Abstract
Yersinia pseudotuberculosis is a recognized zoonotic food-borne pathogen; however, little is known about the ecology and epidemiology of diseases caused by the bacterium in California. The objective of the current study was to contribute to the knowledge of the diseases caused by Y. pseudotuberculosis in goats, the animal species most frequently reported with clinical yersiniosis to the California Animal Health and Food Safety Laboratory System, to better understand the epidemiology of this disease. A 23-year retrospective study was conducted to characterize the syndromes caused by the bacterium in goats and their temporospatial distribution, and to determine the number of cases in other animal species. Yersinia pseudotuberculosis–associated disease was diagnosed in 42 goats from 21 counties, with a strong seasonality in winter and spring. Most cases (88%) were observed within particular years (1999, 2004–2006, 2010–2011). The most frequently diagnosed syndrome was enteritis and/or typhlocolitis (64.3%), followed by abscessation (14.3%), abortion (11.9%), conjunctivitis (4.75%), and hepatitis (4.75%). Among other animal species, 59 cases were diagnosed in non-poultry avian species and 33 in mammals other than goats.
Introduction
Yersinia pseudotuberculosis, a Gram-negative aerobic or facultative anaerobic rod-shaped bacterium in the family Enterobacteriaceae, is a significant and well-known cause of disease in numerous species of domestic and wild mammals and birds,10,26,32 and is also a recognized zoonotic food-borne pathogen. 16 The bacterium has a worldwide distribution and is a common inhabitant of the intestinal tract of a wide range of asymptomatic birds, rodents, livestock, primates, and laboratory animals. 2 Transmission generally occurs by the fecal-oral route, after ingestion of contaminated food or water. 2 The organism survives for a long period of time outside the host in the environment and can replicate in temperatures as low as 4°C. 2
Infection and associated disease have been described in many species, including human beings,17,29 sheep, 27 cattle,26,33 pigs,27,29 deer,14,29,37 horses, 8 buffalo, 13 exotic ungulates, 2 birds, 32 lagomorphs (rabbits and hares),22,29 rodents (hamsters, guinea pigs, beavers),10,22,29 bats, 7 and nonhuman primates. 5 Many carnivores including dogs and cats are relatively resistant to the clinical disease; however, they can shed the bacteria in the feces. 2
In human beings, the disease is considered a food-borne infection and several individual cases or community outbreaks have occurred in the past decades after ingestion of contaminated milk products, 23 undercooked or raw meat (e.g., pork),2,12 fresh vegetables (carrots, lettuce),17,20 vegetable juices, water, and sandwiches. 30 A sudden onset of pseudotuberculosis in human beings was reported in France in the winter of 2004–2005, although a food-exposure analysis did not identify a potential common food source, and the increased incidence was presumed to be due to increased prevalence of the pathogen in rodent reservoirs. 31 Little is known about the epidemiology in the United States, where diseases caused by Y. pseudotuberculosis infection in human beings are rarely reported. 18
The diseases in human beings usually consist of mesenteric lymphadenitis, enteritis with acute abdominal pain (pseudoappendicitis), septicemia, 2 and rarely arthritis. 16 Patients with diabetes mellitus, hepatic cirrhosis, and hemochromatosis or hemosiderosis are at higher risk of developing potentially lethal systemic infections, 16 and the septicemic form has also been described in patients with Human immunodeficiency virus infection.1,21 Rarely, the bacterium has been associated with hemolytic uremic syndrome.9,23
In ruminants in general and goats in particular, Y. pseudotuberculosis has been associated with enterotyphlocolitis and drop of milk production, 24 lymphadenitis, 24 abortion and neonatal death, 36 mastitis,6,15,25 septicemia, and ocular disease.19,34,35 However, most cases of Y. pseudotuberculosis published in goats are of individual animals or small groups of goats; no systematic studies of large numbers of Y. pseudotuberculosis cases in goats have been published. The objective of the current study is, therefore, to characterize the main syndromes caused by Y. pseudotuberculosis in a large number of goats diagnosed at the California Animal Health and Food Safety Laboratory System (CAHFS) during a 23-year period, aiming to contribute to the knowledge of this zoonotic disease in California.
