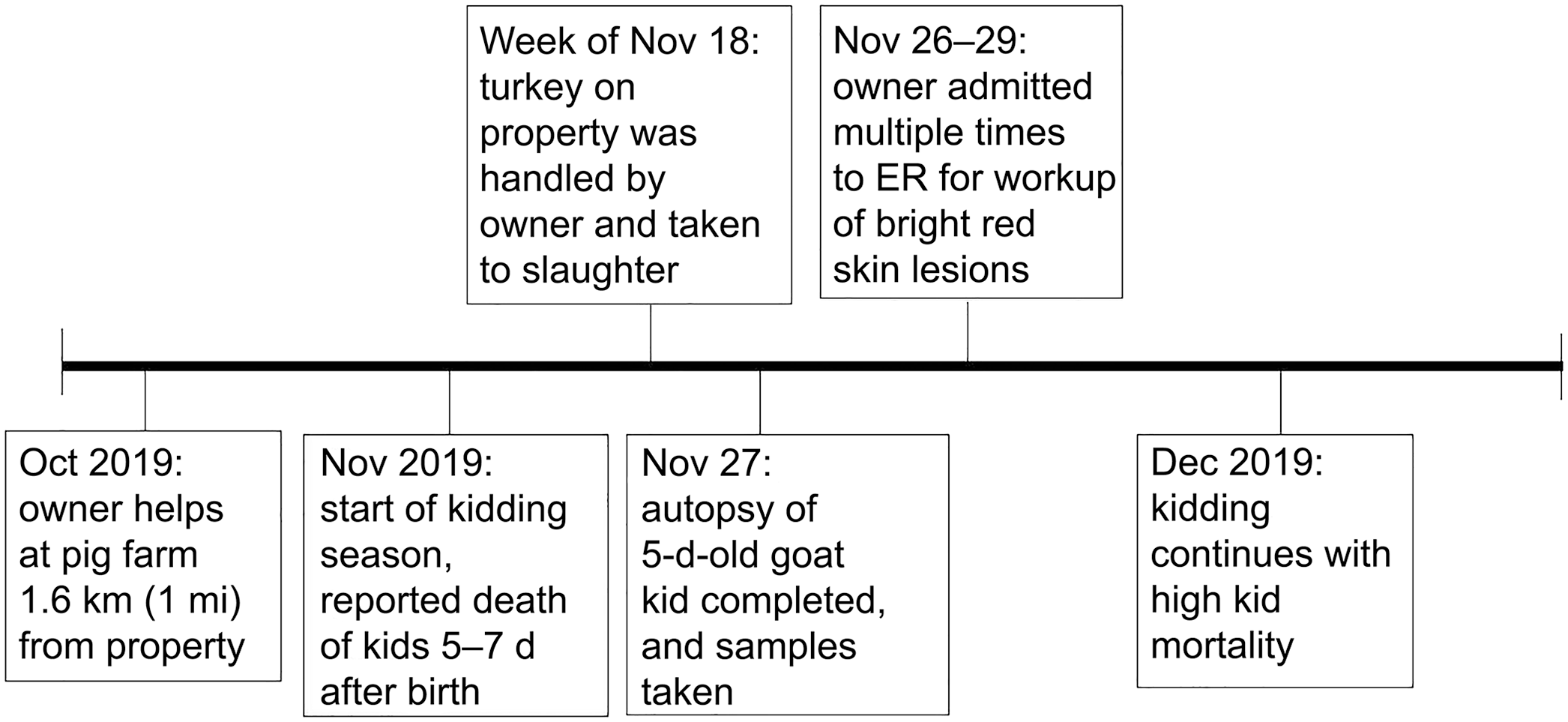
Timeline of Erysipelothrix rhusiopathiae exposure to owner, farm, and subsequent deaths of kid goats.
At autopsy, the goat kid carcass was fresh and in adequate nutritional condition. Gross lesions in this goat were mild and nonspecific. The umbilicus was grossly unremarkable. There was moderate, focally extensive hemorrhage along the remnants of the urachus and umbilical arteries and the apex of the urinary bladder. The heart had moderate diffuse pallor. The abomasal mucosa had numerous brown petechiae. No significant lesions were observed in other tissues.
