Abstract
Seven colostrum-deprived, 3–4-wk-old Rambouillet–Hampshire lambs were inoculated via the mucous membranes with deer adenovirus (DAdV) and monitored for clinical signs for 21 d post-inoculation at which time animals were euthanized and postmortem examinations were performed. Pre-inoculation and post-inoculation serum samples were tested for antibodies to DAdV, ovine adenovirus 7, bovine adenovirus 7, and goat adenovirus 1. Evidence for DAdV infection was determined by virus isolation, PCR tests, and histopathology with immunohistochemistry tests for DAdV. No clinical signs or lesions consistent with adenoviral hemorrhagic disease (AHD) in deer were seen in the lambs, and the lambs did not seroconvert to DAdV. DAdV was not detected by PCR, virus isolation, or immunohistochemistry in any of the samples tested from the lambs. A positive control deer similarly inoculated with DAdV developed fatal AHD 1 wk post-inoculation. Our colostrum-deprived lambs did not become infected when inoculated with DAdV.
Keywords
A newly recognized deer adenovirus (DAdV; species Deer atadenovirus A, genus Atadenovirus, family Adenoviridae) was the cause of an outbreak of hemorrhagic disease that caused high mortality in mule deer in California during 1993 and 1994 9 and has since been frequently detected as a cause of hemorrhagic disease in mule deer in the western United States 14 (20100821.2923 ProMED-mail post, https://www.promedmail.org/post/20100821.2923). Sporadic disease has been diagnosed in white-tailed deer (WTD; Odocoileus virginianus) and/or moose in Iowa, Colorado, Wyoming, and Canada.4,5,7 Pulmonary edema, intestinal hemorrhage, and/or ulceration in the upper alimentary tract associated with vasculitis are manifestations of adenoviral hemorrhagic disease (AHD). The disease was successfully reproduced experimentally in black-tailed deer (BTD; Odocoileus hemionus columbianus) fawns and yearlings as well as in WTD fawns.10,11,12 DAdV is most closely related phylogenetically to bovine adenovirus 7 (BAdV-7), goat adenovirus 1 (GAdV-1), and ovine adenovirus 7 (OAdV-7),3,15 which raises the question of whether domestic livestock are susceptible to infection with DAdV. A 2008 study determined that bovine calves do not develop clinical disease or become infected with DAdV, 13 but no studies have been performed in ovine species. Livestock mix with deer in California and are moved around the state and could move DAdV to regions of uninfected susceptible deer. We designed a study to determine if colostrum-deprived lambs can be infected with DAdV, if they develop clinical disease or asymptomatic infection, and if virus can replicate in and be shed by lambs.
Nine newborn Rambouillet–Hampshire lambs were obtained from the Sheep Unit at the University of Wyoming’s Livestock Center (Laramie, WY) prior to receiving colostrum. For the first 3 wk, the lambs were raised in an isolated room and bottle-fed with milk replacer (Super Lamb instant milk replacer, Merricks, Middleton, WI). At 3 wk of age, lambs were transported to the James C. Hageman Sustainable Agriculture Research and Extension Center (SAREC; Animal Biosafety Level 1 [ABSL-1]) near Lingle, WY. Lambs were bottle-fed twice daily with milk replacer for the remainder of the study and provided with 4-way grain mix (Ranch-Way Feeds, Fort Collins, CO). All lambs were vaccinated with 2 mL of Clostridium perfringens types C & D and tetanus antitoxin (Boehringer Ingelheim Vetmedica, St. Joseph, MO) 3 d post-inoculation (dpi) with DAdV.
