Abstract
A previously reported Erysipelothrix-specific polymerase chain reaction (PCR) was used to detect Erysipelothrix bacteremia in chickens. The sensitivity of PCR using 3 DNA extraction methods (boiling method, commercial gene matrix, and DNA extractor kit) was compared by using a serial 10-fold dilution of a chicken isolate of Erysipelothrix rhusiopathiae strain in chicken blood. Of the techniques used, the DNA extractor kit, followed by PCR, provided the most sensitive method for the detection of the E. rhusiopathiae strain in chicken blood (approximately 100 CFU/0.1 ml of blood). Two E. rhusiopathiae infection experiments were then attempted. In a total of 10 inoculated chickens, bacteremia developed in 9 chickens, consisting of all 5 chickens used in the first trial (ranging from 5.1 × 101 to 2.0 × 103 CFU/0.1 ml of blood) and 4 of the 5 chickens used in the second trial (ranging from 1.0 × 100 to 3.3 × 102 CFU/0.1 ml of blood). In the second trial, the 3 detection techniques were applied to the chickens with bacteremia, and the organism could be detected by using the DNA extractor kit in blood specimens from the 3 chickens exhibiting bacteremia of ≥4.2 × 101 CFU/0.1 ml of blood. This observation suggests that most E. rhusiopathiae–infected chickens develop more critical bacteremia than the detectable level by PCR with the DNA extractor kit, and the PCR detection method can be used as a first-line screening of avian erysipelas.
Avian erysipelas is an acute septicemic disease characterized by generalized parenchymal necrosis and hemorrhage. It is caused by the small Gram-positive bacterium Erysipelothrix rhusiopathiae, which is also responsible for swine erysipelas, other infectious diseases in a wide variety of animal species, and erysipeloid in humans. 5 The genus Erysipelothrix contains 2 main species: E. rhusiopathiae and E. tonsillarum. The latter can be distinguished from the former by its fermentation of saccharose, serology, and its lack of pathogenicity in pigs and chickens. 20,23 The primary economic importance of erysipelas is as a disease of turkeys, which seem to be particularly susceptible to infection with E. rhusiopathiae. 5 It occurs sporadically in several countries, including the United States. 2,8 Economically significant outbreaks in chickens have also occasionally occurred in Japan 16,18 and other countries. 4,12,15,25 It is reported that, in Japan, 5.5% of chicken sera examined had high titers of growth agglutination antibody against E. rhusiopathiae, 22 implying that the bacterium could potentially be present among chickens in the field. Erysipelas in the poultry industry might therefore be an animal hygiene issue to be controlled by turkey and chicken farmers.
Sensitivity of polymerase chain reaction detection of Erysipelothrix rhusiopathiae in chicken blood with DNA templates extracted using 3 different techniques.*
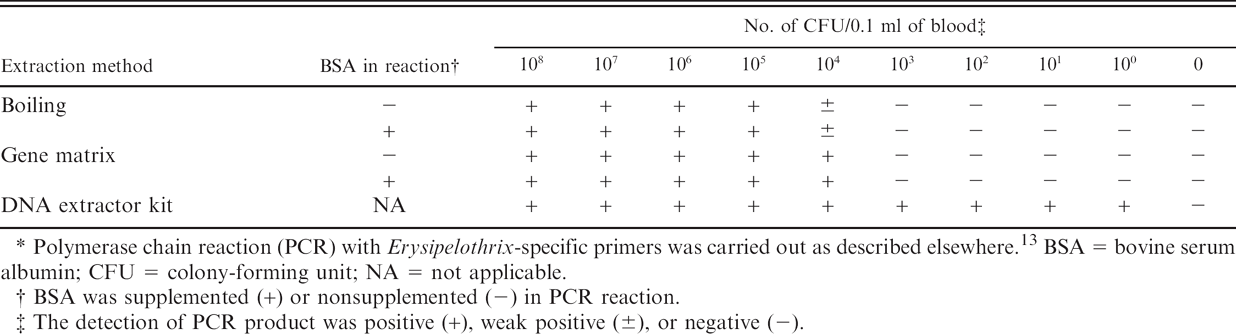
Polymerase chain reaction (PCR) with Erysipelothrix-specific primers was carried out as described elsewhere. 13 BSA = bovine serum albumin; CFU = colony-forming unit; NA = not applicable.
