Abstract
Two meerkats (Suricata suricatta) housed in the same zoological garden in Japan died due to Yersinia pseudotuberculosis serotype 4b infection. Gross and microscopic lesions included necrotizing enteritis and enlargement of the spleen and liver with multifocal necrosis. Inflammatory cells, primarily neutrophils, and nuclear debris were associated with clusters of Gram-negative bacilli. Additionally, there were aberrant organism forms that were larger than bacilli and appeared as basophilic globular bodies. Immunohistochemical examination showed that the bacilli and globular bodies were strongly positive for Y. pseudotuberculosis O4 antigen. The globular bodies were considered a shape-changed form of Y. pseudotuberculosis, and these morphologically abnormal bacteria could present a diagnostic challenge.
Yersiniosis due to Yersinia pseudotuberculosis is an important zoonotic disease with a global distribution. 6 Typically, the bacteria are acquired orally through the ingestion of food and water contaminated with infectious feces from free-living rodents and birds. 12 Yersinia pseudotuberculosis invades the lymphoid follicles of the Peyer’s patches of the small intestine resulting in enteritis, mesenteric lymphadenitis, and occasionally septicemia in humans and other animals.4,12 Stress factors such as a cold environment, poor nutrition, immunosuppression, young age, and transport can predispose to yersiniosis. 14 In zoological gardens, yersiniosis due to Y. pseudotuberculosis has been reported in mammals belonging to the orders Artiodactyla, Rodentia, Lagomorpha, and Chiroptera, nonhuman primates, and birds.1,2,18,20 Nonhuman primates in particular are highly susceptible to Y. pseudotuberculosis infection.3,9,19 Among species in the order Carnivora, only a few sporadic cases have been reported.7,15 Dogs and cats may be asymptomatic carriers of this pathogen. 7 In this report, we describe 2 cases of yersiniosis due to Y. pseudotuberculosis in captive meerkats (Suricata suricatta) in Japan.
In April 2007, a 7-year-old female meerkat (meerkat 1), 1 of 3 meerkats housed together in a Japanese zoological garden, was found in a languid state. Her appetite was depressed and feces were soft. Enrofloxacin a was administered orally (10 mg/kg) the following day but the animal died the next morning. The remaining 2 meerkats appeared clinically healthy. In February 2008, a 9-year-old female meerkat (meerkat 2), 1 of 2 meerkats housed together and one of the animals previously housed with meerkat 1, was found in a moribund state. The cephalosporin antibiotic, cefoperazone, b was administered intravenously (20 mg/kg) twice that day but she died the next morning. A complete autopsy was performed on the 2 meerkats.
Samples of visceral tissues were fixed in 10% neutral phosphate buffered formalin and embedded in paraffin wax by standard procedures for histological and immunohistochemical analysis. Sections were cut at ~4-μm thickness and stained with hematoxylin and eosin and Gram stain. Immunohistochemical analysis was performed using a commercial rabbit anti–Y. pseudotuberculosis sera set (O1, O2, O3, O4, O5, and O6). c Bacteriological examination of the liver, spleen, and rectal swabs from the 2 meerkats were performed as previously described. 9 Briefly, the samples collected from the dead meerkats were plated on CIN (cefsulodin-irgasan-novobiocin) agar plates (Yersinia selective agar base and Yersinia selective supplement) d and incubated at 25°C for 48 hr. Colonies morphologically similar to those of Yersinia spp. were subcultured on trypticase soy agar. e Biochemical characteristics were examined on triple sugar iron medium f and lysine indole motility medium. g The isolates identified as Y. pseudotuberculosis were examined for virulence-associated characteristics by autoagglutination at 37°C by using tryptic soy broth. e Serotyping of Y. pseudotuberculosis was performed by slide agglutination with a commercial rabbit anti–Y. pseudotuberculosis sera set. c
Gross lesions were similar in each of the 2 meerkats, but were more severe in meerkat 2. The liver and spleen were enlarged and friable and had multifocal, 1–3 mm in diameter, yellow-to-white nodules. Similar nodules as well as occasional petechial and ecchymotic hemorrhages were scattered throughout the colonic and cecal mucosa. Intestinal contents were brown to red and mildly watery.