Four yearling WTD that served as the positive control animals were raised from neonates in an outdoor pen at the Wyoming Game and Fish–Tom Thorne/Beth Williams Wildlife Research Center at Sybille (ABSL-1). When the deer were yearlings, they were moved to a covered outdoor pen at the Wyoming State Veterinary Laboratory (WSVL; Laramie, WY; ABSL-1). Animals were fed natural forage at the research center and then alfalfa hay and alfalfa pellets ad libitum once moved to WSVL. All animals in our study were housed and cared for in compliance with the Animal Welfare Act, U.S. Public Health Service Policy on the Humane Care and Use of Laboratory Animals and the Guide for Care and Use of Agriculture Animals in Agriculture Research and Teaching (1999, IACUC A-3216-01).
DAdV was isolated in fetal WTD lung (FOdvL) cells from lung homogenates of a deer that died of natural infection (California strain D94-2569) 9 as described previously, and used as viral inoculum. 13 The stock virus contained 104.8 TCID50 DAdV per mL. 8
At 3–4 wk of age, 7 of the colostrum-deprived lambs (1–7) were each inoculated with 2 mL of DAdV (104.8 TCID50 per mL) in phosphate-buffered saline (PBS) in each of 4 sites (mucosal membranes of the eyes, nose, mouth, and trachea) and housed together in an isolated indoor room at SAREC. Two negative control lambs were housed in a separate building at SAREC and were inoculated by similar routes with 2 mL of PBS. The negative control animals were always handled first when feeding, treating, or taking samples. The lambs were observed for clinical signs 2–3 times daily, and temperature, pulse, and respiration were evaluated once daily. Disposable diapers were placed on lambs for urine collection on collection days.
Blood and serum were collected for PCR and the virus neutralization (VN) tests, respectively, pre-inoculation (day of inoculation) and every 4 dpi until the end of the study 3 wk post-inoculation (wpi). Serum was tested for antibodies to DAdV, BAdV-7, OAdV-7 (ES3048), and GAdV-1 using the VN test. Nasal, rectal, tonsil, and pharyngeal swabs were collected pre-inoculation and every 4 dpi until the end of the study 3 wpi and stored at −70°C until tested (virus isolation and PCR). At 3 wpi, lambs were euthanized with intravenous pentobarbital, and postmortem examinations were performed. Postmortem examinations were performed on any lambs that died during the study within 8 hr after death. Portions of brain, kidneys, all lung lobes, tonsil, liver, spleen, thymus, salivary gland, bladder, pancreas, mesenteric lymph node, retropharyngeal lymph node, tonsil, adrenal gland, esophagus, abomasum, forestomachs, small and large intestine, heart, skeletal muscle, tongue, trachea, eyelid, lip, reproductive organ, peripheral nerve, nasal turbinates, and eyes were collected and immersed in 10% neutral-buffered formalin and processed routinely for histologic examination. Tissues representing all organ systems were collected and frozen at −70°C until tested.
Four WTD yearlings were each inoculated with 2 mL of the same DAdV viral inoculum in each of the 4 sites (eyes, nose, mouth, trachea) and observed for clinical signs 3 times daily. Deer were euthanized at 3 wpi, and postmortem examinations were performed. Postmortem examinations were performed on deer that died during the study within 8 hr of death, and a similar set of tissues was collected in 10% neutral-buffered formalin for histopathology and immunohistochemistry, and frozen at −70°C.
Immunohistochemistry was performed on lungs, tonsil, retropharyngeal lymph node, and nasal turbinate tissues from all lambs and deer using rabbit polyclonal antibody to DAdV (H Lehmkuhl, NADC; California 1994 DAdV) as described previously. 13 Formalin-fixed lung sections from a deer infected with DAdV in a previous study 11 were used as positive controls; a universal negative rabbit control (Dako North America, Carpinteria, CA) was used as a negative control.
VN tests were performed to determine antibody titers in the lambs to ovine, bovine, and goat adenoviruses and antibody response to DAdV as described previously.1,2 Biologically cloned cervid, bovine, goat, and ovine adenoviruses were used to prepare stock pools of the viruses used in the VN tests. Virus titers were estimated to provide 100 TCID50 per well for the VN test, and tests were performed in 96-well microtitration plates. Known titered rabbit polyclonal antiserum was used for the positive control serum.
