Abstract
Amyloidosis is diagnosed by the histologic detection of amyloid deposits; however, this method has limitations such as a prolonged diagnosis time and the need for histologic proficiency. We aimed to develop a rapid and simple method for diagnosing amyloidosis by targeting amyloid-specific endogenous fluorescence, which has not been reported previously, to our knowledge. Fluorescence fingerprint analysis of amyloid extracts and tissue homogenates derived from amyloid A (AA) amyloidosis-affected cattle exhibited a specific intrinsic fluorescence pattern. Furthermore, principal component analysis using analytical data revealed that AA could be identified by peaks near λex 350 nm and λem 430 nm. Fluorescence spectrometry analysis using tissue homogenates, which does not require special histochemical staining, enables the rapid detection of bovine AA.
Amyloid is generated by the misfolding of host proteins and is characterized by the cross-β-sheet structure. 1 Amyloid A (AA) amyloidosis, which is characterized by systemic AA deposition, is one of the most common forms of systemic amyloidosis in animals and humans. In AA amyloidosis patients, amyloid deposits are observed in the kidneys, liver, and gastrointestinal mucosa, and patients with severe amyloidosis die with an acute course. 2 The diagnosis of amyloidosis is made by the histologic detection of amyloid deposits using biopsy or autopsy specimens. However, histochemical detection of amyloid with Congo red staining, 3 which has been used for ~100 y, has significant limitations such as a prolonged diagnosis time (1–3 d) and the need for histologic proficiency. Therefore, the development of a rapid and simple detection method is desirable.
Intrinsic fluorescence of several amyloid fibrils has been reported,4,5 and is considered to be a structure-specific fluorescence based on the cross-β-sheet structure. Therefore, the use of amyloid-specific intrinsic fluorescence may provide a rapid and simple method for diagnosing amyloidosis. 6 However, the intrinsic fluorescence of AA has not been identified. We aimed to detect the intrinsic fluorescence of bovine AA using fluorescence fingerprint analysis (FFA) and principal component analysis (PCA). 7
Liver and kidney samples from AA amyloidosis–affected cattle and liver from healthy cattle were used for amyloid extraction (Suppl. Figs. 1–3). Amyloid was extracted from 3–5-g samples of each tissue using the water extraction method. 8 By polarized microscopy of Congo red–stained specimens, amyloid was confirmed in the liver and kidney extracts from cattle with amyloidosis (Suppl. Figs. 4, 5), but not in the extracts of liver from healthy cattle (Suppl. Fig. 6). In amyloid-positive livers, few lesions other than amyloid deposits were observed, such as fibrosis and inflammation. The protein concentration of each extracted amyloid solution was determined (BCA protein assay kit; Takara Bio). After standardizing the protein concentration in each extract (0.25 mg/mL), FFA was performed using a fluorescence spectrophotometer (FP-8300; Jasco) at room temperature. The measurements were performed 5 times on each sample. In the amyloid-positive extracts, peaks were observed at λex 350 nm and λem 430 nm, in addition to the aromatic amino acid–derived peak at λex 280 nm and λem 330 nm (Suppl. Figs. 7, 8, 10, 11), but not in the amyloid-negative extract (Suppl. Figs. 9, 12).
Next, we analyzed tissue homogenates from 4 different cases (2 amyloid-positive livers, 2 amyloid-negative livers). All tissues were stored at −30°C until use. The homogenates were prepared by homogenizing 10 mg of each tissue with distilled water at a ratio of 1:100 w/v on ice using a bead homogenizer (µT-12; Taitec); 1 mL of each homogenate was subjected to FFA at room temperature; FFA was performed 5 times on each homogenate. The total time from sample preparation to the end of the FFA was 40–70 min per sample. As with the extracts, a peak at λex 350 nm and λem 430 nm was observed in amyloid-positive samples (Fig. 1; Suppl. Figs. 13, 14), but not in amyloid-negative samples (Fig. 2; Suppl. Figs. 15, 16).