BSA was supplemented (+) or nonsupplemented (-) in PCR reaction.
The detection of PCR product was positive (+), weak positive (±), or negative (-).
In veterinary medicine, clinical diagnosis of avian erysipelas is difficult because the clinical signs can resemble those of other infectious diseases, such as fowl cholera, Escherichia coli infections, salmonellosis, and Newcastle disease. 5 Most field cases of avian erysipelas 8,15,18 have therefore been diagnosed based on postmortem inspection, including pathological examination and the identification of E. rhusiopathiae isolated from the lesions. However, these diagnostic methods cannot be performed antemortem, and confirmation of diagnosis is a slow process. The development of an efficient and rapid method for antemortem diagnosis is therefore needed to assist a prompt management of avian erysipelas in the field. The use of the polymerase chain reaction (PCR) in microbial diagnostics is a valuable alternative to time-consuming traditional methods. Previously, an Erysipelothrix-specific PCR method was developed based on the bacterial DNA sequence encoding its 16S ribosomal RNA. 11 The application of this method in assisting diagnosis of swine erysipelas has already been demonstrated. 21 A vian erysipelas was also diagnosed using this PCR method, with DNA extracted from formalin-fixed, paraffin-embedded liver tissue of pheasants, 7 indicating that this method was suitable for the diagnosis of avian erysipelas using tissues. In the present study, the applicability of the Erysipelothrix-specific PCR method to detection of E. rhusiopathiae in chicken blood was examined for the rapid antemortem screening of avian erysipelas in domestic birds.
E. rhusiopathiae strain Sayo-SP belonging to serotype 1a, which was isolated from chickens affected with avian erysipelas and confirmed to be pathogenic for chickens by experimental infection, 23 was used in the current study. Different techniques for the extraction of E. rhusiopathiae DNA from blood samples were compared in a preliminary in vitro study. A 10-fold serially diluted suspension of Sayo-SP strain was prepared from approximately 108–100 colony-forming units (CFU)/0.1 ml by diluting fresh cultures of the strain with ethylenediamine tetra-acetic acid (EDTA)-treated blood from specific pathogen–free (SPF) chickens. EDTA was used as an anticoagulant for the blood samples because E. rhusiopathiae has the ability to clot sodium citrate–treated plasma, 24 and heparin has been thought to act as a PCR inhibitor. 9
Each diluted suspension was centrifuged for 40 sec at 1,700 × g for the removal of erythrocytes, and then 100 μl of the supernatant was centrifuged for 10 min at 10,000 × g. After removal of the supernatant, the cell pellets were subjected to 3 different DNA extraction procedures: 1) DNA extraction by boiling was carried out by resuspending the pellet in 100 μl of autoclaved distilled water and incubating the cell suspension for 15 min at 100°C, then centrifuging it for 10 min at 10,000 × g. 2) DNA extraction using a commercial gene matrix a was carried out by adding 100 μl of the matrix to the cell pellet; the remainder of the process was performed according to the manufacturer's instructions. 3) DNA extraction using a commercial DNA extractor kit b was carried out according to the manufacturer's instructions, after adding the lysis solution. Harvested DNA pellets were dissolved in 3 μl of TE buffer (10 mmol Tris-HCl [pH 8.0], 1 mmol EDTA). Finally, 1 μl of each supernatant obtained from the first or second procedure, or 1 μl of solution from the third procedure, was used as a template for PCR with genus-specific primer sets. 11 Briefly, reaction mixtures were prepared in a volume of 25 μl containing 2.5 μl 10× Taq DNA polymerase buffer c ; 1.5 mmol MgCl2; 200 μmol each 2′-deoxyadenosine-5′-triphosphate, 2′-deoxycytidine-5′-triphosphate, 2′-deoxyguanosine-5′-triphosphate, and 2′-deoxythymidine-5′-triphosphate; 50 pmol (each) primers; 2.5 U of Taq DNA polymerase d ; and 1 μl of template DNA. DNA amplification was performed according to the previous protocol. 11 Amplified 407-bp DNA products were separated on 1.5% (wt/vol) agarose gels and stained with ethidium bromide.