Nasal swabs collected from all lambs pre-inoculation, 4-dpi, and tissues collected at the time of autopsy were stored at −70°C in viral transport medium (VTM) with streptomycin (100 mg/mL) and gentamicin (100 mg/mL) until processed for virus isolation. Pooled tissues (lung and tonsil) collected at postmortem were homogenized, and supernatants were inoculated onto flasks of FOdvL cells. Flasks were incubated for 7 d (medium was refreshed 24 h post-inoculation) and were observed daily for cytopathic effect (CPE). Three blind passages were completed to determine if virus was present.
Isolation attempts for OAdV-1–6, OAdV strain RTS-151, BAdV-7, and GAdV-1 were carried out in ovine fetal turbinate (OFTu) cells. In addition, OFTu cells were used for isolation of other ovine respiratory viruses. Stock pools of the prototype adenoviruses (kindly provided by Dr. BM Adair, Veterinary Research Laboratory, Stormont, Belfast, Northern Ireland) were prepared for biological cloning. Antisera to prototypes of OAdV-6, BAdV-7, and GAdV-1 prepared in rabbits were used in cross-neutralization tests for serotype determination.
Nasal, pharyngeal, and rectal swabs were immersed in 1 mL of VTM with glass beads, vortexed vigorously, and allowed to incubate at room temperature for 2 h. DNA was extracted from the pre-inoculation, 4-dpi, and 8-dpi nasal, pharyngeal, and rectal swabs collected from every lamb (Blood and body fluid spin protocol, QIAamp DNA blood mini kit, Qiagen, Valencia, CA). Samples were stored at −20°C until tested by PCR. DNA was extracted from blood samples from all lambs (pre-inoculation, 4-dpi, and 8-dpi; QIAamp blood mini kit).
Urine collected in diapers at each collection date (pre-inoculation, 4-dpi, and 8-dpi) was tested for DAdV (QIAamp viral RNA mini kit, Qiagen). A section of diaper was inoculated with the DAdV viral inoculum used in the lambs and processed to obtain a positive sample. Each section of diaper was placed in a 15-mL centrifuge tube with 4 mL of VTM (Eagle minimal essential medium) and glass beads.
All tissues collected from the lambs at postmortem examination and lung collected from the deer that died were stored at −70°C until tested by PCR. The tissue protocol (QIAamp DNA mini kit, Qiagen) was used to extract DNA from all tissues.
The primers used for PCR were designed (Primer v.3.0 software, Life Technologies, Grand Island, NY), and thereafter analyzed by the Lawrence Livermore National Laboratories Bioinformatics group and the KPATH signature design system 6 as reported previously. 14 The forward and reverse primers are from the fiber portion of the virion and amplify a 187-bp fragment (DAdV810-997).
For each specimen, 50-μL reactions were prepared by adding 10 μL of DNA extract to the master mix in 1.2-mL tubes. The master mix for each reaction was made of 29.75 μL of nuclease-free water, 5 μL of 10× PCR buffer (containing 15 mM MgCl2), 1 μL of dNTP, 2 μL each of the forward and reverse primers, and 0.25 μL of HotStarTaq (Qiagen). Amplification in a thermocycler (DNA Engine PTC-200, Bio-Rad Laboratories, Hercules, CA) consisted of an initial round at 95°C for 15 min, 60°C for 30 s, 72°C for 1 min, 94°C for 30 s, 57°C for 30 s, and 72°C for 30 s, followed by 35 cycles at 94°C for 30 s, 57°C for 30 s, and 72°C for 30 s, then 72°C for 1 min, and 4°C hold.
PCR products were separated by electrophoresis in 1.5% agarose gel (Fisher Scientific, Pittsburg, PA) containing ethidium bromide solution (Sigma-Aldrich, St. Louis, MO). All PCR runs included positive (DAdV-infected lung tissue and/or swabs) and negative control samples to ensure proper amplification and that contamination did not occur between samples.
