Abstract
Sudden death of 9 deer occurred in a large enclosed deer farm with approximately 400 heads of cervids. Fatal yersiniosis was diagnosed in 2 deer that were submitted for laboratory diagnosis. Histopathologically, the disease was characterized by multifocal pulmonary hemorrhage and mild interstitial pneumonia, marked diffuse cholangiohepatitis, minimal myocarditis with mild myocardial degeneration, and mild multifocal suppurative cystic colitis. Yersinia pseudotuberculosis was isolated from the lungs and colon of the affected animals. The isolates were PCR-positive for genes virF, inv, yopB, and yopH, which are essential for invasion and colonization of host intestine and lung. The isolates reacted with polyclonal antibodies against serotype O:3 antigen. The O-genotyping patterns of the isolates were identical with each other, but different from those of the 21 O-genotypes (or serotypes) reported previously. In addition to the O-antigen genes possessed by classical serotype O:3, a gene (wbyK) encoding a mannosyltransferase-like protein was detected in these isolates. The wbyK gene of the isolates showed 94% of DNA sequence homology with the wbyK gene harbored by Y. pseudotuberculosis O:1b. On the basis of pathology, bacteriology, and serology, the authors concluded that the acute deaths of these deer were caused by Y. pseudotuberculosis infection. Molecular characterization of the isolate revealed a genetic heterogeneity in the O-antigen gene cluster of Y. pseudotuberculosis serotype O:3.
Yersinia pseudotuberculosis, a member of the family Enterobacteriaceae, infects a variety of avian, mammalian, and reptilian species. 3,5,7,13,14,16 The organism is ubiquitous and carried asymptomatically by a range of animal species, such as wild birds and rodents. 9 Infections are usually sporadic, but outbreaks causing high mortality in captive or farmed cervids have been documented. 16 The prevalence of yersiniosis increases in winter in part because of the cold-stress for animals, as well as the organism's enhanced ability to grow in cold temperature. 11 The clinical symptoms and pathologic changes associated with Y. pseudotuberculosis infection vary among different host species. The common manifestations of the disease in primates, cervids, and other mammals are necrotizing, ulcerative enteritis and mesenteric lymphadenitis. 3,5,7,16 Septicemia can lead to the involvement of visceral organs, especially liver and spleen. 11–13
The pathogenic Yersinia isolates harbor a 70- to 75-kb virulence plasmid that encodes a type 3 secretion system (TTSS) and a number of secreted proteins called Yops (Yersinia outer proteins), whose expression is regulated by VirF, a transcriptional activator. 8,18 Among the Yops translocated by the TTSS, YopH and YopB are important for lung colonization and dissemination. 8,19 The chromosomally encoded Inv product enables the organism to invade Peyer's patch M-cells. 9,19 Genotyping on the basis of these virulence-associated genes has been used to predict the phenotypes of field isolates. Yersinia pseudotuberculosis strains can be divided into 21 different O-serotypes or O-genotypes on the basis of the difference in the lipopolysaccharide (LPS) O side chain (O-antigen) or in the O-antigen gene cluster. 4,18 O:1 to O:3 are frequently associated with enteric infections in farmed deer. 3,16 In this study, an outbreak of fatal yersiniosis in farmed deer caused by infections with a Y. pseudotuberculosis O:3 isolate that harbors a wbyK gene in its O-antigen gene cluster is reported. To the authors' knowledge, this is the first attempt to characterize the O-antigen genes of Y. pseudotuberculosis isolated from deer in North America.
Between December 2006 and February 2007, 2 white-tailed deer (Odocoileus virginianus), 4 Axis deer (Cervus axis), 1 Sika (Cervus nippon), and 2 Black Bucks (Antilope cervicapra) were found dead in an enclosed deer farm with approximately 400 head of cervids. All affected animals were less than 8 months old, and deaths occurred with no prior history of illness. A blood sample was taken from a 6-month-old white-tailed deer exhibiting lethargy and depression. The white-tailed deer was subsequently euthanized by the attending veterinarian. Complete postmortem examinations of the white-tailed deer (animal no. 1) and an Axis deer (animal no. 2) that had been dead for about 24 hr were performed by the attending veterinarian. No significant gross abnormalities or pathologic changes, other than hypothermia and the presence of ticks (Ixodes scapularis) on the carcasses, were seen. Specimens, including 10 ml of serum; fresh and formalin-fixed tissues of heart, liver, lung, and colon of animal no. 1; and fresh and formalin-fixed lung tissues of animal no. 2 were submitted to the Mississippi Veterinary Research and Diagnostic Laboratory (MVRDL; Mississippi State University, Pearl, MS) for laboratory diagnosis. Tests for bluetongue virus (BTV), brucellosis, bovine viral diarrhea virus (BVDV), epizootic hemorrhagic disease virus (EHDV), Johne's disease, and leptospirosis were requested by the submitting veterinarian.
