Abstract
We describe herein fatal hepatic sarcocystosis in a free-ranging grizzly bear (Ursus arctos horribilis) cub with apicomplexan infection of the liver and brain, both demonstrating 100% homology for Sarcocystis canis and S. arctosi. Fatal hepatic sarcocystosis in dogs has been etiologically associated with intrahepatic schizonts of S. canis. In black and polar bears, a S. canis–like organism produces schizonts in the liver and massive hepatic necrosis. Although intramuscular sarcocysts, taxa S. arctosi and S. ursusi, have been described in healthy brown and black bears, respectively, they have not been detected in bears with hepatic sarcocystosis, to our knowledge, and it is currently unknown whether bears represent an aberrant or intermediate host.
Hepatic sarcocystosis is a fatal disease first reported in dogs in 1991. 10 The disease has since been reported in dogs,1,7,18 a chinchilla, 16 a horse, 4 a Stellar sea lion, 21 a striped dolphin, 17 a bottlenose dolphin, 2 and captive black3,23 and polar 11 bears. Structural examination of schizonts within hepatocytes in the original canine case revealed that asexual reproduction occurred by endopolygeny without a parasitophorous vacuole, placing the organism in the genus Sarcocystis. 10 The organism did not react with Toxoplasma gondii, Neospora caninum, or Caryospora spp. antiserum on immunohistochemistry (IHC), further supporting identification as Sarcocystis spp. 10 Given that Sarcocystis spp. are generally host specific in the asexual phase, the taxon Sarcocystis canis was proposed. 10
Sarcocystis is an apicomplexan genus in the family Sarcocystidae, which has a 2-host, prey–predator life cycle. 10 The definitive host, usually a carnivore, defecates sporocysts and/or oocysts produced by sexual reproduction, which are ingested by the intermediate host, usually a herbivore, resulting in the formation of intracellular schizonts that produce merozoites asexually. 10 Release of merozoites into the systemic circulation leads to formation of sarcocysts containing bradyzoites in muscle and brain, which, when eaten by the definitive host, perpetuates the life cycle. 10 With the exception of S. neurona and S. canis, apicomplexans are generally host specific for the intermediate host and do not form schizonts in carnivores.6,10
Immunologic studies of ursine hepatic sarcocystosis identified Sarcocystis spp. 3 but not S. neurona in liver,3,11 suggestive of S. canis–like infection. S. canis–like schizonts have been reported only in affected ursine liver,3,11,23 whereas sarcocysts have been identified only in skeletal muscle of bears unaffected by hepatic sarcocystosis. 9 Intramuscular sarcocysts have unique morphology, and 2 species have been described: S. arctosi in 2 brown Alaskan bears 9 and S. ursusi in a black Pennsylvanian bear. 8 Molecular analysis of a partial sequence of the 18S ribosomal RNA (rRNA) gene subsequently revealed that S. arctosi shared 100% homology with S. canis detected in polar bear liver, indicative of either a close evolutionary relationship between the 2 species or schizont and sarcocyst stages of the same species. 9 Partial sequencing of the 18S rRNA gene from the liver and brain of the captive black bear also revealed 100% homology withS. canis and S. arctosi. 3
The life cycle of S. canis has not yet been described, rendering unequivocal diagnosis of this infection challenging and reliant on unique serologic and molecular tests. 6 Attempts to further speciate the polar bear schizont looked at a conserved segment (18S) and variable segment (ITS-1) of the rDNA gene, which most closely supported an identity ofS. canis. 6 Molecular evaluation of schizonts from the liver of a bottlenose dolphin for cox1, a mitochondrial cytochrome C oxidase subunit, revealed 98% identity with S. canis from polar bear, leading to diagnosis of S. canis–like hepatic sarcocystosis in this marine mammal. 2 Thus, a number of molecular tests have demonstrated that schizonts associated with ursine hepatic sarcocystosis exhibit a close relationship to S. canis.
In October 2016, an adult grizzly bear (Ursus arctos horribilis) sow with 2 cubs was living around a small northern coastal community in British Columbia (BC), Canada. Both cubs appeared healthy and of similar size. On October 15, one of the cubs was observed to be lagging behind the family group, holding its head down. The following day, the cub was found dead whereas the other cub remained healthy. The dead cub was shipped to the Animal Health Centre in Abbotsford, BC, for autopsy.
