Abstract
Helcococcus ovis is a newly established species in the genus Helcococcus. The clinical significance of this organism in sheep has not been reported. In the current report, isolation of H. ovis from a 6-month-old mixed-breed ewe lamb that died of respiratory disease is described. Pathologic examination revealed severe, focally extensive, chronic necrotizing pleuritis with intralesional coccobacilli and mild, multifocal, subacute mucopurulent bronchopneumonia, indicating a bacterial etiology. A Gram-positive bacterium was isolated in heavy growth from the lung tissue. DNA sequence analysis on the 16S rDNA gene demonstrated that the isolate was H. ovis. To the authors' knowledge, this is the first report of isolation of H. ovis associated with infection in sheep with pleuritis and bronchopneumonia.
Keywords
Helcococcus kunzii was created as a new genus and species in 1993 to accommodate some uncommon catalase-negative, Gram-positive cocci of unknown taxonomic position isolated from human clinical sources. 3 The organism is a facultatively anaerobic coccus. 3 Helcococcus ovis was added to the genus in 1999 as a new species 5 and was originally isolated from sheep in mixed cultures from animals from different geographical locations. 5 Since then, H. ovis has been reported to be associated with valvular endocarditis in cattle 10 and in a horse from a pulmonary abscess. 12 However, the pathogenic role of this bacterium in sheep has never been established. To the authors' knowledge, the present study is the first report of isolation of H. ovis from a sheep with pleuritis and bronchopneumonia.
A flock of 80 lambs assembled from auctions and other farms experienced an outbreak of acute respiratory disease that resulted in death in 4–5 days. Morbidity was approximately 7%, and 3 animals died. A 6-month-old mixed-breed ewe lamb died 4 days after showing respiratory signs. Fresh and formalin-fixed lung tissues were submitted to the Ohio Department of Agriculture Animal Disease Diagnostic Laboratory (Reynoldsburg, OH). The referring veterinarian reported that a large amount of pleural fluid and severe consolidation of the lungs with hemorrhages were grossly visible on necropsy. Histopathologic examination of the submitted lung sample showed expansion of the pleural connective tissue along one aspect of the section as a result of an increased amount of fibrous connective tissue heavily infiltrated by mixed mononuclear cells including lymphocytes, histiocytes, and a few plasma cells with infrequent neutrophils (Fig. 1). The pleural surface contained a dense linear zone of necrotic round cells with pyknotic nuclei admixed with abundant karyorrhectic debris and many very small coccobacilli (Fig. 2). The parenchyma of the lung was markedly collapsed (atelectasis). A small number of bronchi and bronchioles contained viable and degenerate neutrophils in the lumen, admixed with amphophilic fibrillar material (mucin), and a few small foci of degenerate neutrophils were present in some alveoli (Fig. 3). Light cuffing of airways and vessels by lymphocytes and plasma cells was noted, as was marked congestion. Gram stain of the lung section revealed large numbers of Gram-positive cocci within the exudate adherent to the pleural surface (Fig. 4). The morphologic diagnoses of severe, focally extensive, chronic necrotizing pleuritis with intralesional bacterial cocci and mild, multifocal, subacute mucopurulent bronchopneumonia, supported by Gram stain results, were indicative of a bacterial etiology.
Lung tissues were inoculated onto Trypticase Soy Agar II plates containing 5% sheep blood. a The plates were incubated at 37°C in both aerobic and 7% CO2 atmosphere. Aerobic culture yielded few Nocardia spp. and Bacillus spp. within 24 hr. After 6 days of incubation in CO2, heavy and pure growth of pinpoint colonies satelliting around the streaked Staphylococcus aureus was recovered. The colonies were pinpoint, shiny, grayish, convex, and circular with no hemolysis. Examination of a Gram-stained smear of the culture showed Gram-positive cocci, occurring singly, in pairs, or in short chains. The organism was negative for catalase and oxidase.