Microscopically, the nodules in the liver and spleen were characterized by multiple foci of necrosis that contained central or peripheral clusters of Gram-negative bacilli, neutrophils, and occasional lymphocytes and macrophages (Fig. 1). In the liver, foci of necrosis were randomly scattered throughout the parenchyma. Additionally, basophilic Gram-negative globular bodies were within necrotic foci in the liver and spleen of meerkat 2 (Fig. 2). The globular bodies were round to oval, ~4–5 μm in diameter, and occasionally contained basophilic fine granular material. There were dozens of globular bodies in the liver and fewer in the spleen. The mucosa of the small intestine, cecum, and colon contained foci of necrosis with clusters of Gram-negative bacilli, desquamation of the mucosal epithelium, and hemorrhage. In the small intestine, areas of necrosis and hemorrhage with intralesional Gram-negative bacilli were most severe within the Peyer’s patches. Gram-negative bacilli in the liver, spleen, and intestine of both animals, and globular bodies in the liver and spleen of meerkat 2, were strongly positive by immunohistochemical staining using anti–Y. pseudotuberculosis serogroup O4 sera (Figs. 3, 4). Yersinia pseudotuberculosis serotype 4b was isolated from the livers, spleens, and rectal swabs of the 2 meerkats. No other bacteria were isolated.
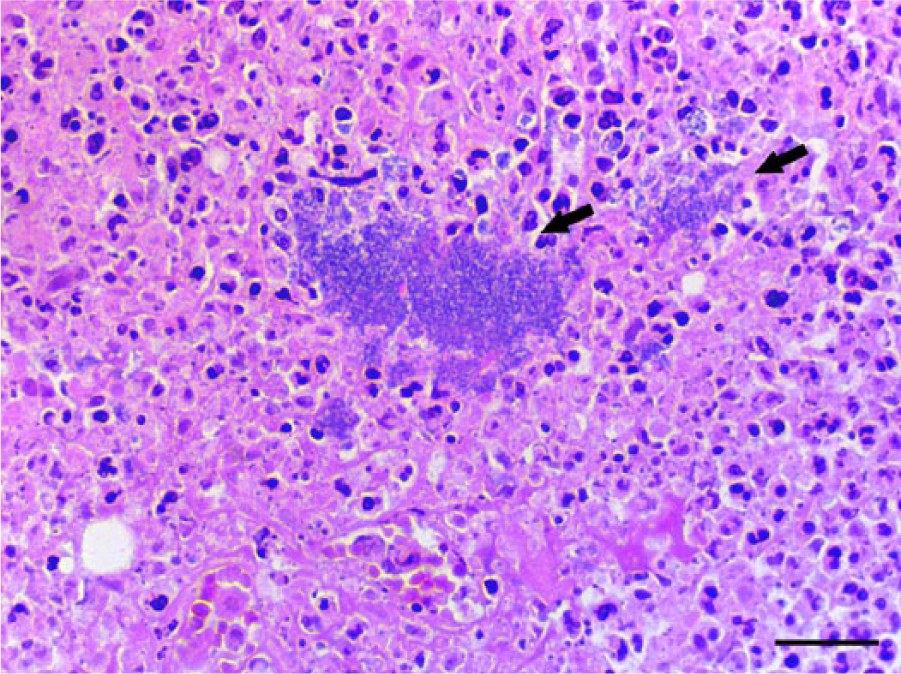
Liver; meerkat 1. Focal hepatocellular necrosis with central bacilli (arrows). Hematoxylin and eosin. Bar = 25 μm.
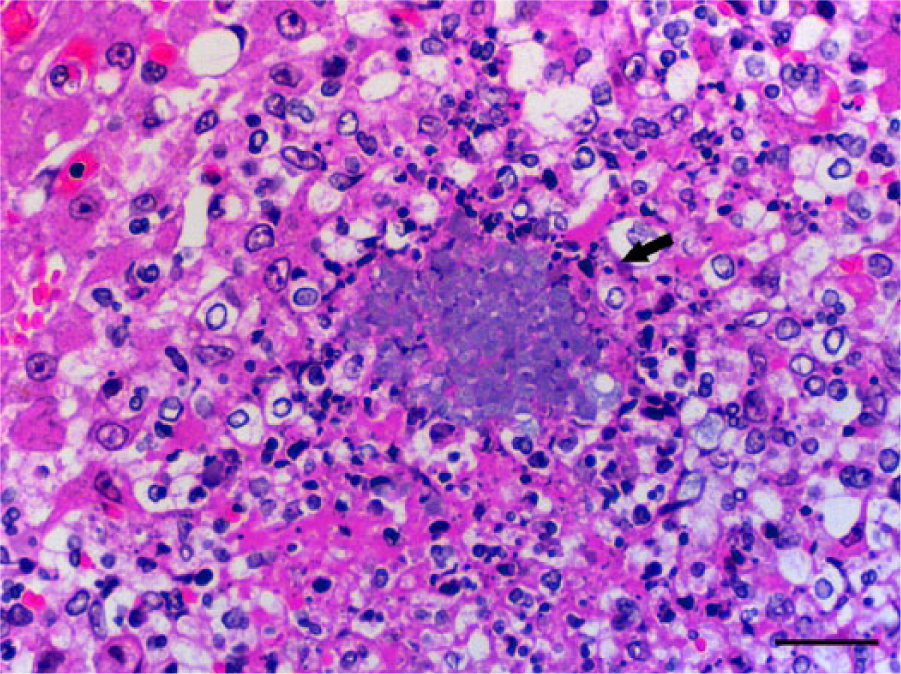
Liver; meerkat 2. Focal hepatocellular necrosis with central basophilic globular bodies (arrow). Note that the shape and size of the bacterial colonies are different from those in the liver of meerkat 1. Hematoxylin and eosin. Bar = 25 μm.
