Abstract
From 2014–2019, 8 juvenile black bears (Ursus americanus) from different geographic regions were presented to the California Department of Fish and Wildlife because of emaciation, alopecia, and exfoliative dermatitis that resulted in death or euthanasia. Autopsy and histopathology revealed that all 8 bears had generalized hyperkeratotic dermatitis, folliculitis, and furunculosis. Skin structures were heavily colonized by fungal hyphae and arthrospores; fungal cultures of skin from 7 bears yielded Trichophyton equinum, a zoophilic dermatophyte reported only rarely in non-equid species. Additional skin conditions included mites (5), ticks (2), and coagulase-negative Staphylococcus sp. infections (2). No other causes of morbidity or mortality were identified. Molecular comparisons performed at the University of Texas Fungal Reference Laboratory determined that all isolates produced identical banding patterns, potentially representing a clonal population. Dermatophytosis is commonly localized and limited to the stratum corneum of the epidermis and hair follicles. Generalized disease with dermal involvement is rare in immunocompetent individuals; illness, malnutrition, age, or immunosuppression may increase susceptibility. Underlying causes for the severe disease impact in these bears were not evident after physical or postmortem examination. The mechanism by which bears from different geographic locations had severe, T. equinum–associated dermatophytosis from a potentially clonal dermatophyte could not be explained and warrants further investigation.
Keywords
The California Department of Fish and Wildlife (CDFW) evaluated 8 juvenile bears (Ursus americanus) from 2014–2019 with severe, generalized alopecia, crusting and scaling dermatitis, emaciation, and weakness that progressed to death (2) or euthanasia (6). Five male and 3 female cubs 3–18-mo-old were recovered from 6 different counties throughout the state: 4 from northwest California (Trinity [2], Humboldt, Mendocino counties), 3 from northern Sierra Nevada (Nevada [2], Placer [1]), and 1 from southern California (Santa Barbara). All but one bear was presented between the months of November and March.
Autopsy examinations, performed at the CDFW’s Wildlife Investigations Laboratory (Rancho Cordova, CA, USA) and the California Animal Health and Food Safety (CAHFS) Laboratory at the University of California–Davis, identified decreased muscle mass, little-to-no subcutaneous or visceral fat reserves, and severe hyperkeratotic dermatitis and alopecia of the neck, trunk, abdomen, flanks, dorsum, perineum, and head (Fig. 1). Two bears had large numbers of Ixodes pacificus ticks attached to the skin. No other gross lesions were identified.
