Abstract
A concurrent outbreak of infection with Yersinia pseudotuberculosis occurred in adult captive African lions (Panthera leo). Two 17-y-old male lions and one 14-y-old female lion developed respiratory distress, lethargy, ataxia, and hyporexia. Within 3–5 d of the onset of clinical signs, one male and the female lion died and were submitted for postmortem examination. Macroscopically, the liver and spleen had multifocal-to-coalescing, semi-firm, pale-tan nodules throughout the parenchyma. The lungs were non-collapsed and marked by petechiae. Histologic examination identified lytic, necrosuppurative foci in the liver, spleen, lungs, and kidney, with abundant intralesional gram-negative coccobacilli in the male lion. Similar findings were seen in the female lion in the liver, spleen, kidney, and mesenteric lymph node; however, the intralesional bacterial colonies were more pleomorphic, comprising rod and filamentous morphologies. Aerobic bacterial culture of the liver, spleen, and lung revealed Y. pseudotuberculosis growth. The source of infection is unknown, and an epidemiologic study was performed. Sources to be considered are from the predation of rodent and/or bird reservoirs, or contaminated soil or water. Mortality associated with Y. pseudotuberculosis has been described in an African lion cub, however, to our knowledge, Y. pseudotuberculosis has not been reported in adult African lions, and this is only the second report of Y. pseudotuberculosis with aberrant bacterial morphology observed histologically.
African lions (Panthera leo) are listed as vulnerable by the IUCN 1 given loss of habitat and prey species, human–animal conflict, and illegal trade for traditional medicine. 12 As of 2016, there were 364 African lions housed by 108 Association of Zoos & Aquariums–accredited institutions 12 with an even larger number estimated at conservations centers and private collections within the United States. Knowledge about potential infectious disease threats will not only inform but also enhance health and husbandry practices in these animals under human care. Yersinia pseudotuberculosis is a gram-negative, facultative anaerobic bacteria closely related to, and speculated to have evolved from, the causative agent of plague, Y. pestis. 5 Y. pseudotuberculosis infects a wide variety of animals, including dogs, pigs, ruminants, rodents, foxes, deer, monkeys, kangaroos, and felids.10,11,13 Yersiniosis has been described in domestic cats, 17 cougars,2,13 a Siberian tiger, 14 and an African lion cub. 15 Birds and rodents are considered to be the principal reservoir of Y. pseudotuberculosis; the bacteria are reported to survive up to 20 d in water and 540 d in soil. 10
The route of transmission of Y. pseudotuberculosis is oral, with subsequent bacterial attachment to the intestinal mucosa and selective crossing of the intestinal barrier through attachment to M (microfold) cells with migration to the underlying lymphoid tissue (Peyer patches). 10 Virulence factors that are encoded on the low-calcium response (Lcr) plasmid allow invasion and systemic dissemination including the invasions (Inv) protein, attachment invasion locus (ail), Yersinia outer membrane proteins (Yops), and the O antigen.5,10
In non-domestic and domestic felids, opportunistic predation of wild rodents and birds is the most likely route of infection. 16 Y. pseudotuberculosis infection can occur in acute and subacute-to-chronic forms. 10 Acute yersiniosis is characterized by enterocolitis, fulminating sepsis, and death within 1–3 d. 10 The subacute-to-chronic form of yersiniosis is characterized by weight loss, listlessness, anorexia, severe diarrhea, respiratory distress, and incoordination. 10 We describe here an outbreak of a subacute-to-chronic form of yersiniosis with pleomorphic bacterial morphologies in a group of captive adult African lions.
Two 17-y-old intact male sibling lions and cage mates, developed lethargy, anorexia, and depression, and progressively became dyspneic. One of the lions was found dead after a 5-d clinical course of disease. The cage mate lion eventually recovered uneventfully. Approximately 1 wk later, a 14-y-old, female lion at the same conservatory institution, housed in an adjacent enclosure, developed acute lethargy and anorexia. Despite being treated with a single dose of cefovecin (8 mg/kg SQ) and supportive treatment, clinical signs worsened, the female lion became obtunded, and died 4 d later. Both lions were sent for postmortem examination.