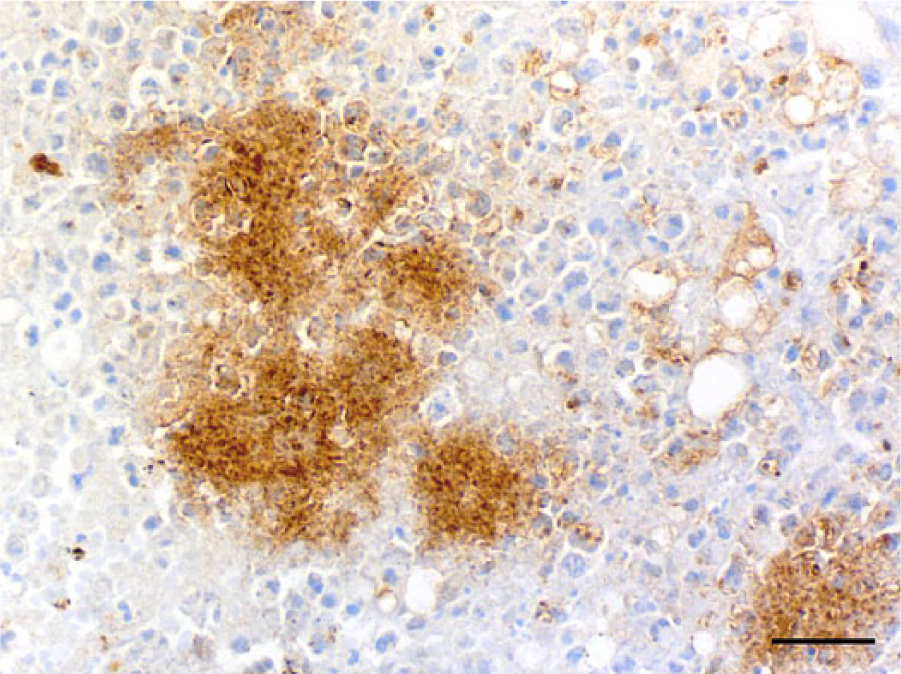
Liver; meerkat 1. Bacterial clusters showing strong positive reaction to immunoperoxidase staining with anti–Yersinia pseudotuberculosis O4 sera. Immunohistochemistry, counterstained with hematoxylin. Bar = 25 μm.
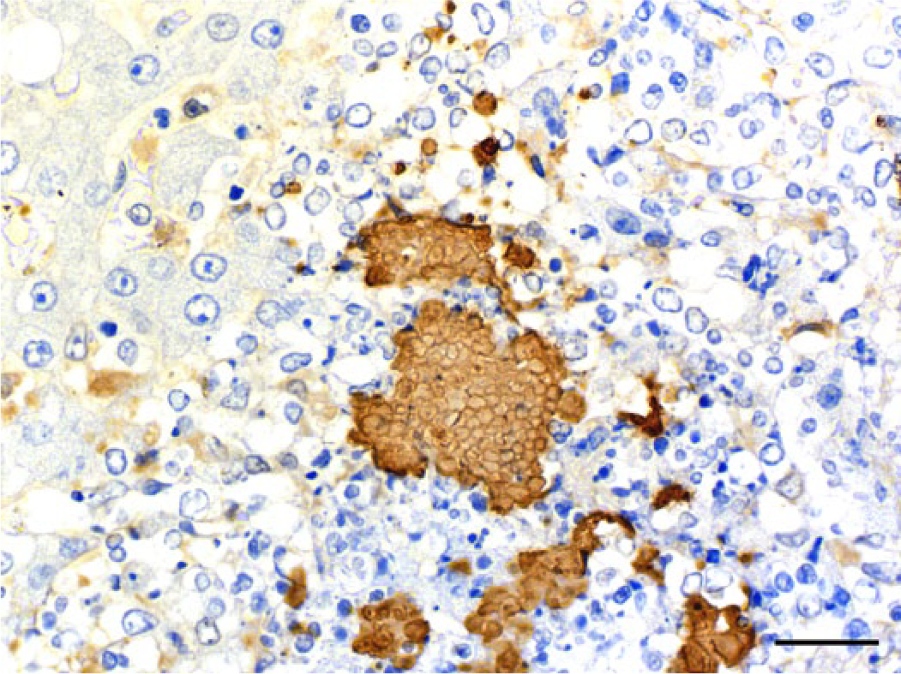
Liver; meerkat 2. Globular bodies showing strong positive reaction to immunoperoxidase staining with anti–Yersinia pseudotuberculosis O4 sera. Immunohistochemistry, counterstained with hematoxylin. Bar = 25 μm.