No lambs developed significant clinical signs consistent with the clinical signs seen in deer infected with DAdV (acute death, hemorrhagic diarrhea, or ulceration and necrosis of the upper alimentary tract). Two lambs had a mild cough 2 d prior to inoculation. Lung sounds were normal on auscultation, so we decided to proceed with the study. However, most lambs developed cough and nasal discharge as the study progressed, including the negative control lambs. Lambs 1, 5, and 6 separated from the group and developed distended abdomens and a bracing position stance 3 dpi (compatible with gastrointestinal disease). Lamb 6 died 24 h after first showing clinical signs (4 dpi). Lamb 4 developed a distended abdomen and respiratory signs, and died 7 dpi. Lamb 8 (negative control) developed respiratory signs and was found dead 2 dpi. One of the 4 WTD yearlings was found dead without clinical signs 1 wpi. No clinical signs or mortality occurred in the remaining 3 deer.
Of the lambs that died prior to the end of the study, lambs 4 and 8 had bronchopneumonia, and lamb 6 had abomasitis. Gross and microscopic changes in lambs 4 and 8 were compatible with bacterial bronchopneumonia from which Mannheimia haemolytica was isolated. Lamb 6 had gross and microscopic changes consistent with Sarcina-like abomasal infection.
Three wk post-inoculation, the remaining 6 lambs were euthanized, and postmortem examinations performed. All 6 lambs had pneumonia of variable severity, including the lambs in the negative control group (Table 1). Lambs 3 and 5 had syncytial cells (one with intracytoplasmic inclusions) suggestive of respiratory syncytial virus (RSV). Lamb 2 had renal medullary epithelial intranuclear inclusions without associated necrosis or inflammation.
Histopathology, PCR, immunohistochemistry, and virus isolation results of colostrum-deprived lambs (1–7) inoculated with deer adenovirus, and negative control, colostrum-deprived lambs (8–9).
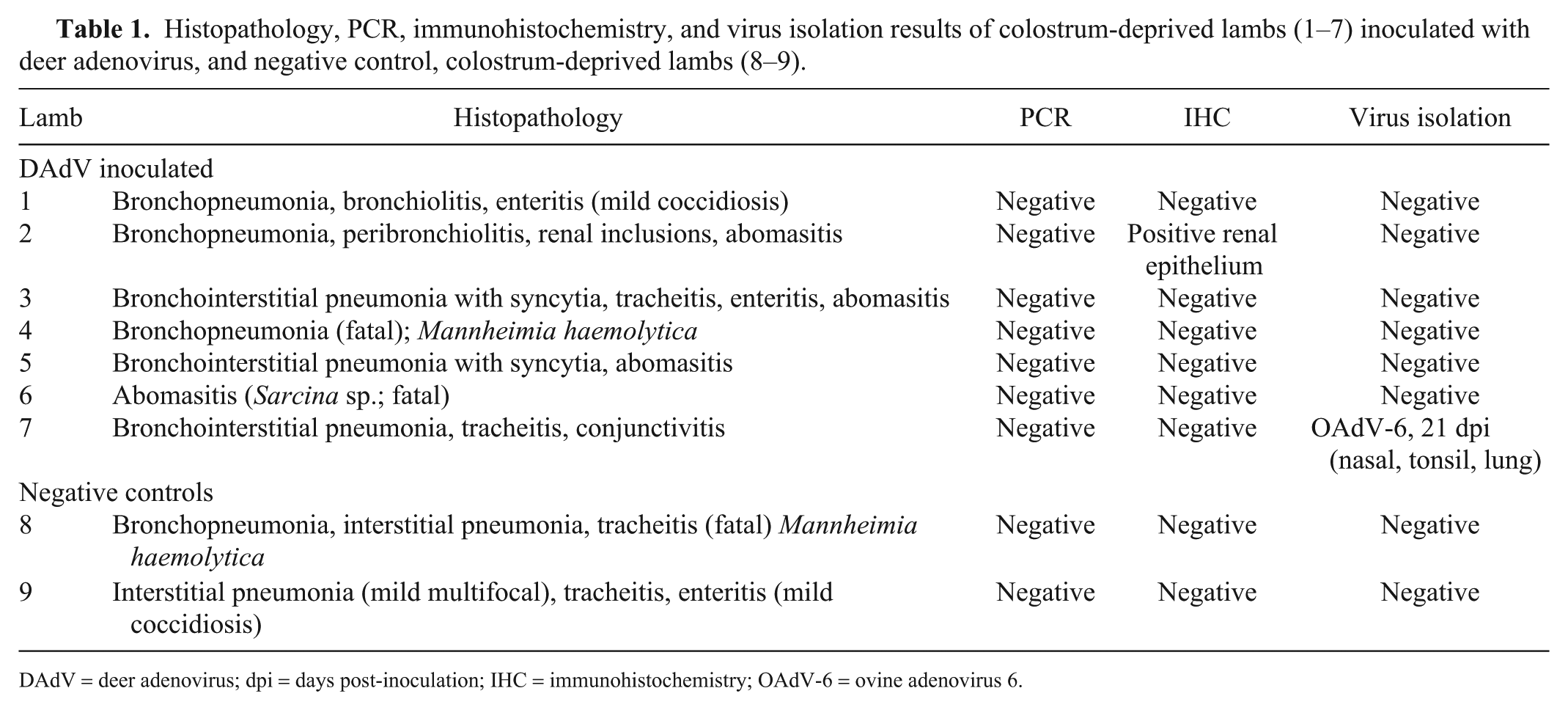
DAdV = deer adenovirus; dpi = days post-inoculation; IHC = immunohistochemistry; OAdV-6 = ovine adenovirus 6.
