Abstract
Two-dimensional electrophoresis and immunoblotting were used to characterize 7 serum amyloid A (SAA) isoforms in cows with amyloidosis or chronic inflammation. Five SAA isoforms (isoelectric point [pI] 5.6, 6.1, 6.4, 6.8, and 7.7) were detected in all 10 amyloidosis serum samples, while 2 isoforms, pI 5.2 and 8.6, were detected in 4 and 9 of the samples, respectively. The same 7 isoforms were also detected in the serum of cows with chronic inflammation, but SAA pI 5.2 and 8.6 were detected in only 1 and 2 of 10 samples, respectively. It was concluded that although an amyloid-specific SAA isoform was not detected, examination of SAA isoform detection patterns may help identify bovine amyloidosis.
Bovine amyloidosis is a systemic disease in which amyloid A (AA) proteins polymerize to form insoluble amyloid fibers with beta structures that accumulate abnormally in various organs. 12,13 Bovine amyloidosis belongs to the same group of diseases as human systemic reactive AA amyloidosis 6 and is characterized by persistent and intractable diarrhea and systemic edema; a rectal examination often confirms nephromegaly. 6,11,13,14,17 Clinicopathological features include nephrotic syndrome-like symptoms, such as severe hypoproteinemia and protein-uria. 6,13,14,17 Histopathological analysis reveals severe amyloid deposition in various organs, especially the kidney and liver. 16,22 A kidney biopsy is needed to confirm the diagnosis, 6,17 but as it is difficult to perform this procedure in a clinical setting, some cows may die without a diagnosis being made.
Serum amyloid A (SAA) concentrations and isoform patterns of cows with amyloidosis (A), cows with chronic inflammatory disease (B), and healthy cows (C).*
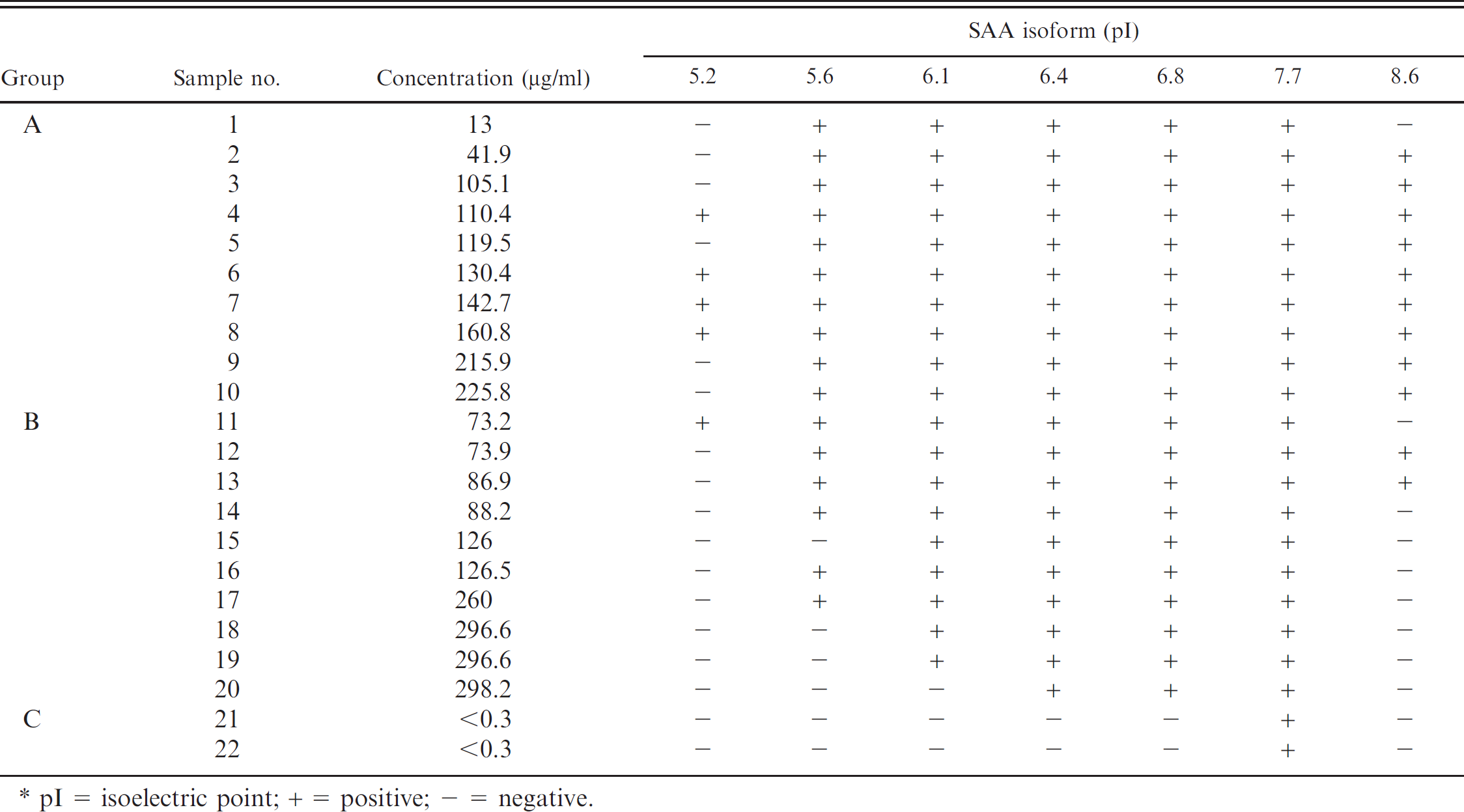
pI = isoelectric point; + = positive; - = negative.