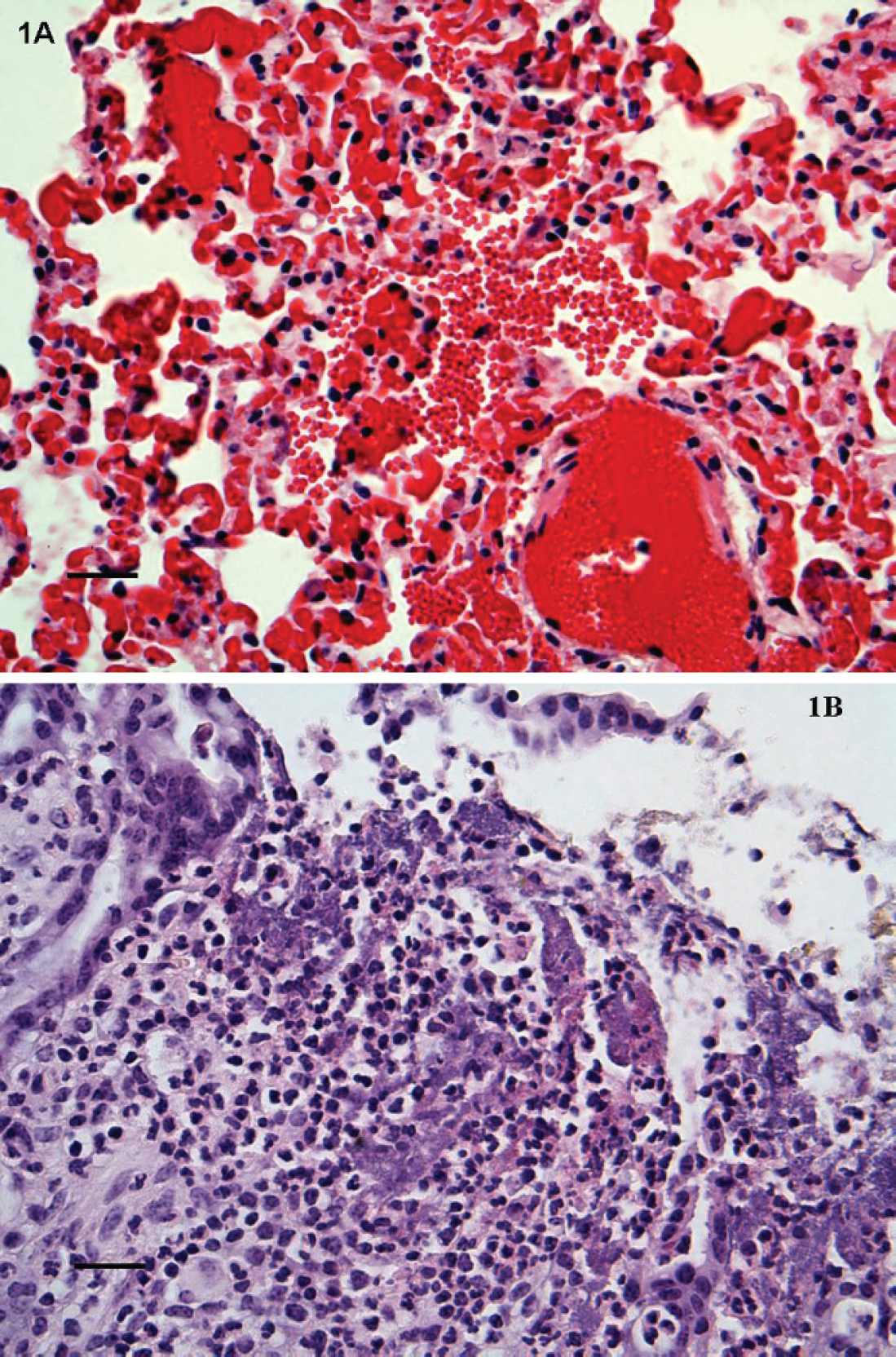
Upon arrival at MVRDL, tissues fixed in 10% buffered formalin were embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin (HE) for microscopic examinations. Histopathology revealed pulmonary hemorrhage, congestion, edema, and interstitial pneumonia in both animals. The lungs were characterized by multifocal intra-alveolar extravasation and edema, diffusely engorged alveolar capillaries with patchy foci of interstitial hemorrhage and fibrin deposition, minimal to mild interstitial infiltrations of lymphocytes, and neutrophils (Fig. 1A). Some of the affected alveoli contained a few macrophages performing erythrophagocytosis. Hemorrhage was also observed in the lumen of occasional bronchioles. Rare granulomas were also scattered within the lung parenchyma. Infrequent small nematodes (possibly Dictyocaulus species) were found in the alveoli and bronchioles of animal no. 1, but not animal no. 2. Other microscopic lesions were also present in the tissues of animal no. 1, such as marked diffuse cholangiohepatitis with sinusoidal leukocytosis, minimal myocarditis with mild multifocal myocardial degeneration, and mild multifocal suppurative cystic colitis with crypt microabscessation and focal erosion (Fig. 1B).