Materials and methods
A retrospective search using the term “yersin” in the diagnosis field for caprine submissions received between January 1, 1990 and December 31, 2012 (23 years) was performed using the CAHFS database. All of the cases in which Y. pseudotuberculosis had been isolated and considered a primary pathogen were selected. The data on each final report was analyzed on a case by case basis to retrieve information related to the age and production class of the goat, pathological findings, concurrent diseases, and hepatic levels of copper and selenium. The bacteriologic techniques used to isolate the bacterium were also evaluated. These included regular aerobic bacterial cultures in sheep blood and MacConkey agars with incubation at 35–39°C in 5–10% CO2 for 48 hr, and Yersinia cold enrichment. The cold enrichment involved placing 0.5–1 g of sample in 5 ml of sterile phosphate buffered saline, refrigerating at 3–5°C, subculturing weekly for 3 weeks to Yersinia selective agar (CIN agar) and MacConkey agar, with aerobic incubation at 23–26°C for 42–48 hr. Yersinia pseudotuberculosis isolates were identified by standard phenotypic methods, colony morphology, growth characteristics, conventional biochemical tests, and a commercial identification kit a as previously described. 4 These tests were performed according to CAHFS standardized operating procedures. The date of submission of the carcasses to the laboratory (temporal distribution) and the geographic location of the animals approximated by the zip code of the collection site or the owner’s address were also analyzed and mapped using commercial software. b For interspecies comparison, a similar search was performed to retrieve the number of cases diagnosed in avian species, cattle, sheep, llamas, pigs, horses, and other mammals in the same period, although these cases were not analyzed in detail.
Results
Yersinia pseudotuberculosis was considered a significant pathogen in 42 goat cases included in 41 submissions (1 submission included 2 different cases). Thirteen of the 42 cases (30.9%) occurred in meat breeds, 11 in dairy breeds (26.2%), 1 in a fiber breed (2.4%), and no breed information was available in 17 of the 42 cases (40.5%). In 16 of the 41 (39%) submissions, the animals were on pasture (with or without grain or mineral supplementation), while in 1 (2.4%) they were housed with hay as a main food source. No information was available in the remainder 24 of the 41 (58.6%) submissions.
Thirty-five of the 42 cases were diagnosed at CAHFS Davis in northern California from a total of 2,682 goat necropsies performed at this laboratory in the study period (1.3%). The remaining 7 cases were diagnosed at CAHFS Tulare (central California) from a total of 1,007 goat necropsies (0.7%) performed in the same period at this laboratory. No cases were diagnosed at CAHFS San Bernardino (the southernmost location of the laboratory system) despite the fact that 766 goat necropsies were performed at this site in the same study period.
The numbers of cases of different syndromes associated with Y. pseudotuberculosis infection by age are shown in Table 1. All but 2 cases from 1 flock in Idaho were from California (Fig. 1). Cases showed overall increased secular trend, with high apparently cyclical occurrences in 1999, 2004–2006, and 2010–2011, periods in which 88% of the cases occurred. During the remaining years of the series, at most 1 case was reported. Twenty-three of the 42 cases (54.8%) were diagnosed in 3 consecutive years (2004, 2005, and 2006; Fig. 2). Regardless of the syndrome, all 42 cases occurred with marked seasonal clustering between late November and May (Fig. 3).
Number of cases of Yersinia pseudotuberculosis in goats by syndromes and age group in California over the period of 1990–2012.

Age specified as “adult” in diagnostic report.
With (14) or without (13) concurrent detectable septicemia.
Five does aborted 7 fetuses (2 twin pregnancies).