Macroscopically, the male lion had moderately decreased adipose stores (body condition score [BCS]: 2 of 9) and muscling was moderately decreased but symmetrical (muscle mass index [MMI]: 1 of 3). The lungs failed to collapse and were diffusely mottled dark to light-red, heavy, and wet, with multifocal-to-coalescing petechiae and ecchymoses on the pleural surface and throughout the parenchyma (Fig. 1A). Scattered throughout all liver lobes were many, random, pinpoint to 0.2-cm diameter, tan-to-white, spherical, caseous nodules. Within the right lateral lobe of the liver, there was a focal, multilobulated, thin-walled cystic structure of 1.5 × 0.5 × 0.3 cm, considered to be an incidental biliary cyst. There were endocardial hemorrhages.
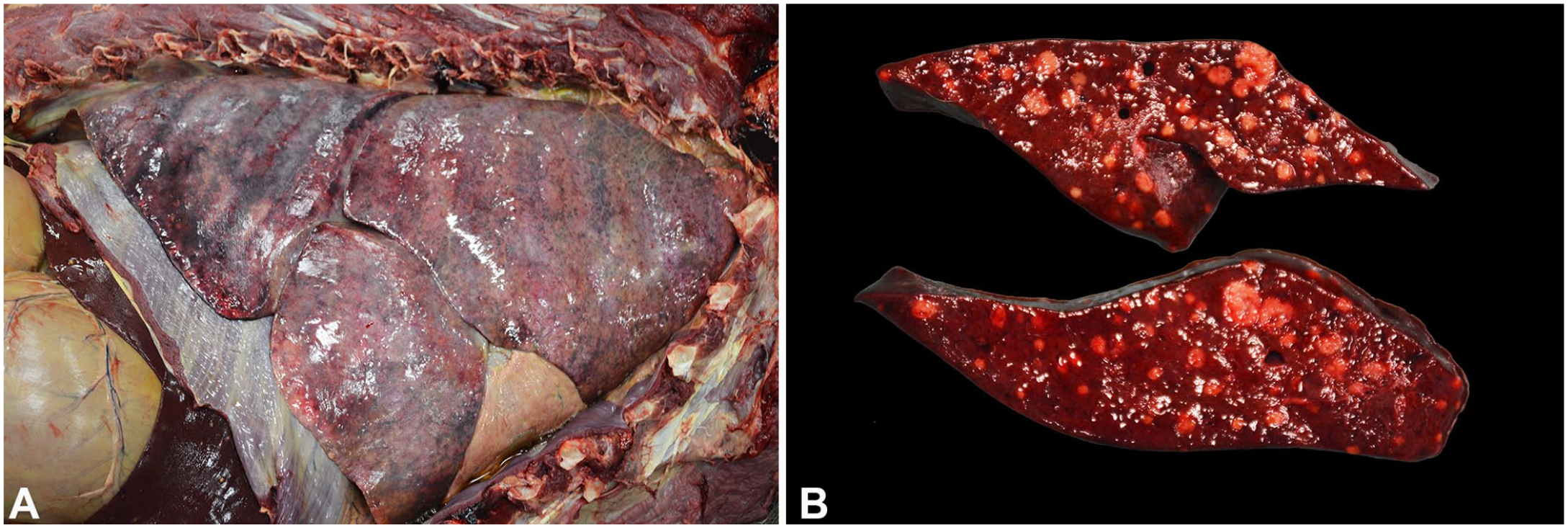
Yersinia pseudotuberculosis infection in African lions in human care.
The female lion had adequate adipose stores and muscling (BCS: 5 of 9; MMI: 3 of 3). The peritoneal cavity contained ~1 L of dark-red to brown, watery fluid mixed with fibrin strands. Similar to the male lion, there were many, pale-tan to white, caseous nodules of up to 0.8-cm diameter within the liver parenchyma (Fig. 1B), as well as moderate, diffuse interstitial pneumonia with petechiation. The spleen had 2 firm, expansile masses ~2.0 and 1.5 cm in diameter, respectively, which were mottled pale-tan to dark-red on cut surface. Bilaterally, the renal cortical parenchyma had pale-tan, soft nodules of ~0.3-cm diameter that bulged on cut surface. The renal medullary parenchyma had dark-red, wedge-shaped discolored foci.