On postmortem examination, the female 39.5 kg cub was moderately autolyzed, with adequate muscle mass and internal fat stores. The spleen was markedly enlarged and congested. The liver had rounded edges with mild generalized tan pallor. The stomach was devoid of content save for gray-black mucus loosely adhered to the mucosa.
Samples of spleen, lung, liver, and intestine were taken for bacterial culture and inoculated onto Columbia blood agar with 5% sheep blood (Oxoid, Nepean, ON, Canada) and MacConkey agar (Oxoid) and incubated at 35°C ± 2°C in 5–10% CO2 and aerobic conditions, respectively. Agar plates were observed at 24 and 48 h for bacterial growth, and bacterial isolates were further subcultured to obtain a pure isolate for identification purposes. Bacterial identifications were performed based on colony morphology, growth characteristics, Gram stain, biochemical testing, and molecular techniques. Samples of all organs including brain and skeletal muscle were placed in 10% formalin, processed routinely, and stained with hematoxylin and eosin for histologic evaluation. IHC was conducted for S. neurona, Sarcocystis spp., T. gondii, and N. caninum on liver as described previously.3,12
Samples of lung, liver, spleen, kidney, and brain were frozen at −70°C for molecular testing. Thawed liver and brain were diluted 1:10 in minimum essential medium, homogenized in a stomacher for 2 min at medium speed, centrifuged briefly, and DNA extracted (QIAamp DNA mini kit, Qiagen, Toronto, ON, Canada) according to the manufacturer’s instructions. The DNA extract from liver was tested for T. gondii and N. caninum by conventional PCR assays as described previously.14,22 DNA extracts from liver and brain were also evaluated for Sarcocystis spp. using conventional PCR assays that target the 18S rRNA gene.3,13