DNA sequence analysis of the 16S ribosomal DNA (rDNA) from the isolate was carried out using 2 sets of primers as previously described, 7 with minor modifications. Briefly, DNA samples were extracted from 2 colonies suspended in DNase- and RNase-free H2O b using a commercial extraction kit c following the manufacturer's instructions. Polymerase chain reaction (PCR) was carried out in a 100-μl volume containing 10 μl of the extracted DNA, 50 μl of Takara's 2X PCR Premix Taq kit, d 2 μl (10 pmol) of each primer, and 36 μl of DNase- and RNase-free H2O. The PCR reactions were carried out in a commercial thermal cycler e ; cycling conditions were dena-turation, 30 sec at 94°C; annealing, 15 sec at 55°C; and extension, 45 sec at 72°C. A final extension step of 5 min at 72°C was included. The PCR products were then purified using a commercial PCR purification kit f following the manufacturer's instructions. After purification, the samples were sent to the Plant Microbe Genomic Facility of The Ohio State University (Columbus, OH) for sequencing. A search of the GenBank database was carried out using the BLAST algorithm (Basic Local Alignment Search Tool; http://www.ncbi.nlm.nih.gov/blast/Blast.cgi). The 16S rDNA entries yielded a 100% match to H. ovis with both primer sets and were equally distantly related to both H. sueciensis and H. kunzii, with an overall identity of 94%.
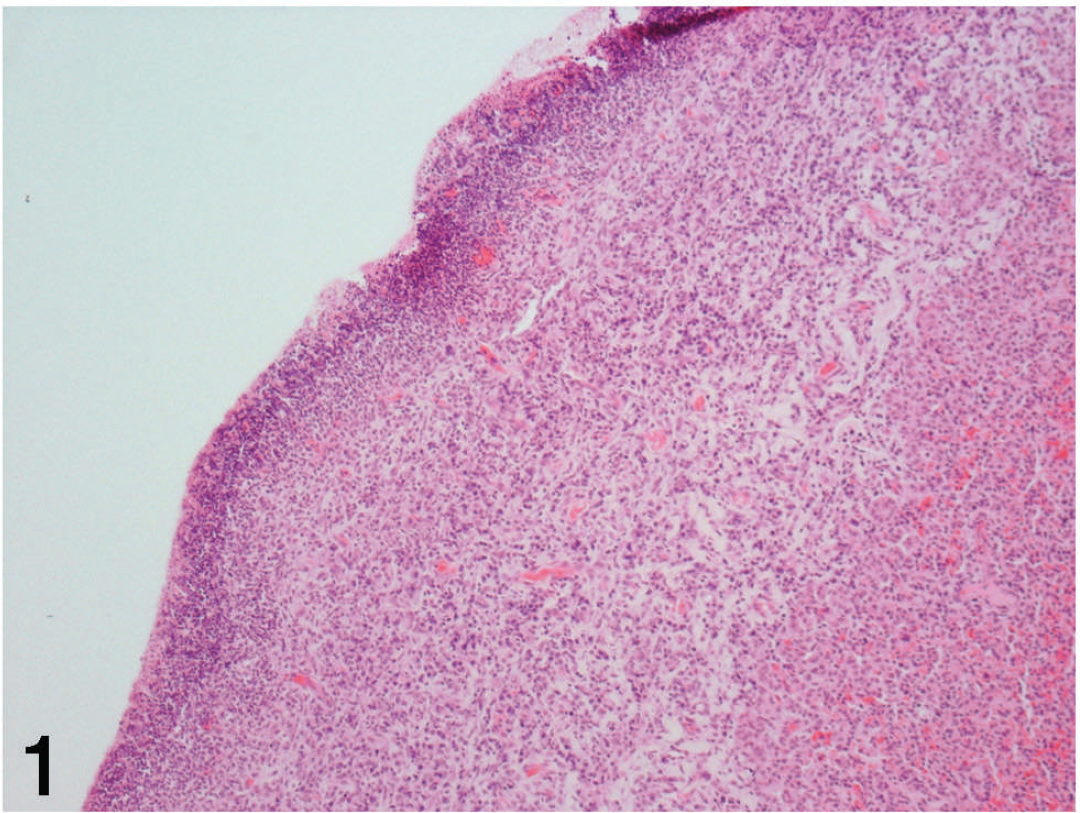
Lung; feedlot lamb. Immature fibrous connective tissue expands the subpleural interstitium and is infiltrated by mixed mononuclear cells. Note the dense linear zone of necrotic debris admixed with neutrophils and bacterial cocci near the pleural surface on the left and atelectatic lung parenchyma on the right. Hematoxylin and eosin. 100×.