Preliminary experimental results on Erysipelothrix rhusiopathiae bacteremia in 5 inoculated chickens.*

CFU = colony-forming unit; dpi = day postinoculation.
Two E. rhusiopathiae infection experiments using chickens were attempted in this study. The first infection trial was conducted by using five 3-week-old SPF white leghorn chickens to preliminarily verify the level of bacteremia. All chickens were confirmed by blood culture to be free of Erysipelothrix spp. before inoculation. Each chicken was inoculated intramuscularly with 0.5 ml of the fresh Sayo-SP strain culture (approximately 0.5 × 10 9 CFU per chicken), which was prepared according to a previous study. 23 After inoculation, the chickens were observed for clinical signs every day for 12 days postinoculation (dpi). To confirm the presence of bacteremia caused by infection of E. rhusiopathiae, the chickens were bled on 2, 3, 5, and 12 dpi, and 0.1-ml aliquots of blood, anticoagulated with 10 mg/ml EDTA, were cultured by pouring into tryptose phosphate agar medium containing 0.1% Tween 80 and 0.75% agar. After incubation for 48–72 hr at 37°C, colonies were counted, and biochemical and serotype identification was conducted. All chickens were euthanized on 12 dpi and were subjected to necropsy and macroscopic examination. In the second trial, five 7-week-old SPF white leghorn chickens were used for PCR detection of E. rhusiopathiae associated with bacteremia. This trial included the following procedures in addition to those in the first trial. Blood specimens collected on 2, 3, 5, and 12 dpi were subjected to the 3 DNA extraction techniques (boiling, the gene matrix, and DNA extractor kit methods), followed by PCR detection of E. rhusiopathiae, according to the protocols described above. Levels of latex-agglutination (LA) serum antibody at both times of preinoculation and death were determined using an available commercial kit. e Both infection experiments were conducted in accordance with the Japan National Veterinary Assay Laboratory Guide for the Care and Use of Laboratory Animals and the Animal Welfare Acts.
Table 1 shows the PCR detection sensitivity with substrate DNA prepared by the 3 DNA extraction techniques. The boiling and gene matrix extraction methods did not result in good DNA recovery, with a detection limit of ≥approximately 104 CFU/0.1 ml when applied to the detection of E. rhusiopathiae in avian blood. In addition, these detection sensitivities were not improved even when PCR was reperformed by adding bovine serum albumin (400 ng/μl reaction), which can relieve the inhibition induced by substances such as hemin in blood, as described elsewhere 10 ; this may possibly be explained by removing erythrocyte as a first step for preparing the sample (i.e., centrifugation of 40 sec at 1,700 × g). In the boiling and gene matrix extraction methods, cell lysis and DNA release are achieved by heat treatment. In addition, the gene matrix can absorb cell lysis products that interfere with the PCR amplification. Although these extraction methods are simplified, the present results suggest that these methods are not suitable for detecting low numbers of E. rhusiopathiae in chicken blood specimens. DNA extraction using the DNA extractor kit resulted in the high sensitive detection of bacteria in avian blood (≥approximately 100 CFU/0.1 ml of blood), which is substantially superior to the other 2 methods, even allowing for approximately 30 times smaller volumes of the final elution made by the kit (3 μl) than that made by the other 2 extraction methods (100 μl). The advantage of the DNA extractor kit, compared with the other 2 extraction techniques, has been also demonstrated in the authors' previous study regarding the detection of E. rhusiopathiae in porcine joint fluid. 21 The method of cell lysis used by the DNA extractor kit is based on consecutive reactions with surfactant, protease, and sodium iodide. The present study demonstrated that the DNA extractor kit greatly improved the sensitivity of PCR detection of E. rhusiopathiae in blood specimens, although the procedure was more time-consuming than extraction by boiling or using the gene matrix.