According to the client's request, the serum sample was first tested for the presence of antibodies specific for Anaplasma spp., Brucella abortus, and BTV. Competitive enzyme-linked immunosorbent assays (cELISA) were performed at MVRDL with the Anaplasma Antibody Test Kit a and the Bluetongue Virus Antibody Test Kit b according to the manufacturers' instructions. A Brucella card test (Rose-Bengal test) was performed at Texas Veterinary Medical Diagnostic Laboratory (TVMDL; College Station, TX). Competitive inhibition ELISA (ciELISA) detecting antibody against a common epitope of all known members of the closely related malignant catarrhal fever (MCF) group of viruses was performed at the Washington Animal Disease Diagnostic Laboratory (Pullman, WA). The results of serological tests suggested that animal no.1 was negative for brucellosis, BTV, and MCF, but positive for anaplasmosis. To rule out EHDV, BTV, BVDV, and leptospirosis, molecular diagnostic tests were performed at MVRDL as described previously. 1,2,10 Johne's disease diagnosis was carried out with the use of VetAlert Johne's Real-Time PCR reagents. c Polymerase chain reaction (PCR) results indicated that animal nos. 1 and 2 were free of BTV, BVDV, EHDV, and leptospirosis; animal no. 1 was also negative for Johne's disease.
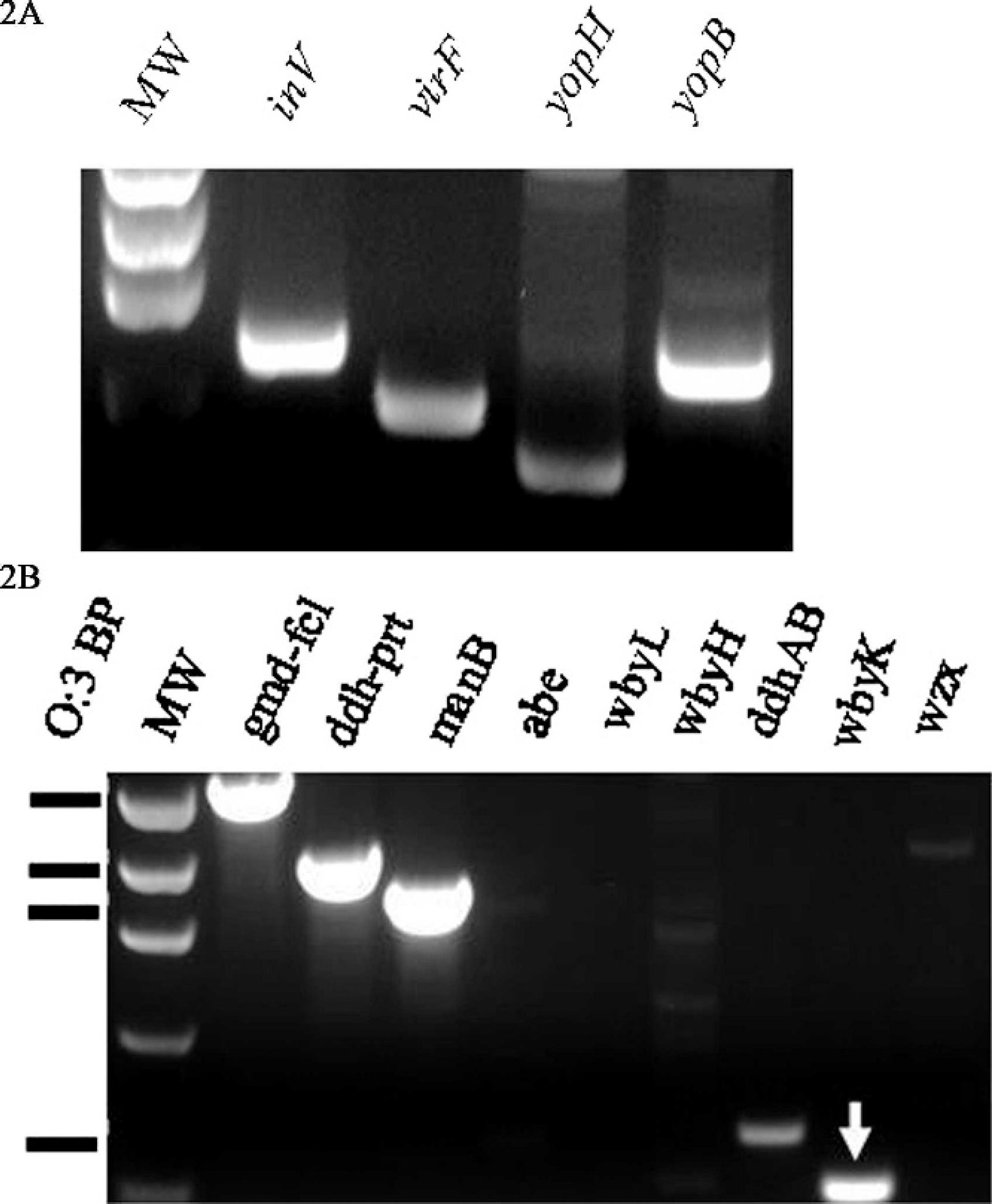
To identify bacterial etiologic agents, fresh tissues of heart, lung, liver, spleen, and colon contents of animal no. 1 and lung tissue of animal no. 2 were cultured on blood and MacConkey agars and incubated at 35°C for 24 hr. Light to moderate growth of Y. pseudotuberculosis was isolated in pure culture from the lung tissues of both animals. Moderate growth of Y. pseudotuberculosis mixed with Escherichia coli and Enterococcus spp. was isolated from the colon content of animal no.1. Identification of the organisms was based on colonial morphology, growth characteristics, and standard biochemical tests using RapID ONE System. d To determine the pathogenic potential of the Y. pseudotuberculosis isolates, PCR assays were carried out to amplify