Tissue samples were fixed in 10% neutral-buffered formalin and processed routinely for histologic examination. Samples of the liver (both lions), spleen, and lung (male lion) were submitted for aerobic bacterial culture. Swabs of the right and left nasal turbinates were submitted for SARS-CoV-2 (COVID-19) real-time PCR (rtPCR) testing to the NC State Microbiology and Molecular Diagnostics Laboratory (Raleigh, NC, USA) because of the presence of pneumonia.
Microscopically, both lions had similar changes. Within the liver (2 of 2), spleen (2 of 2), kidney (2 of 2), lung (1 of 2), and mesenteric lymph node (1 of 2), random, necrosuppurative foci were composed of lytic necrosis, karyorrhectic debris, and fibrin exudate mixed with viable and degenerate neutrophils, lymphocytes, macrophages, and plasma cells (Fig. 2A–C). Moderate numbers of intralesional aggregates of gram-negative coccobacilli were frequent in the male lion (Fig. 2A–F). Surprisingly, the intralesional bacterial colonies observed in the female lion had marked pleomorphism, varying from long chains of large, rod-shaped bacteria to filamentous strands, as well as clouds of coccobacilli (Fig. 2G–I). Additionally, fibrin thrombi were present in the spleen and kidney (1 of 2). The medullary sinuses of mesenteric lymph nodes were flooded by hemorrhage, fibrin, foamy histiocytes, neutrophils, and similar pleomorphic bacteria (1 of 2). Cortical renal tubules had severe acute multifocal coagulative necrosis (1 of 2).
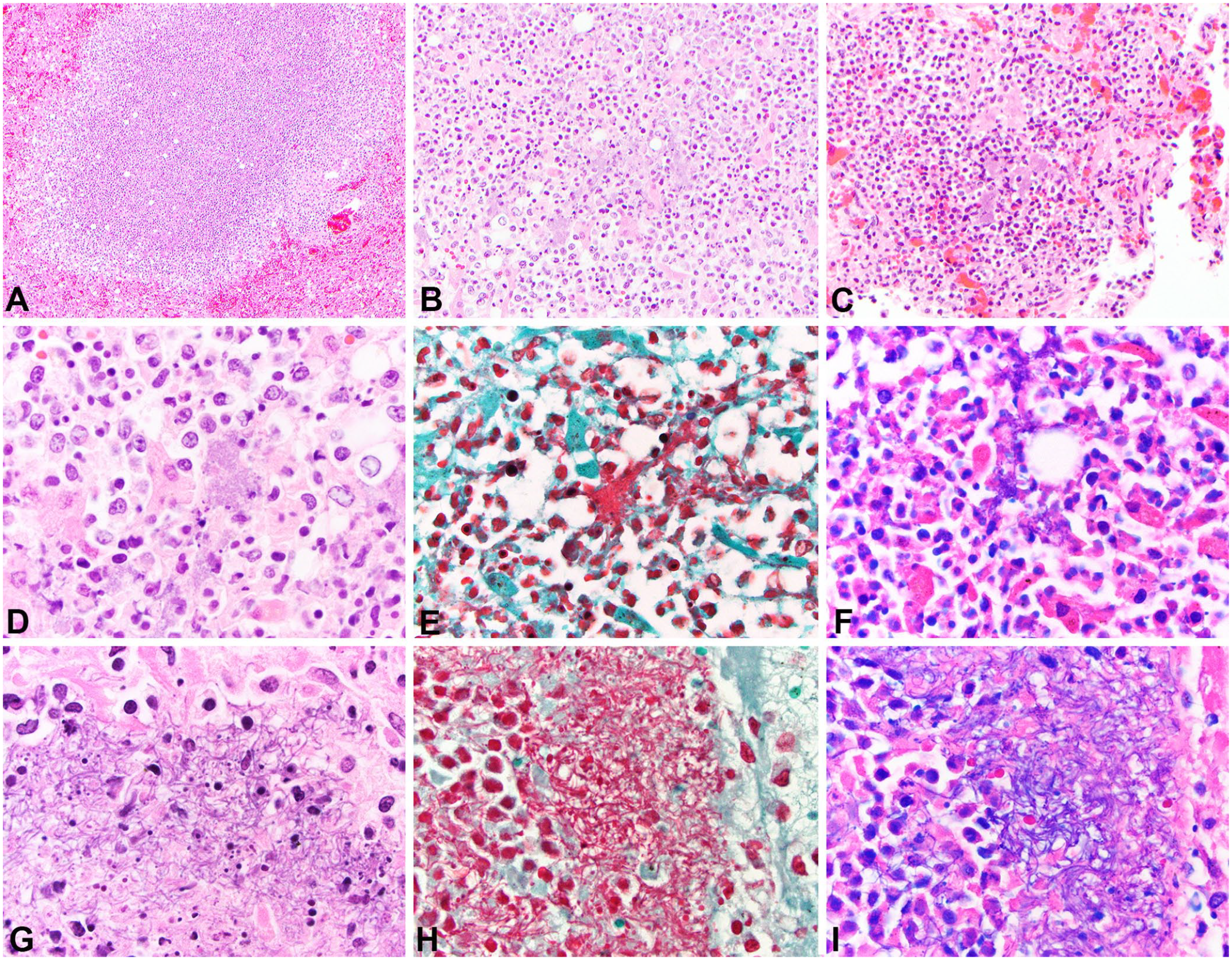
Histologic findings associated with infection with Yersinia pseudotuberculosis in African lions in human care.
Within the lesser affected areas of the pulmonary parenchyma (2 of 2), the alveolar spaces were multifocally expanded and filled by edema, acute hemorrhage, and fibrin mixed with low numbers of neutrophils and rare macrophages. The alveolar septa of the lungs were markedly expanded by mature