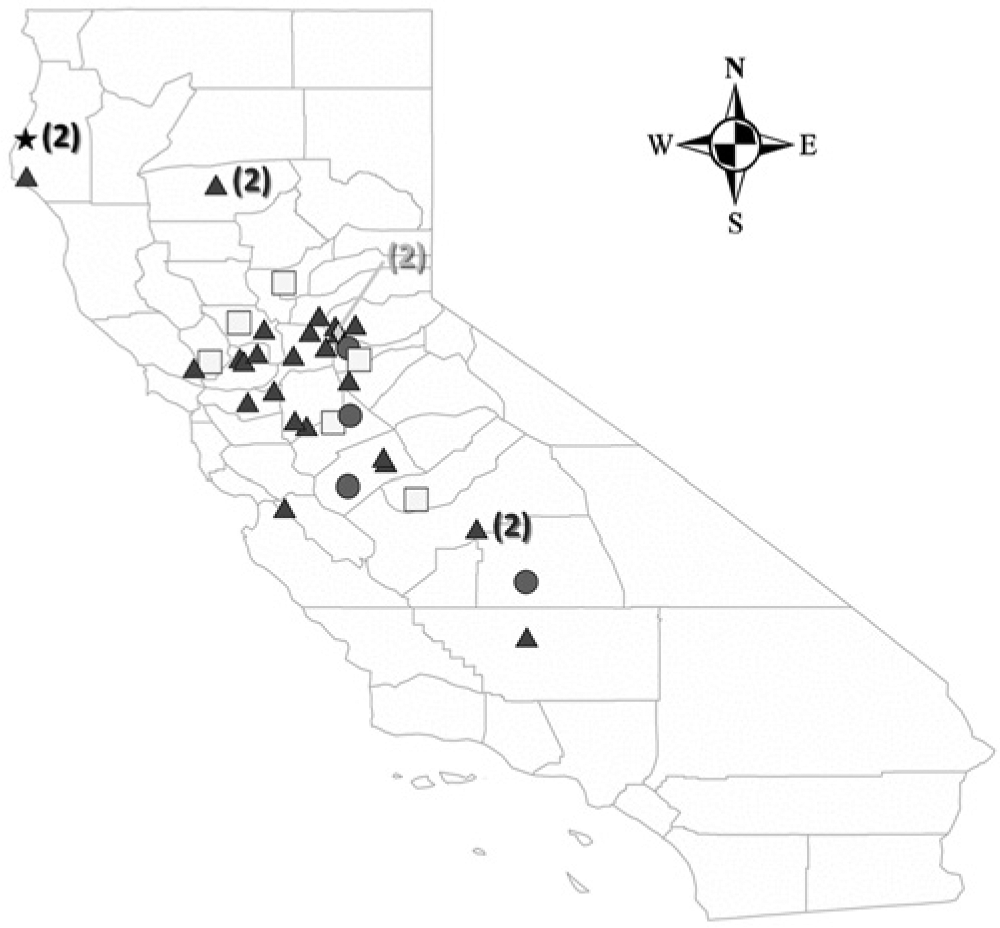
Geographic distribution of 40 cases of Yersinia pseudotuberculosis infection in goats from California. Triangles represent 26 cases of enteritis/typhlocolitis, squares represent 6 cases of abscessation, circles represent 4 cases of abortion, the rhombus represents 2 cases of hepatitis, and the star represents 2 cases of conjunctivitis. One case of abortion and 1 case of enteritis/typhlocolitis from Idaho are not represented in this map. In California, cases were diagnosed in goats from the following counties: Amador, Calaveras, Contra Costa, El Dorado, Fresno, Humboldt, Kern, Madera, Merced, Napa, Placer, Sacramento, San