Serum amyloid A (SAA), a precursor of AA with a molecular weight of approximately 12–14 kD, is an apoprotein and one of the serum acute phase reactive proteins. 3,10,15 During inflammation, SAA is present in high concentrations within serum lipoproteins, especially HDL3, 3 and plays a role in the repair of tissue damaged by the inflammatory response. 20 The amino acid sequences of purified AA and the N-terminus of SAA are highly homologous; thus, the incomplete degradation of SAA presumably results in formation and deposition of the AA fragment. 8 Human SAA has several isoforms 4,19 ; those isoforms that tend to cause amyloid accumulation are thought to be more closely associated with the onset of amyloidosis. 5,9 While several bovine SAA isoforms have been reported, 1 their clinical significance is not yet well understood. In the current study, 2-dimensional electrophoresis was used to compare the SAA isoform detection patterns of bovine amyloidosis and those of chronic inflammation.
Sera from 10 Holstein-Frisian cows with amyloidosis (samples 1–10) and 10 Holstein-Frisian cows with chronic inflammation, including 3 with mastitis and 7 with arthritis, were obtained (samples 11–20). The animals with chronic inflammation were diagnosed by medical history of a chronic course of clinical disease and laboratory findings such as hypergammaglobulinemia. The causative organisms were not determined, but the cows had been treated with antibiotics and anti-inflammatory drugs for several days. Diagnosis of chronic inflammation or amyloidosis was determined by necropsy and histopathological examination at Obihiro University of Agriculture and Veterinary Medicine (Obihiro, Hokkaido, Japan). Amyloid accumulation was confirmed histologically by Congo red staining. Sera from 2 healthy cows with no lesions were used as controls (samples 21 and 22). Serum AA concentrations were measured using a commercial enzyme-linked immunosorbent assay kit, a which used rat monoclonal antibody against human SAA. Reactivity of the rat monoclonal antibody against human SAA was confirmed by using bovine SAA, which was purified in the authors' laboratory (data not shown).