The positive control deer that died 1 wpi with DAdV had pulmonary edema and abomasal ulceration on gross examination. Microscopic examination of tissues demonstrated systemic vasculitis and multifocal ulcerative abomasitis with systemic vascular endothelial intranuclear inclusions compatible with AHD. The 3 remaining deer had no gross anatomic or microscopic lesions.
Pre-inoculation swabs (nasal, rectal, pharyngeal), urine and blood, and swabs and blood taken at 4 and 8 dpi were all negative for DAdV by PCR. Additionally, DAdV was not detected by PCR in any of the tissues taken at autopsy from each of the lambs, including lamb 2 with the renal epithelial intranuclear inclusions (Table 1).
Immunohistochemistry detected DAdV in endothelial cell nuclei in blood vessels in the lungs of the infected deer that died. IHC results for the lambs in our study did not demonstrate viral antigen in any of the tissues examined microscopically, except for the renal medullary intranuclear inclusions seen in lamb 2 (Table 1).
After the first passage, tissue from lamb 7 produced CPE in cell cultures. The only reference antiserum that neutralized the isolates was antiserum OAdV-6 (Table 1). Preliminary sequencing of the DNA extracted from the isolate using primers targeting the hexon region of Mastadenovirus showed the highest percentage of identity to OAdV-6 hexon gene region. No CPE was detected when both kidneys from lamb 2 with the renal medullary epithelial intranuclear inclusions were cultured on OFTu or FOdvL cells. DAdV was not isolated from swabs or tissues from any of the lambs.
Most of the lambs had low antibody titers (<64) to DAdV post-inoculation (Table 2). Lamb 1 had a 4-fold increase in antibody to DAdV between pre-inoculation (<2) and 12 dpi (32) and a 16 titer at 21 dpi. All lambs except lamb 6 had low antibody titers to OAdV-7 and GAdV-1, and titers to BAdV-7 that varied from 4 to 128. Lamb 6 had antibody titers of 256, 2,048, and 256 to OAdV-7, GAdV-1, and BAdV-7, respectively.