inv, virF, yopB, and yopH with the use of gene-specific primer pairs, including Inv-F/R (5′-CTTCAGATACTGGTGTCGCTGT-3′/5′-ATGCCTGACTAGAGCGATATCC-3′), VirF-F/R (5′-GGCAGAACAGCAGTCAGACATA-3′/5′-GTGAGCATAGAGAATACGTCG-3′), YopB-F/R (5′-GATGGAACATGAGTGCGTTG-3′/5′-TGCCTCTTGGGATATCAGG-3′), and YopH-F/R (5′-GGATACTGGCACGCTATGGT-3′/5′-TACAAGACGCCAAAGTGCTG-3′). e All PCR assays were performed on a Mastercycler ep. f The thermal cycling conditions used to amplify these virulence-associated genes were as follows: a 3-min incubation at 95°C; followed by 30 cycles of 95°C for 30 sec, 55°C for 30 sec, 60°C for 30 sec, and 72°C for 45 sec; and a final incubation at 72°C for 5 min. Amplified PCR products were photographed with an AlphaImager. g Results indicated that the Y. pseudotuberculosis isolates were positive for the 4 virulence-associated genes tested (Fig. 2A). Because serotyping of Y. pseudotuberculosis was not routinely performed by MVRDL, a PCR-based O-genotyping was conducted with 9 sets of primers targeting different regions of the O-antigen gene cluster of Y. pseudotuberculosis. Primers and PCR conditions used for O-genotyping were essentially the same as those described previously, except that individual, instead of multiplex, PCR reactions were carried out. 4 Electrophoresis of the PCR products indicated that the O-genotyping pattern of the isolate did not match with that of any known O-genotypes of Y. pseudotuberculosis (Fig. 2B). In addition to the O-antigen genes possessed by classical O:3, the isolate was also PCR-positive for wbyK, a gene encoding mannosyltransferase-like protein. The wbyK gene (GenBank accession bank it1045184, EU338401) of the isolates shared 94% of homology with the wbyK gene harbored by Y. pseudotuberculosis O:1b. 18 To find out whether the isolates belong to any known serotype of Y. pseudotuberculosis, conventional serotyping was performed at the California Department of Health (Sacramento, CA) with commercial antisera against O side chains of the cell wall LPS. The isolates reacted with anti-sera against Y. pseudotuberculosis O:3 antigen.