In the in vivo study using a total of 10 chickens experimentally inoculated with the E. rhusiopathiae strain, bacteremia developed on 2, 3, or 5 dpi in 9 chickens, consisting of all of the 5 chickens used in the first trial (Table 2) and 4 of the 5 chickens used in the second trial (Table 3). In the second trial, although the PCR was negative in all blood specimens when DNA was extracted by boiling and the gene matrix methods, PCR assay using DNA extracted with the DNA extractor kit detected E. rhusiopathiae in the blood specimens collected from the 3 chickens on 2 or 3 dpi, which contained ≥4.2 × 101 CFU/0.1 ml of blood. In addition, in the first trial, the higher level of bacteremia was observed in all 5 chickens (ranging from 5.1 × 101 to 2.0 × 103 CFU/0.1 ml of blood), and their bacteremia is estimated to have been detected by the PCR with the DNA extractor kit. Thus, it is possible that the PCR assay with the DNA extractor kit will be able to substantially detect E. rhusiopathiae in most bacteremic chickens (e.g., a total of 8 of the 9 bacteremic chickens in the 2 trials). In contrast, by the boiling and the gene matrix methods, all of the 5 bacteremic chickens in the first trial could have been missed, in addition to the result of the second trial, because the bacteremia in these chickens did not reach the detectable level by the 2 extraction methods (≥approximately 104 CFU/0.1 ml). Thus, the DNA extractor kit will contribute to the greatest sensitivity (an estimated approximately 90%) for PCR detection of E. rhusiopathiae associated with bacteremia among the 3 extraction methods.
Polymerase chain reaction (PCR) detection of Erysipelothrix rhusiopathiae in blood samples obtained from 5 experimentally inoculated chickens.*
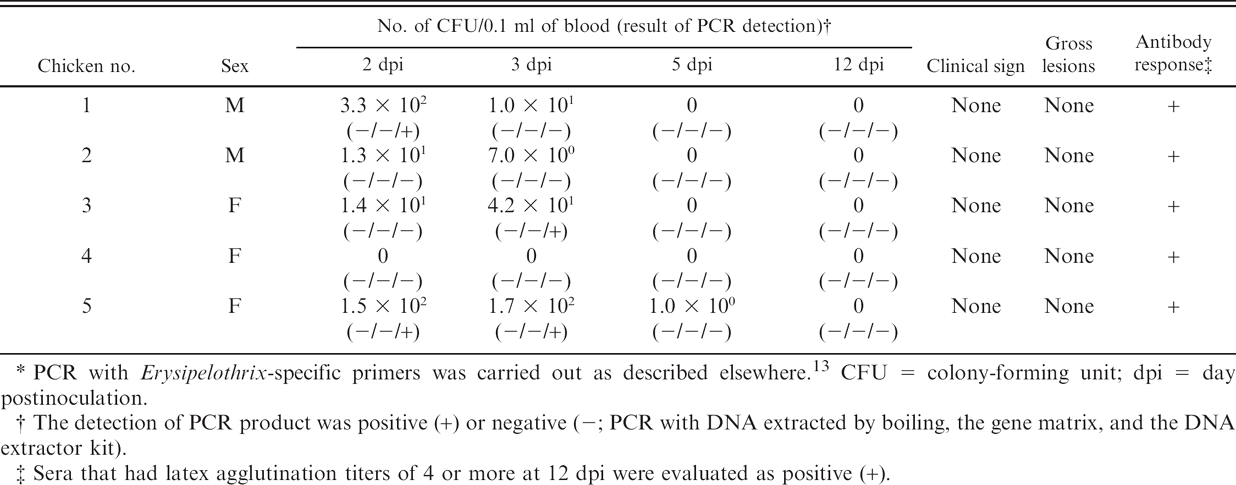
PCR with Erysipelothrix-specific primers was carried out as described elsewhere. 13 CFU = colony-forming unit; dpi = day postinoculation.
The detection of PCR product was positive (+) or negative (-; PCR with DNA extracted by boiling, the gene matrix, and the DNA extractor kit).
Sera that had latex agglutination titers of 4 or more at 12 dpi were evaluated as positive (+).
In the present study, bacteremia persisted only until 3 or 5 dpi in all of the 9 bacteremic chickens. Likewise, it was previously confirmed that bacteremia first appeared on 0.5 dpi and had largely disappeared by 5 dpi in E. rhusiopathiae–infected chickens. 17 The application of the DNA extractor kit described in the current study could therefore assist the PCR screening of avian erysipelas, particularly during the initial phase of infection.