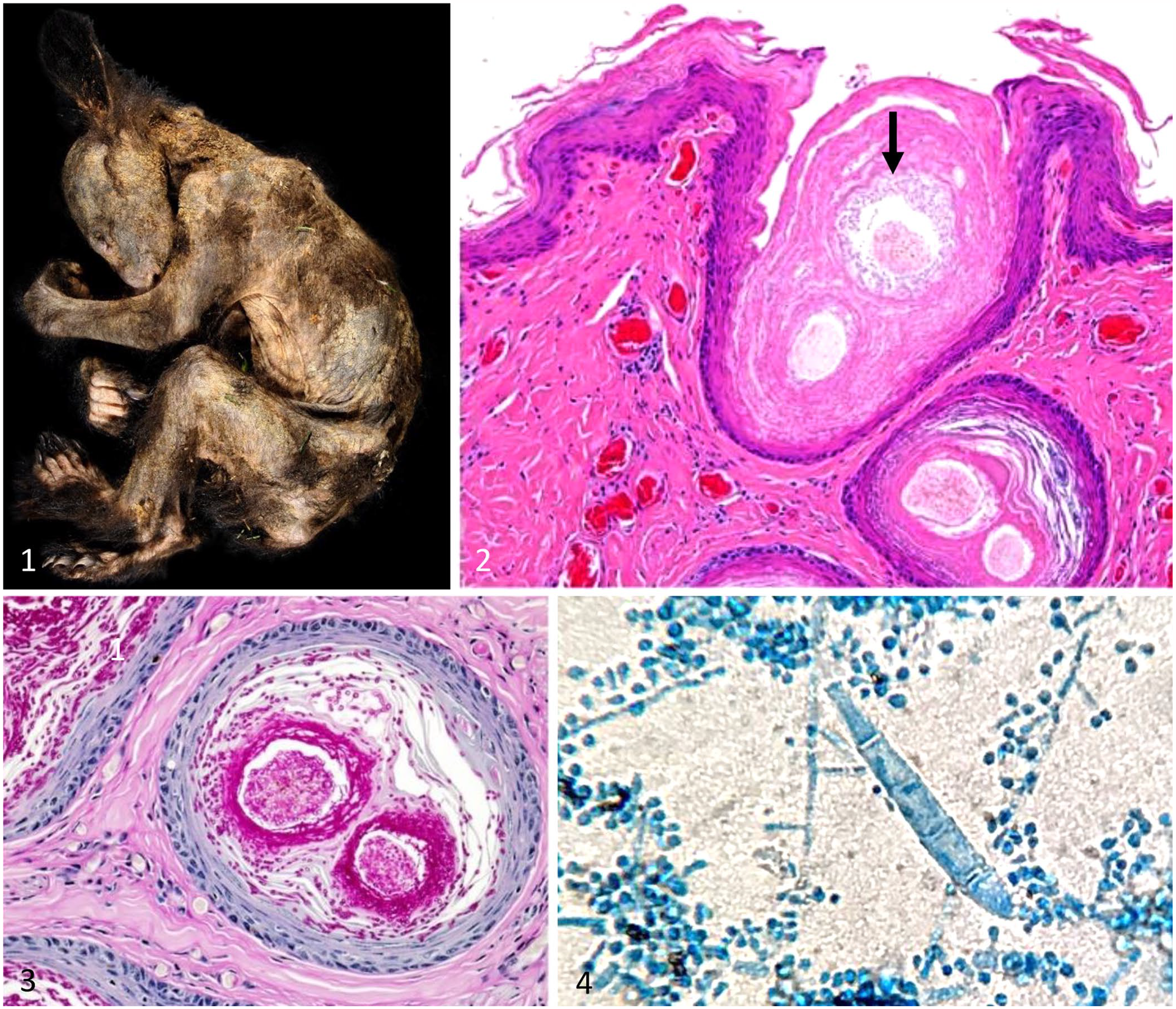
Generalized dermatophytosis caused by Trichophyton equinum in juvenile black bears.
Histologically, all bears were diagnosed with generalized dermatophytosis; hair shafts, follicular keratin, and epidermal keratin were invaded by dermatophyte arthrospores and hyphae. The acanthotic epidermis and follicular infundibula exhibited ortho- and parakeratotic hyperkeratosis that frequently entrapped emerging hair shafts. Some cubs had superficial epidermal pustular dermatitis and neutrophilic folliculitis. Ruptured follicles were associated with granulomas surrounding free hair shafts and keratin (furunculosis). Variable numbers of lymphocytes, plasma cells, and histiocytes surrounded dermal and perifollicular vessels (Fig. 2). Periodic acid–Schiff (PAS) stains revealed the extent of fungal invasion in the epidermal and follicular epithelium (Fig. 3). Two cubs also had foci of full-thickness necrosis with neutrophilic exudates. Additional histologic lesions were uncommon but involved the lungs and liver. Pulmonary lesions included mild lymphohistiocytic interstitial inflammation associated with Emmonsia sp. (1 bear) and bronchopneumonia (1 bear). Random, mild hepatocellular degeneration and lymphocytic hepatitis were evident in the livers of 3 bears.
Histologic examinations of brain, eye, diaphragm, retropharyngeal and prescapular lymph nodes, tonsil, tongue, joint capsules, spleen, liver, gall bladder, adrenal glands, kidneys, esophagus, stomach, small and large intestines, trachea, thyroid glands, larynx, heart, pericardium, ovaries or testicles, and pancreas were unremarkable in all of the cubs.
Coagulase-negative Staphylococcus sp. was recovered on aerobic cultures from both animals with necrosis of the skin. Streptococcus dysgalactiae subsp. equisimilis was recovered from a lung sample in the animal with bronchopneumonia.
Mites consistent with Uriscoptes americanus and Demodex sp. were identified by direct microscopic examination of skin scrapings and/or histopathology in 5 of the 8 cubs examined 8 ; 1 cub had U. americanus and Demodex sp. detected, and 2 had U. americanus identified. Evidence of a chitinous exoskeleton was observed histologically in 2 cubs (suspected mite), but no intact mites were detected, and the identity could not be confirmed.