fibrous connective tissue with mild-to-moderate infiltration by histiocytes, lymphocytes, plasma cells, and fewer neutrophils (1 of 2). The tracheobronchial lymph nodes had moderate follicular lymphoid hyperplasia with prominent germinal centers (reactive lymph nodes). The lamina propria and submucosa of the nasal turbinates were multifocally expanded by edema and low numbers of lymphocytes and plasma cells (1 of 2). The lamina propria of the small intestines was mildly to moderately expanded by mature fibrous tissue at the crypt-villus junction (1 of 2).
Aerobic bacterial culture of the liver (2 of 2), spleen (1 of 1), and lung (1 of 1) grew 3+ to 4+ (liver, spleen) and 1+ (lung) Y. pseudotuberculosis. Additionally, Aeromonas spp. (+1) was identified in lung of the male lion. Special stains were applied to sections of liver and spleen including Gram and Giemsa stains. Giemsa stain is routinely used to highlight Yersinia spp. when Wayson stain is not available. 18 Bacteria observed in histologic sections were gram-negative and Giemsa-positive (Fig. 2D–F, G–I). Coccobacillary bacterial morphology was observed in the male lion whereas the bacterial morphology varied tremendously in the female lion from large aggregates of coccobacilli to large rod-shaped bacteria and filaments (Fig. 2G–I).
SARS-CoV-2 (COVID-19) rtPCR tests of the nasal turbinates were negative for both lions. Immunohistochemical tests for canine distemper virus (Canine morbillivirus; Biocare Reveal Decloaker 20 min, 3% H2O2 10 min; 1:2,000 canine distemper virus monoclonal; Custom Monoclonals International) and feline infectious peritonitis virus (Alphacoronavirus 1; citrate buffer antigen retrieval pH 6.0 at 120°C for 30 s, 1:300; Biocare Medical) in the male lion lung were both negative. Appropriate positive and negative controls were applied. The status of infection by feline immunodeficiency virus in this captive felid population is unknown.