Conventional PCR products were electrophoresed on 2% agarose gels with ethidium bromide and then analyzed (AlphaImager HP imaging system, ProteinSimple, Santa Clara, CA). PCR assays for Sarcocystis spp.,3,13 both targeting segments of the 18S rRNA gene, amplified the expected 521-bp 3 (liver) and 897-bp 13 (liver and brain) amplicons, respectively. Positive PCR results were confirmed by direct sequencing of the PCR amplicons (Big Dye Terminator v.3.1 cycle sequencing kit, Life Technologies, Burlington, ON, Canada) and a genetic analyzer (ABI 3500 genetic analyzer, Thermo Fisher Scientific, Waltham, MA) according to the manufacturer’s instructions. DNA sequences were assembled and analyzed using DNASTAR Lasergene 14 (https://www.dnastar.com/software/lasergene/) and BLAST (http://blast.ncbi.nlm.nih.gov/Blast.cgi).
Bacteriology revealed mixed-species bacteremia associated with heavy growth of Klebsiella spp. and hemolytic E. coli from lung and liver, plus Enterococcus spp. and non-hemolytic E. coli from spleen. Microscopically, the spleen exhibited severe sinus congestion. There was moderate lymphoid depletion of splenic white pulp and thymus. There was moderate multifocal microgliosis of the brainstem. Small multifocal infiltrates of mixed plasma cells and lymphocytes were scattered in the renal interstitium, occasionally associated with adjacent tubular epithelial degeneration and necrosis. The liver exhibited widespread single-cell to multifocal hepatic necrosis with mild periportal to randomly scattered mixed inflammatory cells and moderate multifocal hepatocellular mineralization (Fig. 1). A few intracellular schizonts (Fig. 2) were scattered in the liver parenchyma and were negative on IHC for S. neurona, Sarcocystis spp., and T. gondii. There was positive staining of one schizont for N. caninum. Liver was negative on PCR analysis for T. gondii and N. caninum. Sequence analysis of PCR products from liver (521-bp segment) and brain (897-bp segment) of the 18S rRNA gene were both positive for Sarcocystis spp. with 100% homology for S. canis (GenBank DQ146148.1) andS. arctosi (GenBank EF564590.1)
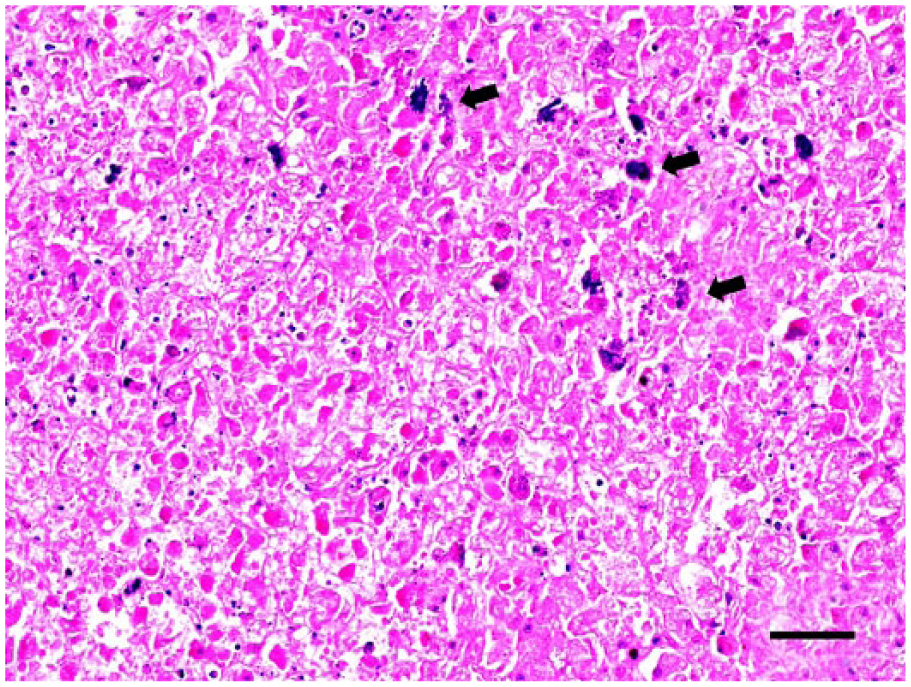
Grizzly bear cub liver. Normal architecture is effaced, and there is widespread necrosis with mineralization (arrows) of hepatocytes. H&E. Bar = 50 µm.
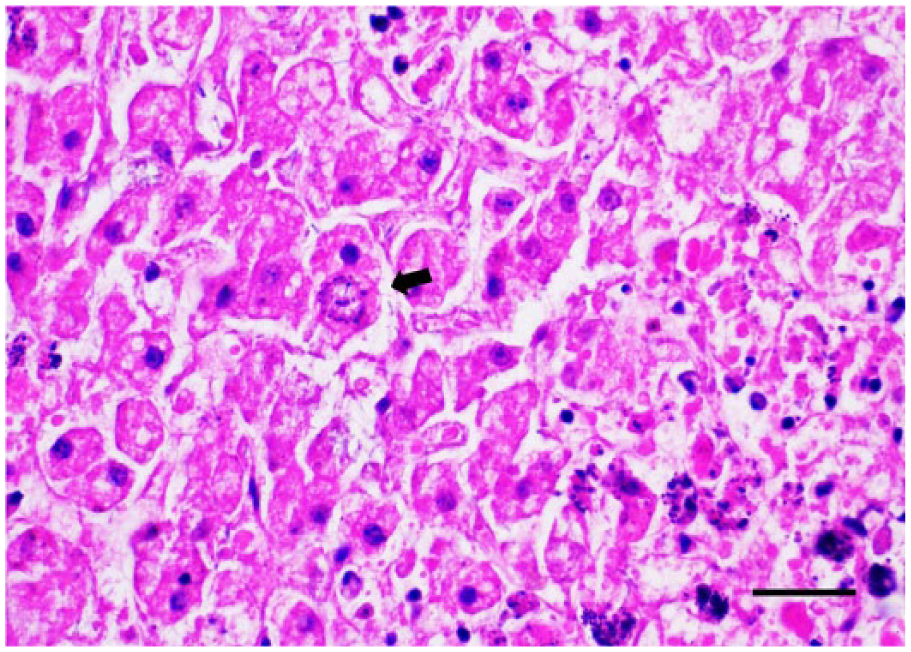
Grizzly bear cub liver with an intrahepatic schizont (arrow). H&E. Bar = 30 µm.