The PCR assay with the DNA extractor kit was not able to detect E. rhusiopathiae of 1.0 × 100 to 1.4 × 101 CFU/0.1 ml in blood specimens collected from infected chickens, although in the preliminary in vitro experiment, the PCR assay had a detection sensitivity of approximately 100 CFU/0.1 ml. All of the studied chickens developed LA antibodies (titers of ≥4 titers), suggesting that an immune response was induced by the inoculation of the E. rhusiopathiae strain. It has been reported that immunoglobulin G 1 and leukocyte DNA, 13 both of which contribute to the immune system in blood, can act as significant PCR inhibitors. Thus, nondetection of E. rhusiopathiae associated with mild bacteremia could have been due to the presence of PCR inhibitors such as immunoglobulins and leukocytes in the blood, resulting from activation of the immune system by E. rhusiopathiae. In the current study, none of the chickens showed any clinical signs or developed gross lesions despite the occurrence of bacteremia, indicating that the chickens were infected latently. It has been, however, previously reported that the bacterial numbers in the blood reached approximately 102 to 103 CFU/0.1 ml in at least half of the E. rhusiopathiae–infected chickens with clinical signs. 23 This previous finding implied that the level of bacteremia in chickens with clinical signs could practically exceed the detectable level by the PCR assay with the DNA extractor kit. In addition, it has been found that E. rhusiopathiae must reach extremely high blood concentrations (106–107 CFU/0.1 ml) before any clinical signs occur in experimentally infected turkeys. 3 Incipient avian erysipelas accompanied by bacteremia could therefore be largely detected by the PCR assay with the DNA extractor kit, although the detection sensitivity may be affected by the presence of PCR inhibitors. The detection sensitivity for E. rhusiopathiae in avian blood could be further improved by using larger volumes of blood sample or by combining it with cultivation in enrichment broth. 19
The Erysipelothrix-specific PCR used in the current study could, in principle, detect not only E. rhusiopathiae but also E. tonsillarum because both nucleotide sequences of the target fragments are exactly identical and can be amplified with the primers. 11 However, E. tonsillarum has never been found in poultry, even in a previous study based on a large collection of Erysipelothrix isolates of diverse origins. 20 It has also been confirmed by experimental infection that E. tonsillarum is avirulent in chickens, 23 which suggests that any Erysipelothrix species detected in avian blood by this PCR method is more likely to be E. rhusiopathiae than E. tonsillarum.
In conclusion, the DNA extractor kit made possible the sensitive PCR screening of avian erysipelas accompanied with bacteremia depending on the level of bacteremia and the time of testing. Avian erysipelas usually occurs in mass outbreaks, 5 and thus it is expected that many samples will be able to be subjected to the PCR detection. This fact would further decrease the risk of missing the occurrence of avian erysipelas in the flocks. The PCR detection with bacterial DNA extracted by the DNA extractor kit, as described in the current study, can be performed rapidly with minimal blood samples and could thus provide a firstline screening test for a prompt management of avian erysipelas ahead of further detailed inspections for the definitive diagnosis.
It is well known that turkeys are the species most commonly affected with E. rhusiopathiae, and thus, in countries with a thriving turkey industry such as the United States, avian erysipelas is one of the problems in animal hygiene. 14 Although in Japan, turkeys are treated as a minor class of livestock, 6 turkey farming has been recently encouraged by the national promotion program in the administration of livestock industry. The growth agglutination antibody test is available for screening methods of avian erysipelas in Japan. 22 In the United States, inactivated vaccines have been widely used for the prevention of avian erysipelas, 5 and therefore, it is hoped that the present PCR detection method using the DNA extractor kit, in addition to the serological test, will help in the screening of the Erysipelothrix infection in turkeys.
Footnotes
a.
InstaGene™ Matrix, Bio-Rad Laboratories, Hercules, CA.
b.
DNA Extractor® WB Kit, Wako Pure Chemical Industries Ltd, Osaka, Japan.
c.
10× Ex Taq™ Buffer (Mg2+free), Takara Bio Inc., Shiga, Japan.
d.
Takara Ex Taq®, Takara Bio Inc., Shiga, Japan.
e.
“Nisseiken” Agtex SE, Nisseiken Co. Ltd., Tokyo, Japan.
