Abstract
Determination of serum or plasma progesterone (P4) concentrations is important to recognize pregnant and non-pregnant ewes, and also to predict the number of carried lambs. The 2 most common methodologies for the detection of plasma P4 are radioimmunoassay (RIA) and enzyme immunoassay (EIA). RIA is very expensive, and not all laboratories are equipped to perform this test; EIA is commercially available for human use, but only a few companies produce species-specific kits, which are expensive. We verified for ovine plasma a less expensive and easily available ELISA kit (DiaMetra) designed to quantify P4 in humans. Pools of ovine and human plasma were used to compare repeatability, accuracy, sensitivity, and stability of P4 measured by the DiaMetra kit. Repeatability data were within 15%, and accuracy values were ~90% for both plasma matrices. Stability data showed a loss of <20% for freeze–thaw and <30% for 30-d storage. All parameters were acceptable under international guidelines for method validation. The human ELISA kit was used successfully to quantify plasma P4 in 26 ewes during pregnancy until delivery. P4 concentrations were also correlated with the number of carried lambs.
Progesterone (P4) plays a central role in reproductive events associated with the establishment and maintenance of pregnancy in all mammals. 4 In the ewe, P4 is synthesized by the corpus luteum during the first 50 d of pregnancy, and then by the placenta until term. 6 P4 is needed during early embryonic development for implantation and placentation; insufficient P4 concentrations can lead to failure of pregnancy and early embryonic loss. 5 P4 concentrations in plasma have been used not only to determine the pregnancy status of the animal but also to predict the number of fetuses by monitoring the dam in the second half of the pregnancy. 8 Thus, the determination of serum or plasma P4 concentrations in sheep is important not only for the early diagnosis of pregnancy, but also to evaluate the number of carried lambs by means of a significant positive correlation with the P4 concentration. 2 Many laboratories use radioimmunoassays (RIAs) for the quantification of plasma P4,8,10,11 but RIA, although very sensitive, is expensive and requires the availability of specialized laboratories and personnel.
Some laboratories have used an enzyme immunoassay (EIA) for determination of P4 in plasma. Most of the commercial kits for P4 assay are validated for use in humans. More recently, several companies have begun marketing P4 ELISA kits specific for ovine plasma.1,2 However, these species-specific kits are ~5-fold more expensive than the kits for the human P4 assay. In our laboratory, in which large numbers of samples are assayed routinely, their use would be uneconomical.
We aimed to verify a P4 ELISA kit, developed for human plasma, to monitor P4 concentrations in ovine plasma. As reported by the manufacturer, the lower and upper limits of detection are 0.05 ng/mL and 40 ng/mL respectively, which are suitable for our manufacturer, the lower and upper limits of detection are 0.05 ng/mL and 40 ng/mL respectively, which are suitable for our analytical purpose. All procedures involving animals in our study were approved by the local Animal Care and Use Committee (authorization 2899 of 17/01/2018).
For method verification, we used five 4–5-y-old Sarda ewes at the end of the seasonal anestrus period. The ewes were penned outdoors, with access to a sheltered area, at the experimental facilities of the Department of Veterinary Medicine at the University of Sassari, Italy. Jugular vein blood samples were collected using 10-mL K2EDTA vacuum collection tubes (Becton Dickinson). Human blood samples were collected from 5 healthy volunteers (women, 60–65 y old, in menopause, without the use of any hormonal therapy) using 10-mL K2EDTA vacuum collection tubes. Blood was centrifuged (500 × g, 10 min, 4°C), and plasma was collected and pooled from the ewes and the women.
A human P4 ELISA kit (DiaMetra) was used to assay P4 in sheep and human plasma using a microplate reader (POLARstar Omega; BMG Labtech), with BMG Labtech software for data analysis. The assay is a competitive ELISA in which the P4 (antigen) in the sample competes with the horseradish peroxidase (HRP)-conjugated P4 supplied in the kit for binding to P4 antibodies coated onto the ELISA plate wells. Quality control reagents provided by the manufacturer with the kit were used to verify the performance of the assay.