In contrast to the positive result reported for the captive black bear, 3 the IHC test for Sarcocystis spp. in our grizzly cub was negative. Yet, PCR detection of Sarcocystis spp. in the cub’s liver was consistent with the previous findings. 3 Controls were positive and schizonts were present in the cub’s test sample, thus this negative result may reflect reduced or unpredictable IHC efficacy when applied to autolyzed and necrotic tissue. 19 Molecular testing of autolyzed tissue is more sensitive than IHC for N. caninum in bovine fetal tissue15,20 and this may also be true for Sarcocystis spp. Paradoxically, there was positive N. caninum staining of one schizont in the cub with a concomitant negative PCR test for the agent. Given that molecular methods are more sensitive and specific than IHC for detection of N. caninum, we concluded that N. caninum was not contributory based on the negative PCR test. 20
Fatal hepatic sarcocystosis caused by a S. canis–like agent was diagnosed in this free-ranging grizzly bear cub based on histopathology and molecular analysis. This finding is consistent with previous reports of hepatic sarcocystosis in captive black and polar bears in which severe hepatic necrosis with intrahepatic schizonts was also demonstrated to be associated with a S. canis–like organism.3,11,23 In earlier studies, the etiologic diagnosis was based on either morphology of the schizonts 23 or morphology plus a negative IHC test for S. neurona. 11 With the introduction of molecular capability, subsequent studies utilized PCR analysis with partial gene sequencing of 18S rRNA for agent identification.3,9 Schizonts from the grizzly bear cub in our study and both the captive polar bear and the BC black bear previously reported were all shown to have 100% homology for S. canis andS. arctosi.3,9
The grizzly bear cub exhibited lesions in organs other than liver. Most notably, multifocal microgliosis was observed in the brainstem. Microgliosis was also reported in the brain of the polar bear 11 and the BC black bear. 3 Although schizonts have not been observed in affected ursine brain, PCR analysis of the BC black bear demonstrated an identical 18S rRNA sequence in both liver and brain, indicative of extrahepatic S. canis–like infection. 3 Schizonts were not observed in our grizzly bear brain, but molecular analysis was positive for Sarcocystis spp., with 100% homology for S. canis and S. arctosi. Encephalitic S. canis infection can occur in dogs in the absence of hepatic or other organ lesions.10,18 In contrast, the polar, black, and grizzly bears had both hepatic and encephalitic lesions suggestive of multisystemic disease, as has been reported for dogs infected with S. neurona.5,7
Once considered a rare disease, hepatic sarcocystosis caused by a S. canis–like organism has now been reported in captive black bears,3,23 captive polar bears, 11 and our free-ranging grizzly bear cub. The presence of schizonts in the liver indicates that bears are acting as either an intermediate or aberrant host for this parasite. Simultaneous infection with a S. canis–like agent in the brain and liver of bears with fatal hepatic sarcocystosis suggests that bears may in fact be an intermediate host of the organism. 10 Aberrant hosts do not form sarcocysts. 5 Although sarcocysts have not been demonstrated in the brains of affected bears, 2 species of intramuscular sarcocysts have been identified in North American bears, S. arctosi and S. ursusi.8,9 S. arctosi has been shown to share identical segments of DNA with S. canis, which, to date, has only been found in intrahepatic schizonts, suggesting that these 2 species of Sarcocystis may be the same agent or very closely aligned on an evolutionary basis. 9 If S. arctosi and S. canis are in fact the same agent, bears would represent an intermediate host for S. canis.
The grizzly bear is now the third ursine species to be reported with fatal hepatic sarcocystosis associated withS. canis–like infection. In order to further elucidate the role of S. canis in the health of bears, molecular screening of deceased bears for S. canis–like infection in organs, brain, and skeletal muscle is warranted.
Footnotes
Acknowledgements
We thank Sandra Etheridge, Joanne Taylor, Fiona Downer, Kristen Moffitt, Chelsey Wood, Erin Zabek, Jaime Battle, Giselle Hughes, Melissa Trapp, Erin Graham, Danielle Lewis, and Ken Sojonky for their contributions and expertise. The BC Conservation Officer Service is much appreciated for the effort in transporting this cub to the laboratory.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