Bovine SAA was purified from a pool of acute phase serum by ultracentrifugation, 21 gel filtration, 7 reverse-phase high-performance liquid chromatography, 18 and ion exchange chromatography. 23 Rabbit polyclonal antiserum to bovine SAA was obtained using the method described in a previous report. 23 After isofocusing of serum, this antiserum recognized the same protein bands as did a commercially available rabbit antibody raised against human SAA. b
Immobilized pH gradient strips (pH range: 3–10) and a commercial isoelectric focusing (IEF) system c were used according to the manufacturers' recommendations. Total protein concentration of each serum sample was adjusted to 1 mg/ml, and 125 μl of each was loaded for IEF. Two-dimensional analysis was performed with 15% sodium dodecyl sulfate-polyacrylamide gel electrophoresis using a commercial system d and subsequent electroblotting onto a polyvinylidene fluoride membrane. Blots were incubated with prepared rabbit anti-bovine SAA polyclonal serum, and signal was detected by enhanced chemiluminescence using a Western blotting detection reagent. e
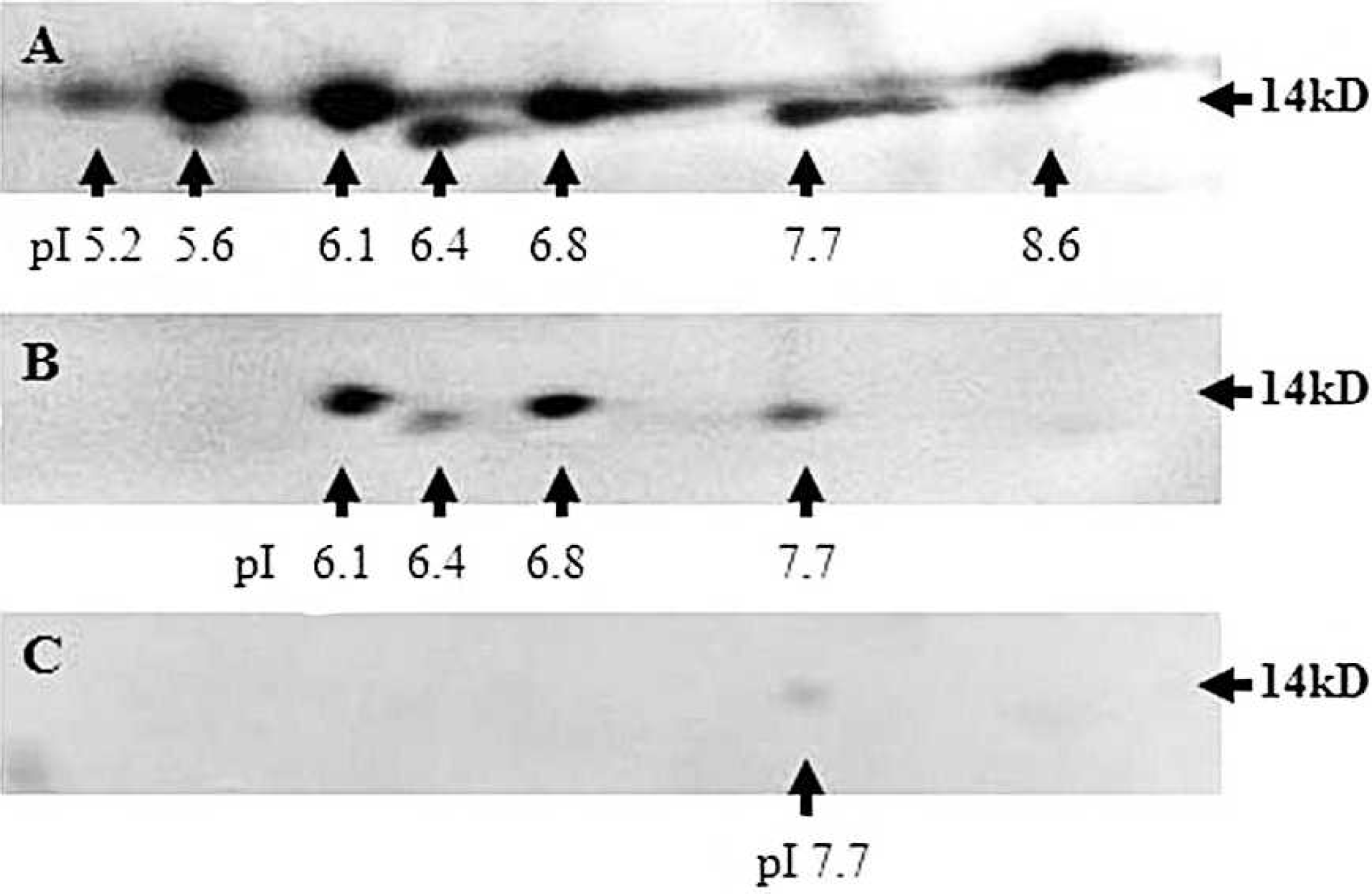
Serum amyloid A (SAA) isoforms detected by 2-dimensional electrophoresis and immunoblotting. SAA isoform patterns of
Serum AA concentrations and isoform patterns of cows are shown in Table 1. Serum AA concentrations of cows with amyloidosis ranged from 13.0 to 225.8 μg/ml (mean = 126.6 μg/ml), and SAA concentrations of cows with chronic inflammation ranged from 73.2 to 298.2 μg/ml (mean = 172.6 μg/ml). Serum AA concentrations of the 2 healthy cows were both less than 0.3 μg/ml.