Based on the gross and microscopic findings combined with the aerobic culture results, a diagnosis of disseminated Y. pseudotuberculosis infection leading to sepsis was considered as the cause of death in both lions. Given an unknown route of exposure for these animals, an epidemiologic investigation was conducted to evaluate potential sources of infection, including wild rodents, diet, and environment. 3
In other species, Y. pseudotuberculosis typically causes primary lesions within the gastrointestinal tract including necrotizing enteritis with invasion into Peyer patches and mesenteric lymph nodes before systemic dissemination, particularly to the spleen, liver, and lung. 12 Interestingly, in the cases reported here, the small and large intestines were histologically unremarkable except for multifocal, mild-to-moderate fibrosis of the lamina propria, and mild crypt necrosis. In other reports of domestic and exotic felids diagnosed with yersiniosis, the intestine was likewise unaffected; therefore, it is possible that the intestine may not be a targeted organ in felids.2,13–15 Additionally, different disease courses of yersiniosis including acute, chronic, and subclinical presentations are described. 10 We speculate that our cases represented a subacute-to-chronic form of infection in which intestinal lesions may have already healed and had become unremarkable on macroscopic and microscopic examination. Histologic findings in the female lion, including moderate interstitial fibrosis in the lungs and mild fibrosis of the lamina propria of the intestine, may provide evidence of previous injury or inflammation in these organs. However, in a study of domestic cats, fibrosis of the lamina propria of the mucosa of the intestine, mainly located at the border between crypts and villi, as seen in our case, was speculated to be associated with chronic inflammation such as inflammatory bowel disease, although, in our cases, there was no evidence of active intestinal inflammation nor reported clinical signs suggestive of inflammatory bowel disease. 6
Although both lions had Y. pseudotuberculosis cultured from the liver, the bacteria observed histologically varied in morphology between the male and female lions. The bacteria observed in tissue sections from the male lion had a more typical and classical morphology for Y. pseudotuberculosis, namely clouds of small coccobacilli. However, the bacterial colonies observed in the female lion had a more filamentous or rod-shaped chain-like morphology admixed with more typical clouds of small coccobacilli. We highly suspect that the bacterial aggregates observed in the female lion represent one of the aberrant forms of Y. pseudotuberculosis described in squirrel monkeys. 11 In humans, pleomorphism, including filamentous shape changes, has been described in Klebsiella pneumoniae 7 and Escherichia coli 8 infections following treatment with beta-lactam antibiotics. Antibiotic administration may explain the different bacterial morphologies in our 2 cases, given that the female lion was treated with cefovecin, a beta-lactam antibiotic, a few days prior to death, whereas the male lion was not treated with any antibiotics. Beta-lactam antibiotics can alter the bacterial cell envelope by attaching to penicillin-binding proteins (PBPs). 11 PBPs are attached to bacterial cytoplasmic membranes and are associated with the synthesis of peptidoglycans. 11 Therefore, theoretically, attachment of beta-lactam antibiotics to PBPs could alter cell shape and bacterial morphology, as potentially seen in our case. 11
Aerobic culture of the lungs of the male lion also revealed 1+ growth of A. salmonicida subsp. salmonicida, a gram-negative bacillus that commonly causes disease in wild and farmed fish. Although anecdotal evidence may suggest that Aeromonas spp. can act as a low-grade pathogen in mammals, A. salmonicida has yet to be reported as a potential cause of disease in mammals. 9 Therefore, we consider this bacterium from the male lion lung to be a contaminant.
The kidneys of the female lion had regional peracute-to-acute cortical tubule coagulative necrosis. Acute tubular injury is caused most commonly by ischemia or nephrotoxins. 4 In our case, ischemia is the most likely cause, secondary to sepsis or systemic inflammation, with renal thrombi causing regional infarction. The endocardial petechiae and ecchymoses in the male lion may be a perimortem or agonal change, or may have occurred secondary to sepsis and inflammatory vascular damage.
Y. pseudotuberculosis infection should be considered as a differential diagnosis in African lions and other exotic felids with nonspecific clinical signs and random necrosuppurative nodules in the liver and spleen on postmortem examination. If such findings are observed, appropriate personal protective equipment should be employed to prevent zoonotic transmission. Additionally, Y. pseudotuberculosis should be considered even if the bacteria exhibit filamentous pleomorphism histologically, especially if the animal had been treated with an antibiotic.
Footnotes
Acknowledgements
We acknowledge the contributions of the technical staff of the North Carolina State University Autopsy and Histology Laboratories, and Dr. Leigh Duke for her clinical assistance. We thank Hannah Fullmer and Mindy Stinner and the rest of the staff at the Animal Park at the Conservators Center for their assistance with these cases.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