Benito, San Joaquin, Santa Cruz, Solano, Stanislaus, Tehama, Tulare, Yolo, and Yuba. Numbers in parentheses represent number of cases.
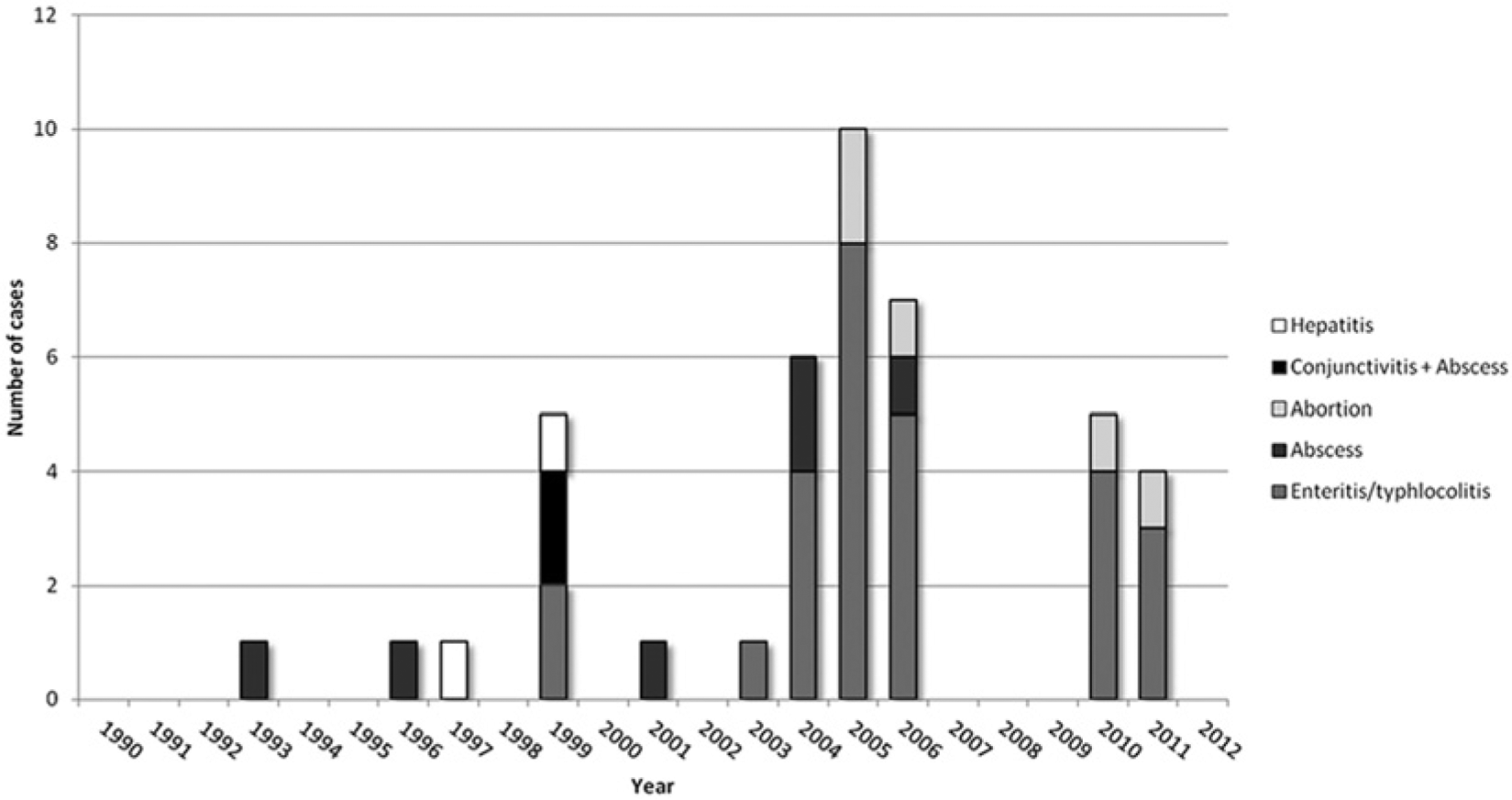
Annual distribution of 42 cases of Yersinia pseudotuberculosis infection in goats by syndrome diagnosed between 1990 and 2012. The disease was diagnosed sporadically in 11 of the 23 years of the series, and 23 of the 42 cases (54.8%) were diagnosed in 3 consecutive years (2004, 2005, and 2006).
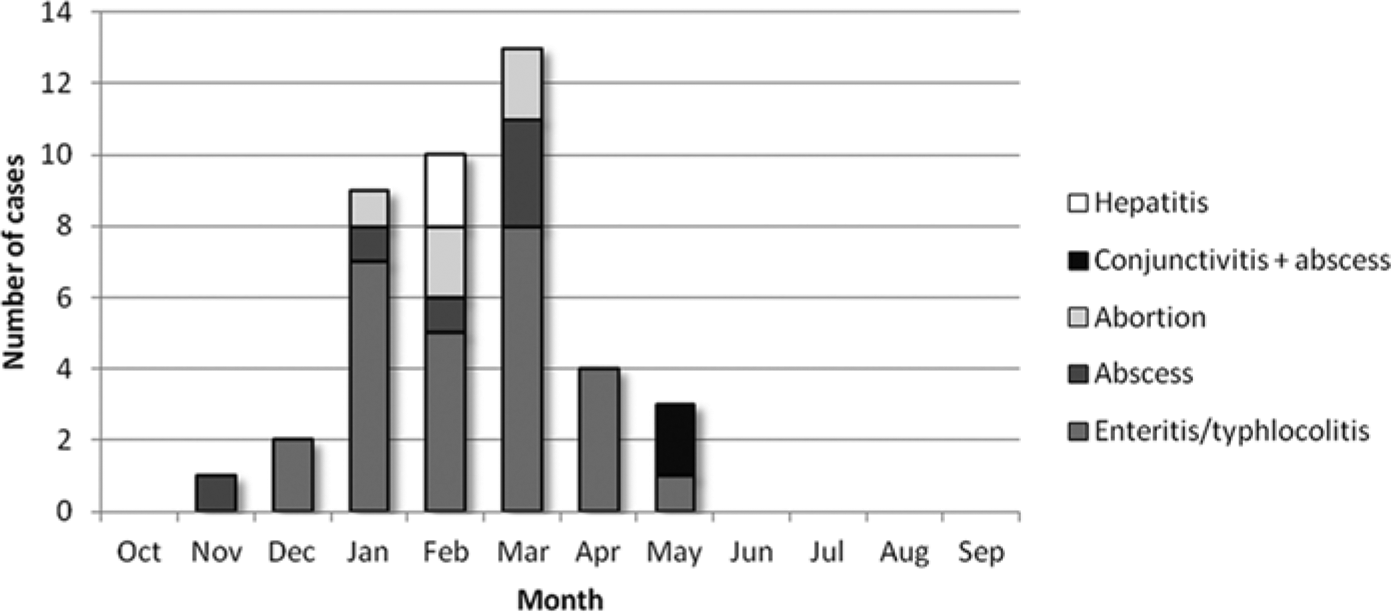
Monthly distribution of the 42 cases of Yersinia pseudotuberculosis in goats by syndrome with marked clustering during winter and spring months.