Both meerkats had necrotizing enteritis and enlarged spleen and liver accompanied by multifocal necrosis with intralesional Gram-negative bacilli. These lesions are typical of those seen in other animals that have died of yersiniosis.2,8 A diagnosis of yersiniosis was confirmed by immunohistochemical detection of Y. pseudotuberculosis serogroup O4 antigen and isolation of Y. pseudotuberculosis serotype 4b in bacterial culture from affected tissue samples and rectal swabs. Based on these findings, yersiniosis due to Y. pseudotuberculosis serotype 4b was diagnosed in these 2 meerkats, and death was attributed to sepsis. Yersinia pseudotuberculosis serogroup O4 has been divided into 2 serotypes, 4a and 4b, with Y. pseudotuberculosis 4b being the most common serotype isolated from the humans and other animals in Japan. 6
The globular bodies in the liver and spleen of meerkat 2 were considered to be shape-changed forms of Y. pseudotuberculosis based on the results of Gram stain, immunohistochemistry, and their location in the necrotic lesions. Similar findings have been reported in squirrel monkeys (Saimiri sciureus) in which shape-changed Y. pseudotuberculosis organisms appeared as spheroplasts and filaments in the spleen, liver, and intestines by using transmission electron microscopy and light microscopy. 13 The globular bodies in the meerkat were similar to the spheroplasts described in the squirrel monkeys. Shape-changed bacilli have often been observed in clinical specimens such as blood, sputum, and urine from human patients receiving antibiotics and are attributed to the pharmacological actions of antibiotics on the bacterial cell wall.10,11 The presence of abnormal forms of bacteria are indicative of inadequate antibiotic concentrations at the site of infection.10,11 Shape-changed bacteria may result in a misdiagnosis or cause a misleading negative bacterial culture result due to bacterial cell wall destruction. 11
The 2 meerkats in the present study died from necrotizing enteritis, hepatitis, and splenitis, caused by alimentary Y. pseudotuberculosis infection, but the source(s) of the infections was unclear. In this zoological garden, yersiniosis caused by infection with Y. pseudotuberculosis has occurred sporadically in squirrel monkeys. In December 2003, 2 young squirrel monkeys died from infection caused by Y. pseudotuberculosis 4b, and in August 2008, a young squirrel monkey died from infection caused by Y. pseudotuberculosis (serotype unconfirmed). It is suspected that carrier animals such as free-ranging rodents or birds may play a role in the transmission of Y. pseudotuberculosis. Although it is possible that there are other captive zoo inhabitants that may serve as subclinical carriers and allow contamination of feed, water, or the environment, or fomite-associated spread, this is considered unlikely because human traffic in the meerkat cages is restricted, and stringent disinfection controls are in place. Cold stress might have contributed to the occurrence of yersiniosis as meerkats 1 and 2 died in April and February, respectively. Additionally, Y. pseudotuberculosis grows better in cold conditions. 17 It is possible that the administration of 2 doses of intravenous antibiotics accelerated the death of meerkat 2 by rapid destruction of the bacterial cell walls. This possibility should be considered in future investigations.
Reports of yersiniosis due to Y. pseudotuberculosis in carnivores, including meerkats, are rare, except for a few cases in domestic and exotic cats.7,15 Mycobacteriosis, another important zoonotic disease, was recently reported in free-ranging and zoo meerkats.5,16 From public health and treatment perspectives, it is important to differentiate Yersinia spp. from other organisms using immunohistochemistry and bacterial identification.
Footnotes
Authors’ contributions
S Nakamura and Y Une substantially contributed to conception and design of the study, and drafted the manuscript. S Nakamura contributed to acquisition, analysis, and interpretation of data. S Suzuki contributed to acquisition of data. A Yonezawa contributed to acquisition and analysis of data. H Hayashidani contributed to analysis of data. H Hayashidani, A Yonezawa, and I Suzuki critically revised the manuscript. All authors gave final approval, and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Baytril, Bayer Yakuhin, Osaka, Japan.
b.
Cefobid, Pfizer Japan, Tokyo, Japan.
c.
Denka-Seiken, Tokyo, Japan.
d.
Oxoid, Basingstoke, UK.
e.
BD, Franklin Lakes, NJ.
f.
Eiken Chemical, Tokyo, Japan.
g.
Nissui Pharmaceutical, Tokyo, Japan.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