Samples of skeletal muscle, heart, liver, lung, kidney, brain (medulla, cerebellum, cerebrum), thymus, rumen, reticulum, omasum, abomasum, small intestine, cecum, and spiral colon were fixed in 10% neutral-buffered formalin, processed routinely, and sections stained with H&E. Liver, urachus, abomasum, and intestine were swabbed for routine aerobic culture, and abomasum and liver were swabbed for clostridial culture. The swabs for aerobic cultures were plated on blood agar (tryptone soya agar with 5% sheep blood; Remel) and incubated in ambient air at 37°C for 48 h. The abomasum and intestinal swabs were also plated on selective medium (Tergitol 7 agar; Remel). The swabs for clostridial cultures were plated on brucella blood agar and phenylethyl alcohol blood agar (Anaerobe Systems) and incubated under anaerobic conditions (Mitsubishi AnaeroPack-Anaero gas generator; Thermo Fisher) at 37°C for 48 h. The plates were examined at 24 and 48 h of incubation, and selected bacterial colonies with distinct phenotypes were subcultured onto nonselective medium, blood agar or brucella blood agar, for identification by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI Biotyper; Bruker) as described previously. 6 Mass spectra data acquisition and analysis were performed (Microflex LT/SH mass spectrometer, MBT Compass software v.4.1.80; Bruker), and spectra were compared with the MBT Compass library (v.9.0; Bruker), which contains 8,468 entries from 540 genera and 2,969 species of clinical importance. A bacterial test standard (Bruker) was used for calibration of the instrument before each run, following the manufacturer’s instructions.
The owner of the farm requested further investigation from the U.S. Department of Agriculture, Animal and Plant Inspection Service (USDA-APHIS) and Pennsylvania Department of Agriculture (PDA). An epidemiologic investigation was conducted through a questionnaire and a walk-through of the farm facilities following the deaths. USDA-APHIS and PDA completed the investigation using the principles of biosecurity. 10 After discovering a lack of farm biosecurity protocols, PDA and USDA used a questionnaire to determine possible mode and time of introduction of the bacterium to the farm. The farm owner explained that before kidding season in October 2019, he had helped on a neighboring pig farm that was located <1.6 km (1 mi) from his own farm. Biosecurity measures for visitors were not implemented on either farm. When the owner arrived home, he proceeded with his normal routine without disinfection of his clothing, boots, or farm utility vehicle.
Microscopic examination of the lung revealed diffuse expansion of alveolar septa up to 6 times normal by fibrin thrombi, macrophages, lymphocytes, neutrophils, and plasma cells. The myocardial interstitium and interstitial blood vessels were infiltrated by few-to-moderate numbers of neutrophils and fewer macrophages, with occasional necrosis of adjacent myofibers. In the liver, there was occasional random individual hepatocyte necrosis, and blood vessels and sinusoids contained numerous neutrophils and macrophages, fewer lymphocytes and plasma cells, and rare clusters of bacilli. Macrophages in the lung (Fig. 2A), heart (Fig. 2B), gastrointestinal mucosa, and renal medullae were distended with intracytoplasmic small bacilli.
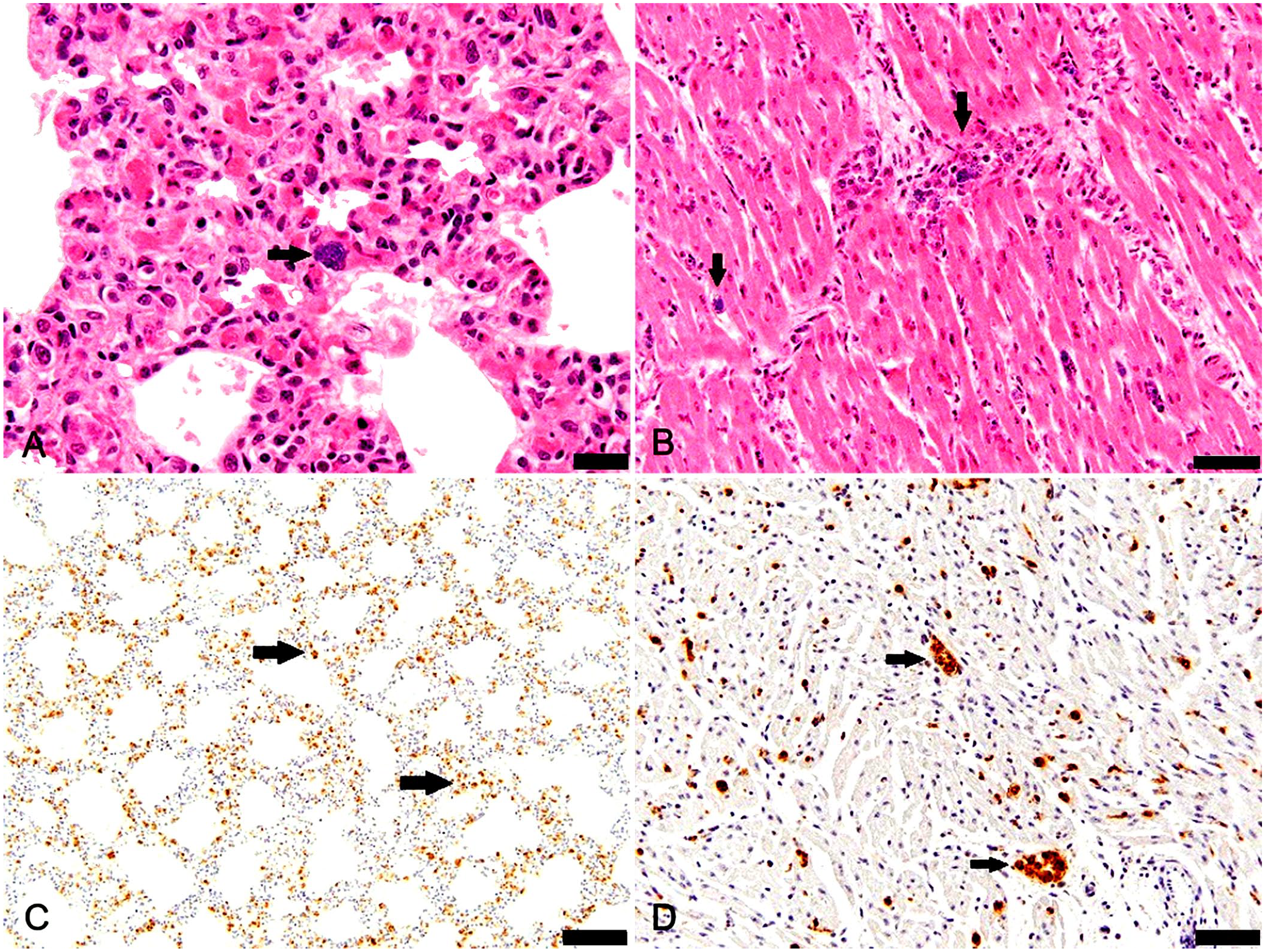
Erysipelas in a goat.