Virus neutralization titer results for colostrum-deprived lambs (1–7) inoculated with deer adenovirus 1, and colostrum-deprived, negative control lambs (8–9).
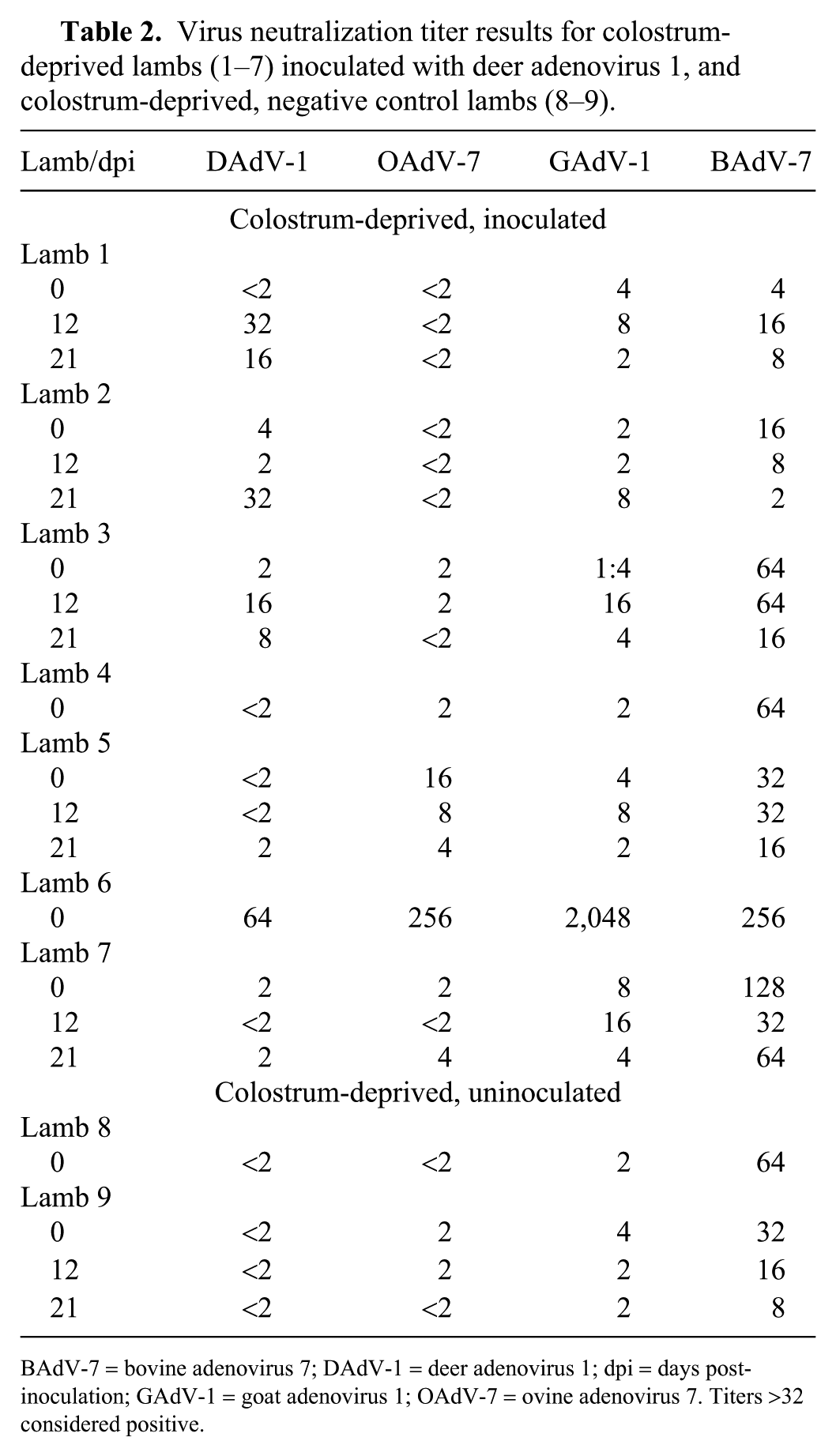
BAdV-7 = bovine adenovirus 7; DAdV-1 = deer adenovirus 1; dpi = days post-inoculation; GAdV-1 = goat adenovirus 1; OAdV-7 = ovine adenovirus 7. Titers >32 considered positive.