Three of the cubs had trace amounts of anticoagulant rodenticide (brodifacoum or chlorophacinone) detected in liver samples, but no evidence of hemorrhage. These compounds are not uncommonly identified in trace amounts (below the limit of quantification of 0.01 ppm), and, because no evidence of coagulopathy was detected in the examined cubs, these results were considered incidental findings (Poppenga R.H., pers. comm., 2020 Jul 02).
Fungal cultures were performed on skin samples from 7 of the bears (1 case had only formalin-fixed tissues available), as described previously. 3 Briefly, sections of affected skin/crusts were inoculated onto 2 bi-plates (Derm-Duet; Hardy Diagnostics) containing dermatophyte-tested medium (DTM) in one section and rapid sporulation medium (RSM) in the other section; 1 plate was incubated at 37°C and the other at 25°C. After 6 d of incubation, plates at 25°C had fluffy white colonies that resulted in a DTM color change to red and a RSM color change to light-blue. Microscopic examination utilizing lactophenol aniline blue (LPAB) revealed septate hyphae with parallel walls and abundant pyriform microconidia. Colonies were subcultured onto potato flake agar and incubated at 25°C to facilitate further identification. After 7 d, fluffy, white colonies that were yellow at the periphery with a tan-peach reverse were observed. Additional microscopic examination with LPAB revealed septate hyphae and abundant microconidia as well as rare fusiform, septate, thin-walled macroconidia (Fig. 4), consistent with Trichophyton equinum. 3
Isolates recovered from 7 of the bears were submitted to the Fungus Testing Laboratory at the University of Texas–San Antonio, for molecular characterization. Isolate identity was confirmed using a combination of phenotypic features and DNA sequencing of the internal transcribed region17,20 and tubulin17,18 genes. Rapid amplification of random polymorphic DNA analysis with primers OPA-15 (5′-TTCCGAACCC-3′) and M13 (5′-GAGGGTGGCGGTTCT-3′)6,7 determined that all 7 isolates produced DNA bands that were distinct from quality control strains but identical to each other, representing a potential clonal population.
T. equinum dermatophyte infections in horses are frequently self-limiting, and generalized cutaneous involvement is rare. 16 Lesions are often identified on skin that stays moist for long periods, such as on the distal limbs and under tack. Concurrent illness, poor nutrition, overcrowding, stress, and immunosuppression are predisposing factors associated with clinical disease.9,16 T. equinum requires specific nutrients including nicotinic acid, which is present in equine hair shafts but not those of other species. 21 We did not detect additional infectious agents (Leptospira sp. [detection by florescent antibody testing], Salmonella sp. [detection by PCR on feces], Brucella sp. [detection by bacterial culture or serology], canine distemper virus [detection by immunohistochemistry]), which may have predisposed these cubs to increased susceptibility to T. equinum.
The severity of equine T. equinum clinical disease is based on fungal strain, cutaneous defenses, environmental factors, and the immune status of the animal.9,11 Young animals are more susceptible than adults and generally have a prolonged course of disease. 11 A defective cell-mediated immune response can predispose an animal to chronic or recurrent dermatophytosis9,19; however, thymus tissue was only examined in one bear and no abnormalities were identified. Evidence of defective immune status was not detected in the cubs in our study. Skin damage from self-trauma or external parasites can exacerbate fungal spread and enhance disease severity.9,11
T. equinum infections in non-equid species are uncommon. Human cases are reported rarely, are generally localized, and can frequently be traced to contact with horses.1,21 In 2015, T. equinum was reported in farmed mink with microscopic skin lesions similar to those described in bears from our report; interestingly, the farmed mink were otherwise clinically healthy and in good body condition, unlike the emaciated cubs described here. 13
Dermatophyte pathogenesis begins with the anchoring of arthroconidia to keratinocytes, protease and sulfite production to facilitate keratin degradation, and hyphal germination and invasion of the stratum corneum. 19 Hydrolytic enzyme secretion is essential for virulence, facilitating digestion of insoluble keratin substrates, release of nutrients from host tissues, and hyphal spread. 12 Hair shaft invasion and follicular damage result in regional alopecia, crusting, and scaling characteristic of dermatophyte infections. Extension into deeper skin structures is uncommon but can lead to inflammation, evidence of self-trauma, and, in severe cases, pyogranulomatous furunculosis.9,16