The pool of ovine plasma and the pool of human plasma were tested for endogenous P4 using the DiaMetra ELISA kit. For both pools, the concentration of endogenous P4 was below the 0.05 ng/mL detection limit. The pools of plasma were designated as blank ovine plasma (BOP) and blank human plasma (BHP). Next, P4 standards of 4 ng/mL and 20 ng/mL were created by dissolving appropriate amounts of P4 (Merck) in aliquots of BOP, BHP, and distilled water to obtain a positive control for each matrix. The controls at 4 ng/mL were designated control water 1 (CW1), control ovine plasma 1 (COP1), and control human plasma 1 (CHP1); similarly, the controls at 20 ng/ml were designated CW2, COP2, and CHP2.
The precision of the method, evaluated at the 2 concentrations in both plasma matrices, was expressed as the percent relative standard deviation (RSD%) and calculated for 5 replicates for intra-day repeatability and over 3 consecutive days for inter-day repeatability (Table 1; Suppl. Tables 1, 2). The repeatability data were within 15%, as required in the guidelines for method validation. 12 Given that BOP and BHP were below the limit of detection, the accuracy, which represents the closeness of the test results to the true values, was determined for 5 replicates using the following formula:
Repeatability, accuracy, and stability of progesterone (P4) measurement in ovine and human plasma.
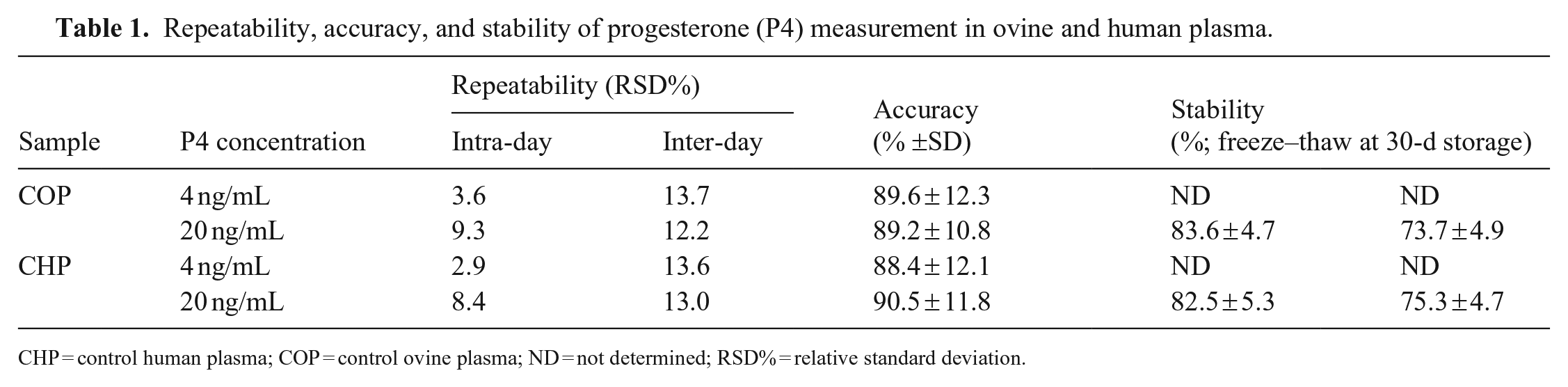
CHP = control human plasma; COP = control ovine plasma; ND = not determined; RSD% = relative standard deviation.