Our results demonstrated that lambs likely do not develop clinical or asymptomatic infections when inoculated with DAdV via the mucous membranes. Lambs deprived of colostrum were inoculated in order to evaluate susceptibility of immunodeficient lambs to DAdV and to ensure no interference from maternal antibodies. The titer of the viral inoculum used was higher than the dose used in a previous experimental study in which 8 of 10 BTD fawns died 11 and the same viral inoculum used in another study in which 2 of 4 BTD fawns in a positive control group died of experimental AHD. 13 One of 4 WTD yearlings died, which is similar to the susceptibility of WTD 12 and to yearlings (vs. fawns) in previous studies 14 and therefore demonstrated that the virus was viable and lethal.
None of the lambs had the classic pattern of antibody response to infection characterized by a 4-fold rise in antibody titer to DAdV post-inoculation, indicating that DAdV infection did not occur. Lamb 1 had a very low (32) but 4-fold rise in antibody titer to DAdV 12 dpi, which waned at 3 wk. This slight antibody response was likely directed against the viral inoculum as seen in a previous study in calves. 13 Lamb 6 had a 64 titer to DAdV but higher titers to OAdV-7 (256), GAdV-1 (2,048), and BAdV-7 (256), indicating that this lamb likely nursed before removal from the ewe. The titer to DAdV was likely a cross-reaction with GAdV-1 (Table 3). Also, this lamb died 4 dpi, before antibody response to the cervid adenovirus was possible.
Results of cross-neutralization tests between members of the ruminant Atadenovirus genus.
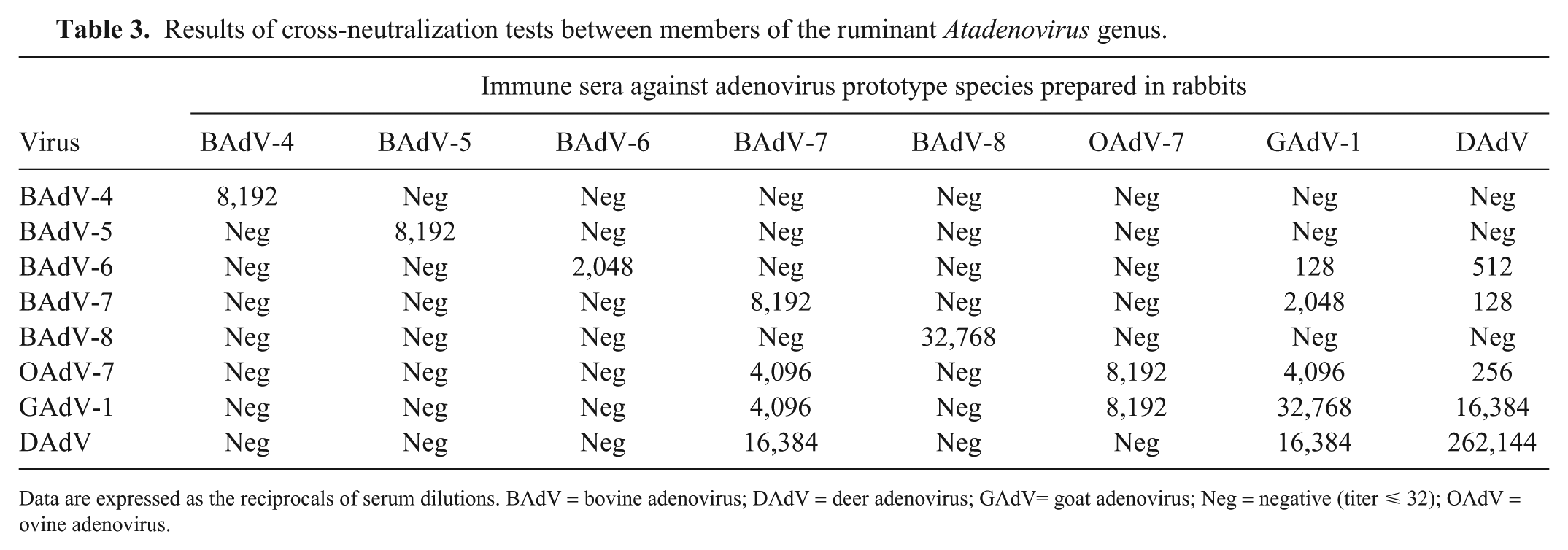
Data are expressed as the reciprocals of serum dilutions. BAdV = bovine adenovirus; DAdV = deer adenovirus; GAdV= goat adenovirus; Neg = negative (titer ⩽ 32); OAdV = ovine adenovirus.
Respiratory agents were circulating within the flock during the study, with the likely source being the ewes prior to lamb removal. Syncytia associated with bronchointerstitial pneumonia in 2 lambs suggest an infection with RSV. OAdV-6 was isolated from lamb 7 and may have been partly responsible for some of the respiratory signs and lesions in the flock.