Clinical history, pathology, and microbiology results collectively suggested that the sudden deaths of these 2 deer were attributable to Y. pseudotuberculosis infection. Although animal no. 1 was seropositive for Anaplasma and had small numbers of pulmonary nematodes, it was less likely that these conditions were the direct causes. In fact, cervids generally do not show overt clinical signs nor develop apparent gross and histopathological lesions after infection with Anaplasma spp. 11 Acute deaths of cervids have sometimes been associated with severe pulmonary nematodiasis, which, however, is frequently accompanied by characteristic postmortem findings such as pulmonary edema, consolidation, and a large quantity of bloody froth in trachea and bronchi containing adult worms. 11 The histopathological changes in the colon, liver, and heart of animal no. 1 were similar to those reported previously. 3,12,16 However, multifocal pulmonary hemorrhage has rarely been described. 12 A recent study demonstrated that intranasal inoculation of mice with Y. pseudotuberculosis resulted in lung colonization, and the bacterial virulence factors, YopB and YopH, were essential for this process. 9 The isolate from this study was PCR-positive for several key genes (inv, virF, yopB, and yopH) involved in invasion and colonization of host intestine and lung. Isolation of the organism from the colon and lungs of the affected animals, as well as the lesions seen in these organs, indicated an initial Y. pseudotuberculosis-induced colitis followed by systemic dissemination and lung colonization. Because mesenteric lymph nodes were not available for pathologic evaluations, it is not known whether the affected animals had mesenteric lymphadenitis, a common manifestation of yersiniosis. Because of a lack of tissue specimens from other dead deer, a causal link between Y. pseudotuberculosis infections and the sudden deaths of the other deer could not be established.
Increasing amount of evidence suggests that O-antigen of LPS is required for virulence in pathogenic Yersinia, including colonization of host tissues, resistance to complement-mediated killing, and resistance to innate immunity. 6 Characterization of O-antigen gene clusters of Y. pseudotuberculosis and Yersinia pestis have lead to the development of a O-genotyping method. 4,17,18 When Y. pseudotuberculosis collections from Asia and Europe were tested, a single O-genotyping pattern for all O:3 isolates was obtained, although discrepancies between O-serotyping and O-genotyping were seen with several strains, especially O:12 and O:13. 4 In this study, the O-genotyping pattern of the isolates was distinct from what has been reported for all O-serotypes or O-genotypes of Y. pseudotuberculosis. The Y. pseudotuberculosis isolate from this study, belonging to classical serotype O:3, acquired a wbyK gene, which suggested a genetic heterogeneity in the O-antigen gene cluster of classical O:3. The wbyK gene encodes a mannosyltransferase whose function is to add mannose residues to the growing side chain of LPS. 15 Reactivity of the isolates with polyclonal antisera against O:3 antigen suggested that acquisition of wbyK did not alter the common O:3 antigenic epitope. However, whether addition of a wbyK gene results in the generation of any additional antigenic epitope(s) in the LPS O-side chain or is related to the pathogenic phenotype remains to be investigated. For bacterial source tracking and disease diagnosis, it is also important to determine whether the distinct O:3 genotyping pattern observed in the current study reflects a geographic-(North America vs. Asia and Europe) or a host species-associated (humans vs. cervids) genetic variation.
Footnotes
a.
Anaplasma Antibody Test Kit, cELISA, VMRD, Inc. Pullman, WA.
b.
Bluetongue Virus Antibody Test Kit, cELISA, VMRD, Inc. Pullman, WA.
c.
VetAlertTM Johne's Real-Time PCR reagents, Tetracore, Rockville, MD.
d.
RapIDTM ONE System, Remel, Inc., Lenexa, KS.
e.
Custom Oligos, Operon Biotechnologies, Inc., Huntsville, AL.
f.
Mastercycler® ep, Eppendorf AG, Hamburg, Germany.
g.
AlphaImagerTM 3400, Alpha Innotech Corporation, San Leandro, CA.