The small bacilli observed on microscopic examination of H&E-stained tissues were identified as E. rhusiopathiae via immunohistochemistry (IHC; rabbit polyclonal antiserum, Iowa State Veterinary Diagnostic Laboratory [ISVDL], Ames, IA, USA). 4 Abundant intracellular and extracellular E. rhusiopathiae antigen–positive bacilli were observed in all tissues stained, including lung, heart (Fig. 2C, 2D), liver, skeletal muscle, kidney, and thymus.
Bacterial isolates from the abomasum and intestine were identified as Escherichia coli. Bacteria isolated from liver and urachus matched E. rhusiopathiae with a highest Bruker score of 2.35; all top 10 hits matched E. rhusiopathiae spectra in the Bruker MBT Compass Library. DNA from a formalin-fixed, paraffin-embedded heart and skeletal muscle tissue block was extracted (DNA extract all reagent kit; Thermo Fisher) after deparaffinization with xylene. The extracted DNA was tested by an Erysipelothrix spp. multiplex real-time PCR assay at the ISVDL, as described previously. 5 The PCR assay targets the noncoding region downstream of the 5S rRNA coding region in the Erysipelothrix genome and is designed to detect and differentiate E. rhusiopathiae, E. tonsillarum, and Erysipelothrix sp. strain 2. The heart and skeletal muscle pool tested positive for E. rhusiopathiae (Ct = 35.9) and negative for E. tonsillarum and Erysipelothrix sp. strain 2. The high Ct value is possibly the result of degradation of nucleic acid in formalin-fixed tissue. 1
Our epidemiologic investigation supported the likelihood that a common pathogen was responsible for the deaths of the goat kids. Although laboratory data were only available for one goat kid, the results from the epidemiology questionnaire provided evidence that the death of all of the goat kids could be attributed to E. rhusiopathiae. The questionnaire identified husbandry issues that promoted exposure and subsequent infection of the goat kids with E. rhusiopathiae as well as with other disease agents that the farm had experienced in previous kidding seasons. The questionnaire also revealed that there were multiple animal species on the farm, including pigs and turkeys, that are particularly susceptible to E. rhusiopathiae. The owner noted that there were sudden deaths among the turkeys present on the property prior to the goats dying. Although erysipelas was not definitively diagnosed in the turkeys, the turkeys were a possible source of infection for the goats. Neither the age of the does, which ranged from 1–8-y-old, nor the feeding methods of kids (bottle vs. dam feeding) was determined to be a factor in the goat kid deaths. The most significant common factor among the deaths was that none of the affected goat kids had 7% iodine applied to their umbilical cord or navel at birth, which is recommended for the prevention of bacterial omphalitis and subsequent septicemia. 2
At the property, USDA APHIS and PDA found many different species of animals living on the farm including steers, horses, dogs, chickens, turkeys, ducks, and pigs (seasonal). Although the poultry species were housed separately from the goats, water runoff from the poultry shed to the rest of the property was noted (Suppl. Fig. 1). The farm owner had not implemented proper biosecurity techniques, such as using a foot bath when entering facilities, changing clothing, kidding in a separate pen, removing sick animals from pens, and disinfecting farm utility vehicles upon use or after returning to the property.