A total of 7 SAA isoforms were characterized in the serum of cows with amyloidosis (isoelectric point [pI] 5.2, 5.6, 6.1, 6.4, 6.8, 7.7, and 8.6). These isoforms, with the exception of SAA pI 5.2 and 8.6, were detected in each serum sample, while SAA pI 5.2 (4 samples) and 8.6 (9 samples) were detected only in some of the amyloidosis serum samples. Figure 1A shows the SAA isoform pattern of sample 6.
The same 7 isoforms were found in the serum of cows with chronic inflammation. However, only SAA isoforms pI 6.4, 6.8, and 7.7 were present in each serum sample, while SAA pI 5.6 (6 samples) and 6.1 (9 samples) were detected in only some of the samples. Moreover, SAA pI 5.2 (1 sample) and 8.6 (2 samples) were infrequently detected. Figure 1B shows the SAA isoform pattern of sample 15. In contrast, only SAA pI 7.7 was detected as a faint spot in sample 21 in serum from a healthy cow (Fig. 1C). The results of the present study indicate the lack of an SAA isoform specific to bovine amyloidosis.
The frequency of each SAA isoform found in cows with amyloidosis and chronic inflammation was compared by Fisher's exact test. The frequency of SAA pI 8.6 in cows with amyloidosis was significantly (P = 0.0027) higher than that in cows with chronic inflammation. The frequency of SAA pI 5.2 in cows with amyloidosis seemed to be higher than that in cows with chronic inflammation; however, the difference was not significant (P = 0.1517). Although SAA pI 5.2 and 8.6 appear to be specific markers for amyloidosis, it is possible that the findings are not unique for amyloidosis. More cases of chronic inflammation and amyloidosis should be analyzed to confirm or refute this hypothesis. Moreover, the metabolic acid/base status of the cows used in the current study might influence the isoelectric position of SAA and should be considered in future studies. Furthermore, only 2 healthy cows were used as negative controls in the present study; it would be preferable to have more control animals to confirm the specificity of the SAA isoform patterns in future studies.
In mice and minks, SAA2 is the isoform that tends to cause amyloid formation (i.e., the amyloidogenic isoform), 5,9 while in humans, SAA1 and SAA2 are both amyloidogenic isoforms. 2 In cows, only 1 amyloidogenic isoform is thought to exist. 18 Based on the results of the present study, both SAA pI 5.2 and 8.6 appear to be potential amyloidogenic isoforms. To confirm this, it will be necessary to determine the amino acid sequence of each isoform and to assess the homology with amino acid sequences of AA proteins deposited in organs.
Acknowledgements. This work was supported in part by a Grant-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (No. 20658072) and an Education and Research Grant under the President's Discretion, Obihiro University of Agriculture & Veterinary Science.
Footnotes
a.
Tridelta Development Ltd., Maynooth, County Kildare, Ireland.
b.
MorphoSys AG, Martinsried/Planegg, Germany.
c.
Ettan™ IPGphor™, GE Healthcare Bio-Science, Tokyo, Japan.
d.
Mini-PROTEAN® 3, Bio-Rad Laboratories, Hercules, CA.
e.
ECL™, GE Healthcare Bio-Science, Japan.