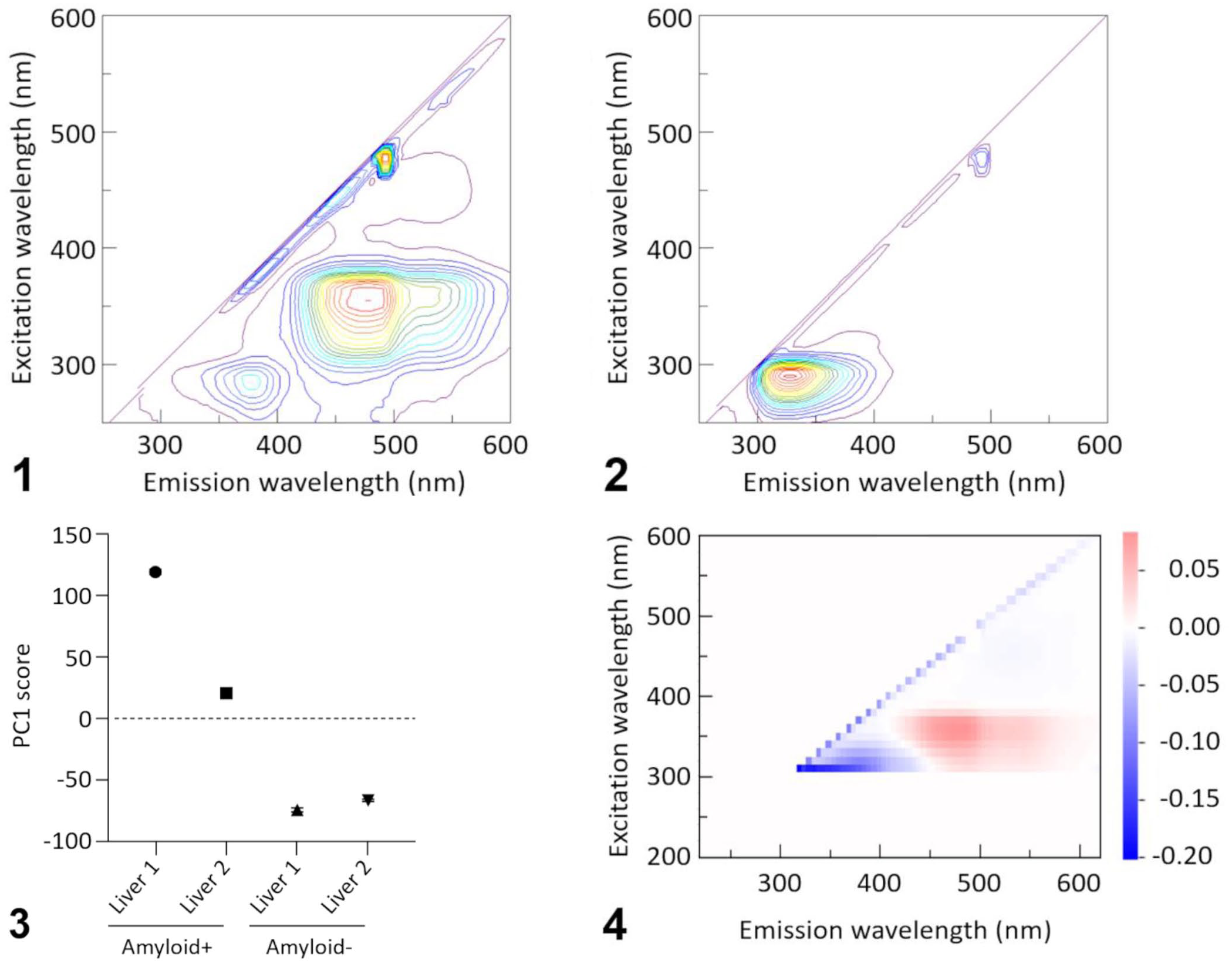
Intrinsic fluorescence of bovine tissue homogenates.
To detect amyloid-specific components, PCA was performed using the 3-dimensional data obtained by FFA. As a pretreatment, data on λex of ≤ 300 nm were removed to rule out the effects of intense fluorescence from aromatic amino acids, such as tryptophan, which are considered inappropriate for the analysis of amyloid-derived peaks. Similarly, data on λex 460–490 nm and λem 485–500 nm, including instrument-derived stray light peaks, were removed. PCA was performed using each combination of excitation and fluorescence wavelengths obtained by FFA as an explanatory variable. Separation of the amyloid-positive or amyloid-negative samples was confirmed in the first principal component score (Fig. 3). The amyloid-positive sample exhibited a positive eigenvector near λex 350 nm and λem 430 nm, whereas the amyloid-negative sample exhibited a negative eigenvector in the same region (Fig. 4), indicating that this peak specifically occurred as a result of the presence of amyloid.
Our results clearly support the intrinsic fluorescence of AA. The fluorescence peaks at λex 350 nm and λem 430 nm are located close to the peak of intrinsic fluorescence exhibited by amyloid fibrils derived from insulin and α-synuclein.9,10 In contrast, amyloid β fibrils exhibit fluorescence spectra at λex 405–450 and λem 455–480 nm.9,11 These facts suggest that the intrinsic fluorescence of amyloid fibrils have different spectra depending on the precursor protein, and the factors that determine these properties may not be limited to the cross-β-sheet structure.
Fluorescence spectrometry analysis using tissue homogenates can be used in the diagnosis of AA amyloidosis without preparing a tissue specimen. This method can be carried out at typical diagnostic laboratories with a fluorescence spectrophotometer. Our results may contribute to the rapid and simple diagnosis of AA amyloidosis.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387211049217 – Supplemental material for Intrinsic fluorescence–based label-free detection of bovine amyloid A amyloidosis
Supplemental material, sj-pdf-1-vdi-10.1177_10406387211049217 for Intrinsic fluorescence–based label-free detection of bovine amyloid A amyloidosis by Naoki Ujike, Susumu Iwaide, Yuki Ono, Takayuki Okano and Tomoaki Murakami in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our research was partially supported by JSPS KAKENHI (grant 20K15660) and the Adaptable and Seamless Technology transfer Program through target-driven R&D (A-STEP; grant JPMJTM20CY) from Japan Science and Technology Agency (JST), and the Program on Open Innovation Platform with Enterprises, Research Institute and Academia (OPERA) from JST.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