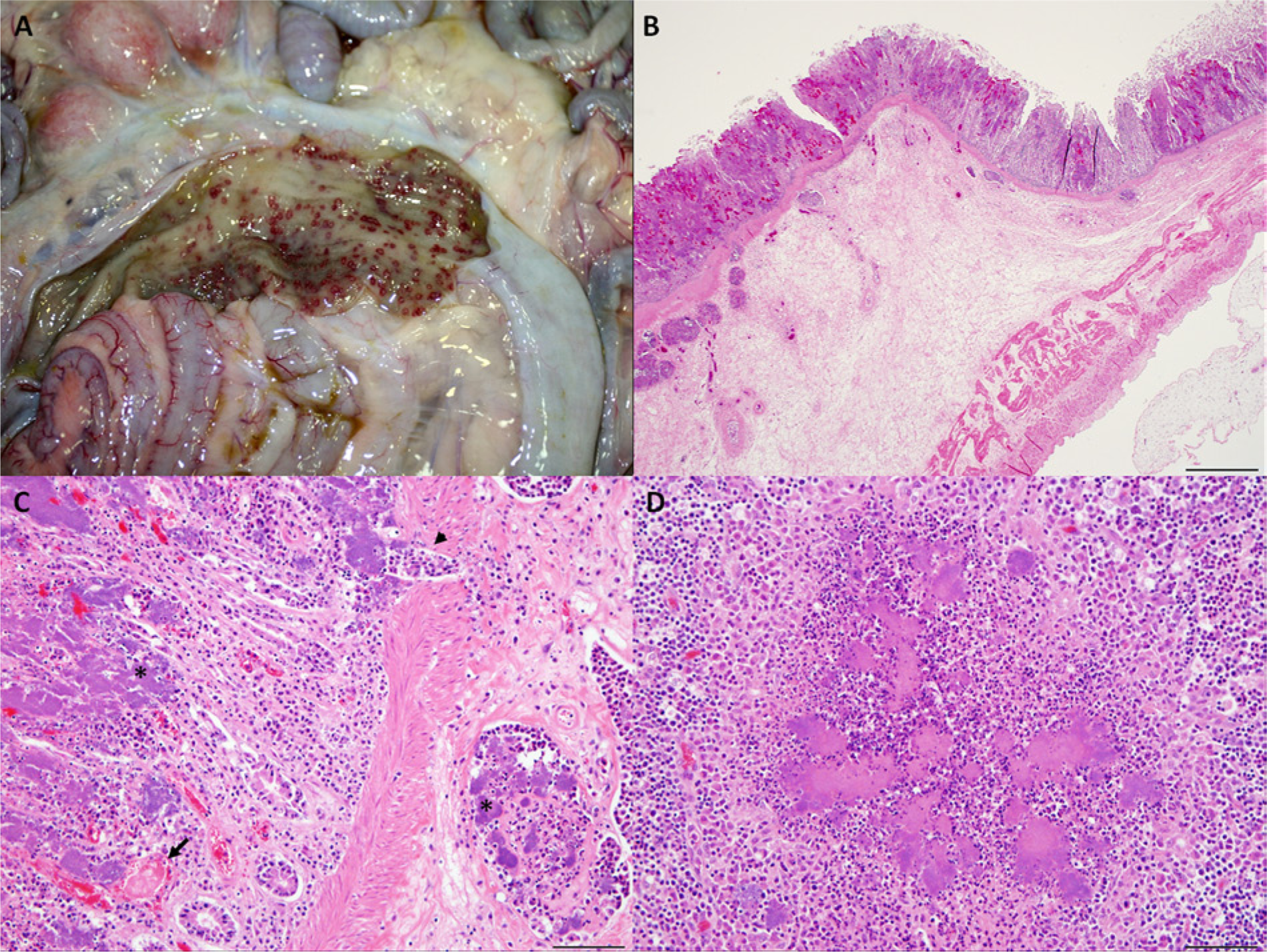
The most frequently diagnosed syndrome was enteritis and/or typhlocolitis with or without mesenteric lymphadenitis or septicemia (27/42 cases, 64.3%; Fig. 4A–4D). All 27 cases of enteritis and/or typhlocolitis occurred in the winter and spring (December–May) with no cases reported in summer or autumn (June–November), showing a marked association with the colder and wetter months in this region. Concomitant gastrointestinal disorders included variable degrees of gastrointestinal trichostrongyle (21/27 cases) and/or coccidia (18/27) infestation, ranging from minimal to severe. The possibility of Salmonella spp. infection was investigated either by specific culture or polymerase chain reaction in feces or intestinal contents in 22 of the 27 cases. Coinfection with Salmonella Eastbourne and Salmonella Manhattan was diagnosed in 2 adult goats, while the remaining 19 cases were negative. Cryptosporidium spp. coinfection was diagnosed in one 5-week-old goat kid. Intestinal and multisystemic coinfection with Trueperella (Arcanobacterium) pyogenes was diagnosed in 2 cases. In 3 cases, Clostridium perfringens enterotoxemia was investigated by a commercially available qualitative capture enzyme-linked immunosorbent assay kit c for the detection of alpha, beta, and epsilon toxin in intestinal contents, with negative results. Extraintestinal diseases included individual cases of Mannheimia haemolytica bronchopneumonia, verminous pneumonia (Muellerius capillaris), and caseous lymphadenitis (Corynebacterium pseudotuberculosis).
Goat.
Thirteen of the 27 cases of enteritis and/or typhlocolitis (48.15%) had normal hepatic levels of selenium or copper, and 12 cases (44.44%) had subnormal or deficient levels of these metals. Combined selenium and copper deficiency was diagnosed only in 1 case, and no data on hepatic levels of selenium and copper was available in 2 cases (7.41%). Enteritis and/or typhlocolitis was diagnosed in similar proportions in animals with normal hepatic levels of copper and selenium, and in animals with subnormal or deficient levels of these metals. In the 6 cases of abscessation, the anatomic location of the abscesses was the subauricular lymph node (1), lumbar vertebral body with extension into the spinal cord (1), submandibular or jaw (2), or widespread with involvement of mesenteric lymph nodes, liver, lungs, and/or brain (2).