Transmission of erysipelas to an individual animal occurs through cutaneous exposure or ingestion of contaminated soil and water particles.7,9 Diseased animals shed the organism in their feces, urine, saliva, and nasal secretions. Domestic swine are considered the most important animal reservoir of E. rhusiopathiae.7,9 In swine, erysipelas can manifest in 3 main clinical forms: acute (septicemia), subacute (urticaria), and chronic (arthritis, lymphadenitis, endocarditis), with all 3 forms clinically present in a herd. 7 In the acute form, mortality is usually high. 7 In sheep, the disease causes arthritis in lambs as a result of umbilical infection. 7 Avian infections result in septicemia with mortality rates of 2.5–25%. 9 Turkeys are the most affected species, with predisposing factors related to environmental conditions. 3
Biosecurity measures are important in controlling the transmission of this organism given that E. rhusiopathiae can survive for an extended time in the environment. 7 Vaccination is a vital control measure in swine and avian species; however, a vaccine is not licensed for goats. 7 After our investigation was completed, USDA-APHIS and PDA did provide a biosecurity plan for the farm. The owners removed all poultry from the premises and implemented changes, which included: general precautionary measures with posting signs and limiting guests, cleaning and disinfection of farm equipment and clothing and shoes, and isolation of sick animals. The owner was also advised on best goat husbandry practices, including navel dipping at birth. On follow up in the 2020 kidding season, the owner was still using the biosecurity plan, and their mortality rate in goat kids had dropped from 70% to 10%.
Differential diagnoses for perinatal death in goat kids include infectious diseases caused by E. coli, Clostridium spp., Chlamydia spp., Leptospira spp., and Toxoplasma spp., as well as colostrum issues, maternal and kid nutrition, and environmental factors. 8 No other aerobic bacteria or Clostridium spp. were recovered from tissue samples, and Chlamydia spp. were not detected in lung by a PCR assay. In ruling out nutritional deficiencies, inductively coupled plasma–mass spectrometry was used to evaluate calcium, copper, iron, magnesium, manganese, molybdenum, selenium, and zinc concentrations in a sample of fresh liver. All concentrations were within reference intervals. Histopathology did not support a diagnosis of toxoplasmosis or leptospirosis.
Limitations of our investigation prevented us from understanding more about this outbreak. Additional work, such as sampling of soil and/or serology on remaining animals would have provided more insight but was not undertaken. Also, further characterization of the isolate by serotyping or other methods was not possible because the organism was lost before being stored.
Our investigation provides evidence that E. rhusiopathiae entered the property through a breach in biosecurity and contaminated the environment. The organism was likely perpetuated among poultry species on the farm, and, without proper biosecurity measures, the owner of the farm spread the organism throughout the property. Failure to properly disinfect the navel of the neonatal kids with 7% iodine provided a point of entry for E. rhusiopathiae with subsequent ascending umbilical infection and septicemia. At the same time, the owner also complained about going to the emergency room 3 times because of diffuse, cutaneous, urticarial lesions on his body that developed after handling the turkeys on the property and caring for the goats. We believe that these lesions could be the result of the owner being an accidental host for E. rhusiopathiae.
Supplemental Material
sj-docx-1-vdi-10.1177_10406387211072831 – Supplemental material for An unusual outbreak of erysipelas on a goat farm in Pennsylvania
Supplemental material, sj-docx-1-vdi-10.1177_10406387211072831 for An unusual outbreak of erysipelas on a goat farm in Pennsylvania by Heather A. Palm, Nagaraja R. Thirumalapura, Lore A. Boger and Stephanie R. Ringler in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the owners of the farm for allowing our team to do an in-depth investigation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