Lung; feedlot lamb. Higher magnification demonstrating infiltrates of lymphocytes, plasma cells, and histiocytes within increased subpleural connective tissue and viable and degenerate neutrophils admixed with nuclear debris and bacterial cocci on the pleural surface. Hematoxylin and eosin. 200×.

Lung; feedlot lamb. Neutrophils and cellular debris are present in the lumen of a terminal bronchiole, as well as within a subjacent alveolar space. Hematoxylin and eosin. 400×.

Lung; feedlot lamb. Gram stain demonstrates the presence of large numbers of Gram-positive cocci within nuclear and cellular debris on the pleural surface. Brown and Brenn. 600×.
Currently, there are at least 4 species in the genus Helcococcus: H. kunzii, H. ovis, H. pyogenes, and H. sueciensis. 1,6,8,13 All have been isolated from clinical samples of human or animal origin. 1,6,8,13 Since its first description, H. kunzii has been isolated from various clinical diseases in human patients that range from localized infection including wound infections, abscesses, and sebaceous cysts to systemic bacteremia. 1,3,9,11,13 Helcococcus pyogenica was isolated from a patient with prosthetic joint infection, 8 and H. sueciensis was recovered from the wound infection of a female patient. 4 An unclassified Helcococcus-like organism causing endocarditis was isolated from an intravenous drug user. 13
To date, H. ovis is the only species in the genus Helcococcus of veterinary importance. The original 2 isolates were isolated from sheep from different geographic locations in the United Kingdom. 5 One isolate was recovered from the lung, liver, and spleen of a postmortem adult male sheep, in mixed culture with Arcanobacterium pyogenes. 5 The second isolate was recovered from a milk sample collected from a sheep with subclinical mastitis. The isolate was found in mixed culture with a Staphylococcus sp. on Columbia blood agar at the periphery of the staphylococcal colonies. Both of the H. ovis isolates from sheep were recovered in mixture with other organisms; the clinical significance of the organism was never confirmed. 5 However, the bacterium was subsequently reported as the primary pathogen from other animal species 10,12 in association with clinical disease. In 2003, H. ovis was reported to be isolated from a 12-month-old Angus bull with valvular endocarditis in North Carolina. 10 In 2004, the bacterium was recovered from a horse with a pulmonary abscess. 12 In the current report, the isolation of the organism in heavy and dominant growth from a sheep is described. The organism appeared to be the primary pathogen for the pleuritis and bronchopneumonia. It also appeared to be responsible for causing morbidity and mortality in the sheep flock. To the authors' knowledge, this is the first report to demonstrate the pathogenic role of this bacterium in sheep. The prevalence of this organism in the sheep population is unknown. The authors believe that the organism may be underreported because it is slow growing and can be easily missed because of the overgrowth of other fast-growing organisms in a mixed infection. In addition, it requires CO2 and Staphylococcus sp. for initial isolation. Moreover, it is biochemically inert, which makes its identification difficult using conventional techniques. The only reliable method currently available for correct identification is by 16S rDNA gene sequence analysis. Helcococcus ovis should be considered in the differential diagnosis for ovine respiratory diseases, especially in cases with pleuritis.
Acknowledgements. The authors thank Scott Fox and Shawn Smith for histologic technical assistance.
Footnotes
a.
Trypticase™ Soy Agar II with 5% Sheep Blood (TSA II), BD Diagnostic Systems, Sparks, MD.
b.
DNase- and RNase-free H2O, Sigma-Aldrich, St. Louis, MO.
c.
DNeasy® Kit, Qiagen Inc., Valencia, CA.
d.
2X PCR Premix Taq Kit, Takara, Otsu, Shiga, Japan.
e.
iCycler™ Thermal Cycler, Bio-Rad Laboratories, Hercules, CA.
f.
QIAquick® PCR Purification Kit, Qiagen Inc., Valencia, CA.