The 5 cases of abortion involved 7 fetuses (2 twin pregnancies), all of which were between 3.5 and 5 months of gestational age (late- and full-term abortion). Reported lesions were necrosuppurative placentitis with intralesional bacterial colonies, and bacterial embolization with necrosuppurative fetal pneumonia, hepatitis, and less frequently, splenitis and nephritis.
In 34 of the 42 cases studied (81%), Y. pseudotuberculosis was isolated by regular aerobic culture only, while in 3 cases (7.1%) the microorganism was isolated by Yersinia cold enrichment only (regular aerobic cultures were attempted but the organism was not recovered), and in 5 cases (11.9%) the bacterium was isolated concurrently by both methods. Thus, regular aerobic cultures were sufficient to isolate Y. pseudotuberculosis in 92.9% (39/42) of the cases.
Among other species, the database search retrieved 59 cases of Y. pseudotuberculosis–associated disease in avian species, including psittacines (23), pigeons (6), turacos (5), finches (5), weavers (4), canaries (2), barbets (2), toucans (2), red-crested cardinals (2), a crested screamer, a western grebe, a mousebird, a troupial, a white-tailed jay, a duck, and 2 non-poultry non-psittacine birds of unreported species. Among major mammalian species other than goats, 33 cases were identified. Fourteen cases were diagnosed in cattle (10 cases of enterocolitis, 2 cases of mid- and late-term abortion, and 2 cases of hepatitis/septicemia), 6 in water buffalo (Bubalus bubalis; all of them cases of enteritis), 5 in llamas (Lama glama; 2 cases of enteritis/colitis, 2 cases of septicemia, and 1 case of mesenteric lymphadenitis/mesenteric abscess), and 3 in sheep (2 cases of enterocolitis and 1 late-term abortion). No cases were diagnosed in pigs or horses. Under the category “other mammals”, 1 case each of septicemia was diagnosed in a deer (unreported species), antelope (Oryx spp.; enteritis and septicemia), mountain lion, rabbit, and rat (unreported species).