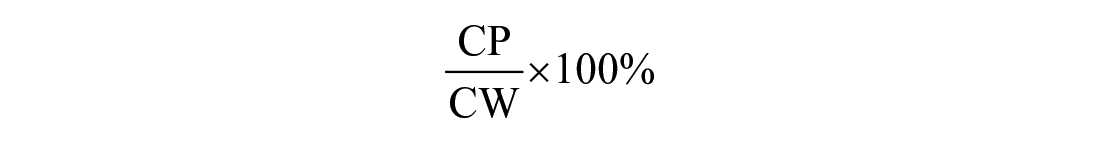
where CP indicates COP or CHP in ovine and human plasma, respectively. The kit had good accuracy of 88.4–90.5% for human plasma and 89.2–89.6% for ovine plasma (Table 1). Hence, the tested ELISA is as accurate in ovine plasma as in human plasma.
The freeze-and-thaw stability of P4 in the samples was determined after performing 3 freeze–thaw cycles on COP and CHP samples at the highest tested concentration (Table 1; Suppl. Table 3). COP and CHP samples (n = 5) were first analyzed as fresh, and then frozen at –20°C for 24 h and thawed for 1 h at room temperature. This freeze–thaw cycle was performed 3 times.
Freeze–thaw loss of P4 was <20% (Table 1; Suppl. Table 3). The 30-d stability of P4 in frozen plasma showed a loss in concentration of <30% (Table 1; Suppl. Table 4). Long-term stability is an important factor because, for our laboratory, it is often necessary to store samples of ovine plasma at –20°C for up to a month before analysis.
The DiaMetra ELISA kit, although designed for human plasma, was found to be suitable for the determination of P4 concentrations in ovine plasma. Moreover, the kit was checked for possible cross-reactivity with other progestogens (testosterone, 17αOH-progesterone), and we found no cross-reactivity in ovine plasma.
We used this assay to quantify P4 in 26 healthy ewes during their 5-mo pregnancy until delivery. Jugular vein blood was drawn from the ewes and stored at –20°C until analysis. Pregnancy occurred after natural breeding following cycle synchronization with intravaginal pessaries impregnated with progestogens (20 mg of fluorogestone acetate; Chronogest CR, MSD-Animal Health) for 12 d plus a single IM injection of 200 IU of equine chorionic gonadotrophin (eCG; Folligon, MSD-Animal Health), concurrent with pessary removal. The day of mating was considered day 0 for experimental purposes. At day 24, pregnancy diagnosis and initial fetal counts were conducted by transrectal ultrasonography, with a real-time B-mode scanner (SSD 500; Aloka) fitted with a 7.5-MHz linear-array probe. The number of carried lambs was confirmed at parturition. Blood sampling and ultrasound scanning were performed on the same day in all ewes. To minimize variables, we analyzed all plasma samples for our experiment on the same day, with the same equipment, same laboratory personnel, and same ELISA kit batch (Fig. 1; Suppl. Table 5).
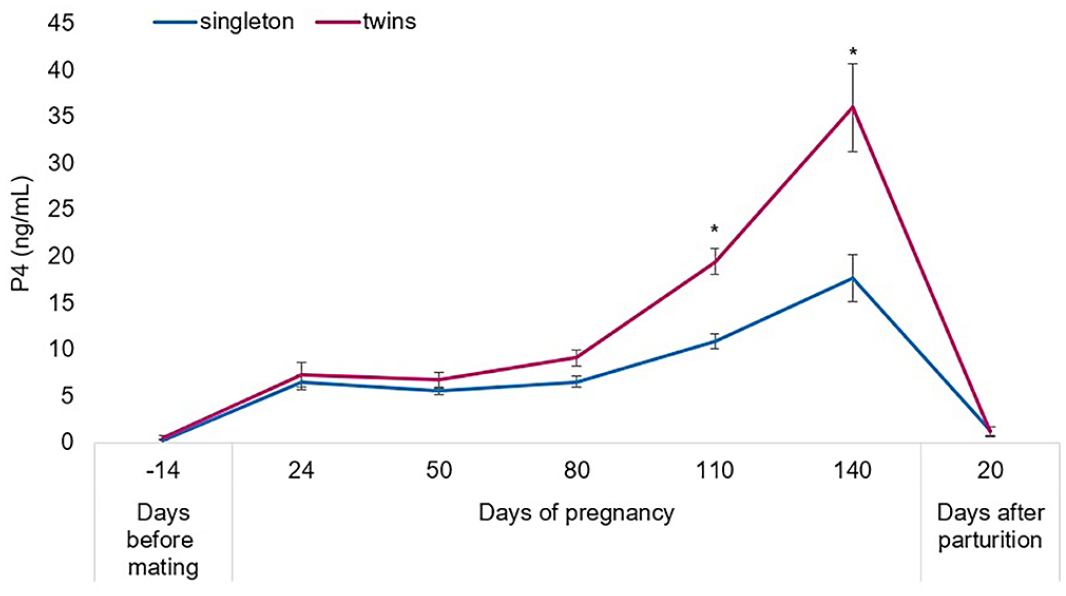
Use of the DiaMetra human progesterone (P4) ELISA kit to measure ovine P4 before, during, and after pregnancy. P4 quantification in 26 ewes from before pregnancy onset until delivery (values are means ± SEM). *Statistical differences from pregnant ewes bearing a single fetus (p < 0.001).