All swabs and blood collected at 4 and 8 dpi were negative by PCR and virus isolation, ruling out early DAdV infection, replication, and shedding through the respiratory, alimentary, and urinary tracts. Negative results on the PCR tests and virus isolation on the tissues indicated that there was no DAdV in any of the tissues tested at the time of autopsy, suggesting that a DAdV infection was not established in any of the inoculated lambs. In pathogenesis studies of cervid adenovirus infection in deer, the presence of virus in tissues was detected as early as 3 dpi. 11
IHC, using a polyclonal antibody directed against DAdV, labeled antigen in the intranuclear inclusions in the kidney of lamb 2, indicating that the virus was either cervid adenovirus or an antigenically related adenovirus. Deer adenovirus was not isolated from the kidneys of this lamb, and the kidneys were negative for DAdV by PCR, indicating this was not DAdV but likely an antigenically related adenovirus. The polyclonal antibody directed against DAdV has been demonstrated to cross-react and label several of the ovine and bovine adenoviruses (L Woods, personal observation).
Unfortunately, respiratory viral and bacterial infections confounded our inoculation study of lambs with DAdV. Concurrent infection of animals with viruses can interfere with interpretation of clinical and pathology results in experimental inoculation studies. However, DAdV was not detected in any of the lambs by any detection test employed, strongly indicating that sheep are not susceptible to DAdV.
Our results have implications for management of sheep production facilities and free-ranging deer populations. Further studies in respiratory disease–free, colostrum-deprived lambs are needed but our results imply that deer infected with DAdV are not likely to pose a threat to domestic sheep health, and domestic sheep herds are not likely to transmit DAdV to susceptible populations when exposed to deer with AHD.
Footnotes
Acknowledgements
We gratefully acknowledge the thesis committee, Everett L. Belden, Geoff J. Letchworth and Steven I. Paisley and the former director of the Wyoming State Veterinary Laboratory at the time of the study, Dr. Donal O’Toole. We also appreciate the excellent assistance by Mercedes Thelen and Paula Jaeger for histology and IHC support, Katie Bardsley for clinical chemistry support, Brent Larson for providing the lambs, Dr. Dave Legg for statistical support, and Lea Ann Hobbs for VI and VN support and training.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funded by the United States Department of Agriculture (grant 58-3625-2-143) and the Wyoming Wildlife/Livestock Disease Partnership.