Discussion
Little information is available on the epidemiology of Y. pseudotuberculosis infections in domestic and wild animals and human beings in the United States. In a study conducted by the Foodborne Disease Active Surveillance Network (FoodNet) from 1996 to 2007, 18 human cases were identified (average annual incidence of 0.04 cases per 1,000,000 persons), 8 (44%) occurred during winter months (December–February), 10 (56%) occurred in western regions of the United States (California or Oregon), and 5 of the 18 (27.78%) were in California. Thirteen (72%) persons with Y. pseudotuberculosis infection required 2–35 days of hospitalization, and 2 deaths were reported (case fatality rate of 11%). 18 In comparison, the annual incidence of Y. enterocolitica infection was much higher (1,355 cases; 0.34 cases per 1,000,000 persons); however, persons with Y. pseudotuberculosis infection were more likely to be hospitalized, have longer hospital stays, die, or have the bacteria isolated from invasive sites (blood, cerebrospinal fluid). 18
Results of the current study show that diseases caused by Y. pseudotuberculosis are endemic among non-poultry birds, and mammals in California. Although the role of goats in the disease epidemiology is still unknown, it is the most frequently reported species with clinical yersiniosis in the CAHFS data set. Animals in both meat and dairy breeds and production classes appear to be similarly affected. The geographic distribution is wide, with cases diagnosed in 21 different counties in northern and central California. The number of cases diagnosed over the total number of goats received diminished with laboratory location in the state from north (Davis) to central (Tulare) California, with no cases diagnosed in southern California (San Bernardino). Although this geographic distribution of cases coincides with the distribution of goat population in California (http://usda01.library.cornell.edu/usda/current/ShpGtInd/ShpGtInd-08-09-2011.pdf), there may also be an association with differences in the climate between these geographic regions (dry weather and warmer winters in southern California), which may interfere with the environmental survival of the bacteria and limit the transmission of the infection. Regarding temporal distribution of cases, 23 of the 42 goat cases (54.8%) occurred in 2004, 2005, and 2006, and peaked in 2005 with 11 cases. All 6 cases in buffalo (1 in 2004, 4 in 2005, and 1 in 2006), 2 of the bovine cases, and 2 of the cases in llamas occurred in the same period (data not shown). It has been proposed that clinical disease and herd outbreaks may be precipitated by stress such as weather conditions, shipping, overcrowding, or starvation. 2 Interestingly, in the autumn of 2004 and winter of 2004–2005, the southwestern United States was affected by a series of Pacific storms that brought extreme and, in some cases, record precipitations in the form of rain and snow to California (http://www.ncdc.noaa.gov/oa/climate/research/2005/california-storms2005.html#records). Rainfalls throughout the state were between 116% and 246% of normal when compared with the historic average between 1971 and 2000 (http://ggweather.com/ca2004rain.htm). The extreme weather conditions registered in this period may have favored the replication of the bacteria in the environment (which can occur at temperatures as low as 4°C) and the transmission of the organism between potentially stressed pastured livestock, resulting in an increased number of cases seen in the period. Surprisingly, in the same winter, a sudden onset of human cases of pseudotuberculosis was registered in France, with 27 cases confirmed nationwide. 31
The different syndromes diagnosed in goats at CAHFS have been described in goats elsewhere.6,24,36 Enteric disease was the most frequently diagnosed syndrome in the data set (64.3% of the cases), and no specific age group was particularly affected. These cases occurred invariably in winter and spring, a seasonal distribution that correlates with what has been previously described in an outbreak of Y. pseudotuberculosis enteritis in dairy goats in Japan, 24 sheep, goats, pigs, cattle, and deer in Australia,14,26,27 and deer in the United States, 37 as well as in human cases in the United States 18 and France. 31
In the current series, 2 cases of Y. pseudotuberculosis–associated conjunctivitis were found. Ocular disease caused by Y. pseudotuberculosis sometimes with concurrent parotid and submandibular lymphadenopathy has been described in goats rarely,19,34,35 and similar lesions occur in human beings as part of the so-called “Parinaud oculoglandular syndrome” caused by the same agent. 34
Yersinia pseudotuberculosis has been associated with mastitis in cattle3,5 and goats,6,15,25 and Y. pseudotuberculosis septicemia and uremic-hemolytic syndrome were reported in a child after ingestion of unpasteurized goat milk. 23 No cases of Y. pseudotuberculosis mastitis were found in the present retrospective search in goats, sheep, or cattle, although the data set mostly included necropsy cases, and it has to be considered that subclinical and nonfatal cases of mastitis can occur without being detected by the search criteria.