Differences were analyzed by a monofactorial ANOVA (Minitab v.18.1). To discriminate between the means, we used the Fisher least-significant-difference procedure. All results were expressed as mean ± SEM, and a probability of p < 0.01 was considered to be significant.
The P4 trend in ewes bearing one fetus was compared with ewes with twin fetuses. For both ewe groups, the P4 concentration started from a very low value before pregnancy, increased with progression of pregnancy, and reached the maximum concentrations in the fifth month (day 140); P4 then decreased to undetectable values after delivery, as reported elsewhere. 3 During the second half of pregnancy, when P4 is produced mainly by the placenta, the P4 concentration was significantly higher in ewes carrying twins versus singletons (Fig. 1; Suppl. Table 5). Plasma P4 concentrations in ewes with 2 fetuses were approximately double those bearing 1 fetus. This evidence can be explained by the presence of 2 placentas in ewes with 2 fetuses, as reported by others. 9 Moreover, there was a significant positive correlation during the second half of pregnancy at 80 (p < 0.01), 110 (p < 0.001), and 140 d (p < 0.001), between P4 concentrations in pregnant ewes and the number of carried fetuses (singleton or twins; Table 2; Suppl. Table 5). This correlation is in accord with reports in the literature.2,7,8 P4 concentrations in nonpregnant ewes were also measured during an induced luteal phase after estrus synchronization with intravaginal pessaries impregnated with progestogens for 12 d plus a single IM injection of eCG. Blood samples were taken on days 6, 8, 10, and 17 after sponge removal. Progesterone plasma concentrations were determined with the same ELISA kit and represent normal P4 values of nonpregnant cycling sheep (Suppl. Table 6).
Progesterone ovine concentration measured using of the DiaMetra human progesterone (P4) ELISA kit before, during, and after pregnancy.

Pearson correlation coefficients (r) between number of fetuses and P4 concentration. Corresponding p-values are shown within parentheses.
Our results show that the DiaMetra human P4 ELISA kit is suitable for ovine P4 analysis. Furthermore, we used the kit successfully to confirm pregnancy diagnosis and the number of carried lambs.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387211043513 – Supplemental material for Measurement of progesterone in sheep using a commercial ELISA kit for human plasma
Supplemental material, sj-pdf-1-vdi-10.1177_10406387211043513 for Measurement of progesterone in sheep using a commercial ELISA kit for human plasma by Valeria Pasciu, Maria Nieddu, Elena Baralla, Cristian Porcu, Francesca Sotgiu and Fiammetta Berlinguer in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our experimental work was supported by funds from Regione Autonoma della Sardegna—Progetti ricerca fondamentale o di base—L.R. 7/2007—annualità 2013 (CRP 78167).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