Advances in molecular characterization and whole-genome sequencing have provided tremendous insights into dermatophyte pathogenicity and virulence.10,12 Comparative genomics of closely related dermatophytes (T. equinum, T. tonsurans, T. rubrum, Microsporum canis, and M. gypseum) identified gene families not present in other pathogenic fungi, including enzymes that permit chitin-binding, proteases that facilitate growth and nutrient acquisition on keratinized tissues, and compounds that mask cell wall components to evade host immune responses.10,19 Hemolytic activity, cytokine induction, and the ability to induce hypersensitivity in the host are associated with increased virulence.9,19
The zoophilic dermatophyte T. equinum and the anthropophilic fungus T. tonsurans, which is the most common cause of tinea capitis in humans, are highly related genetically yet occupy distinct ecologic niches.4,10 Although sexual reproduction in dermatophytes has been difficult to confirm in vitro, the mating type locus and meiosis-specific genes were identified in all dermatophytes examined, including T. equinum. 10 The opportunities for genetic acquisition in these pathogens is considered to be limited; however, recent work has identified other means of genetic exchange (parasexual, hybridization) that can produce novel clones with enhanced pathogenicity or determinants that facilitate spread to novel host species. 5
Arbitrarily or randomly primed PCR analysis utilizes short primer sequences to create a strain-specific DNA fingerprint that can be used for epidemiologic assessments of individual isolates.6,7 The clonal pattern of the T. equinum isolates from our cases indicates that these cubs may have encountered a virulent dermatophyte strain at a time when they were least equipped immunologically to control the infection. Human infections from zoophilic dermatophytes are believed to produce greater inflammatory responses than those from anthropophilic dermatophytes. 4 The severe dermatologic lesions observed in these cubs may be at least partly the result of severe inflammation accompanying the infection by a primarily equine-associated fungal species. In humans, 30–70% of individuals may carry dermatophytes asymptomatically on their skin. 22 Fungal cultures performed on a variety of skin sites from a convenience sample of 4 unaffected bears submitted to CAHFS for autopsy did not recover any dermatophytes; however, this does not necessarily rule out unaffected bears as possible sources of the T. equinum.
During times of environmental extremes such as drought and flooding, bears may be forced to travel large distances in search of food, increasing the likelihood of exposure to humans and domestic animals. 2 Several of these animals were reported to or collected by CDFW from residential neighborhoods. California experienced a severe, persistent drought from December 2011–March 2017, followed by 2 y of flooding, with extensive fire damage over much of the state during this time. During the decade from 2009–2018, more than 3,300 fires burning over 2.8 million ha (7 million acres) were recorded in the state, which is more than twice the area burned in 1979–1998 (https://emlab.ucsb.edu/sites/default/files/documents/wildfire-brief.pdf). The presumed orphan status of the cubs, food insecurity, and habitat stressors could have contributed to dermatophyte susceptibility in the juvenile bears of our report. Droughts can strongly affect the dynamics of animal populations, and extended drought has had the greatest impact on carnivores. 15 Four additional juvenile bears with severe, T. equinum–associated dermatophytosis were identified during the first quarter of 2021, possibly associated with the habitat destruction and food scarcity related to the devastating fires in the state during 2020.
Cases of sarcoptic mange caused by the mite Sarcoptes scabiei have increased in several species, including black bears, in both frequency and geographic region in Pennsylvania in recent years; increased interactions with other S. scabiei–infected hosts may have contributed to this spike in cases. 14 The presence of the cubs in suburban or semi-rural environments may have facilitated contact with locations in which horses had been housed, which may have been a source of T. equinum arthrospore exposure in the bears of our report. Ectoparasites including sarcoptic mites, demodex mites, and ticks were inconsistently identified in our cases, but could have resulted in sufficient skin damage to facilitate severe fungal spread. The significance of T. equinum dermatophytosis as a sole or contributing pathogen in the California bear population warrants further investigation.
Footnotes
Acknowledgements
We thank Dr. Nathan Wiederhold and the staff at the Fungus Testing Laboratory at the University of Texas–San Antonio, for molecular evaluation of these isolates.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our work was supported by the California Department of Fish and Wildlife, Sacramento, CA, USA.