In ruminants, copper and selenium deficiencies have been associated with decreased resistance to bacterial infections. 28 Such deficiencies are endemic in California and commonly diagnosed in small ruminants submitted to the CAHFS laboratories. 11 For this reason, the hepatic levels of selenium and copper were evaluated in the goats with enteritis and/or typhlocolitis in the present series, when available. However, in the current study, enteritis and/or typhlocolitis was diagnosed in similar proportions in animals with normal hepatic levels of copper and selenium, and in animals with subnormal/deficient levels of these metals, suggesting that the development of clinical disease caused by Y. pseudotuberculosis in goats occurred independently of the hepatic levels of these elements.
The bacteriologic techniques used to isolate the agent were evaluated, and the results showed that, in 92.9% of the cases, the bacterium was isolated by regular aerobic culture, and no Yersinia cold enrichment was needed. Yersinia pseudotuberculosis can be classified into 6 genetic groups (1–6) with 21 different serotypes (O:1a–1c, 2a–2c, 3, 4a, 4b, 5a, 5b, 6–15), which have been associated with different hosts and geographic regions. Serotypes 1–5 contain strains that are pathogenic to human beings and farm animals. 2 Serotypes 1 and 3 have been associated with enteritis in goats in Australia, 27 and serotype 3 has also been implicated in an outbreak of caprine enteritis in Japan, 24 and an outbreak of colitis and septicemia in farmed deer in the southern United States. 37 Unfortunately, serotyping or genotyping of Y. pseudotuberculosis isolates was not performed in the current case series.
Fifty-nine cases of Y. pseudotuberculosis submitted to CAHFS in the current study period were diagnosed in a variety of avian species. The 4 weavers were submitted from a single zoo aviary in 2012, and one of the pigeons was also submitted from a different zoo (Giannitti, personal observation 2013). These 5 birds were likely in close contact with people. Four outbreaks of Y. pseudotuberculosis septicemia and osteomyelitis in turkeys from central California have been reported and published in 1982 32 ; however, no cases were recorded at CAHFS in turkeys or chickens (either backyard or commercial) in the period studied. Among mammalian species other than goats, 14 cases were diagnosed in cattle, 6 in water buffalo, 5 in llamas, 3 in sheep, and 1 each in an antelope, deer, mountain lion, rabbit, and rat. No cases were diagnosed in pigs or horses. According to this data, nonpoultry avian species and goats are the species in which diseases caused by Y. pseudotuberculosis are most frequently diagnosed in the CAHFS Laboratory System.
Although the current study provides details about clinical and pathologic cases reported in goats, shedding of the agent without clinical presentation in goats and other animal species still needs to be investigated in order to better understand the ecology and epidemiology of the disease. This information would allow a better understanding of the role of reservoirs and hosts species that maintain and transmit the infection, which may or may not be specific to the serotypes present in California. The frequent disease occurrence in goats may be due to actual higher prevalence of infection or increased susceptibility of goats to clinical disease.
In conclusion, the current work contains novel information on Y. pseudotuberculosis infections in goats in particular, and animals in general, in California, and also provides an overview of different syndromes caused by this bacterium in goats. Given the zoonotic potential of this disease and the fact that human cases have been reported in the state in the past decade, results about the geographic and temporal distribution of cases in goats may be useful background for the future diagnosis of the disease in human beings and animals, and a contribution to elucidation of the Y. pseudotuberculosis ecology and epidemiology in California.
Footnotes
Acknowledgements
The authors thank all California Animal Health and Food Safety (CAHFS) Laboratory System case coordinators who had cases of Y. pseudotuberculosis in goats included in this study (J. Adaska, P. Blanchard, C. Farman, B. Johnson, D. Larochelle, A. Mete, R. Moeller, and R. Walker), those who had cases in other mammals (P. Blanchard, C. Farman, L. Heath, B. Johnson, H. Kinde, A. Mete, R. Moeller, J. Ortega, and L. Woods) and birds (G. Casper, R. Chin, G. Cooper, P. Cortes, R. Crespo, P. Duignan, M. Franca, L. Heath, J. Jeffrey, B. Johnson, D. Larochelle, E. Pendleton, G. Rimoldi, G. Senties-Cue, H. L. Shivaprasad, and L. Woods), as well as Lucy Gomes for technical assistance with the retrospective search, and all CAHFS bacteriology technicians.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interests with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
a.
API-20E strip, bioMérieux SA, Mercy l’Etoile, France.
b.
ArcGIS v. 10, ESRI Inc., Redlands, CA.
c.
Bio-X Diagnostics SPRL, Jemelle, Belgium.
