Abstract
Two aborted Chester White pig fetuses were presented to a veterinary diagnostic laboratory in Illinois. Postmortem examination identified no gross abnormalities. Histologic evaluation revealed multifocal necrosis of chorionic epithelial cells, coalescing areas of mineralization in the placenta, and focal accumulations of viable and degenerate neutrophils in the lung. Intra- and extracellular acid-fast bacilli were identified in the lesions in both the placenta and lungs. Bacterial culture of stomach contents yielded heavy growth of Mycobacterium fortuitum, a rapidly growing nontuberculous mycobacterium (NTM), which was further confirmed through whole-genome sequencing. NTM are opportunistic pathogens commonly found in the soil and in contaminated water supplies. In animals, M. fortuitum is typically introduced through cutaneous wounds leading to infections limited to the skin, with systemic infection being uncommon. To our knowledge, abortion caused by M. fortuitum has not been reported previously.
In July 2019, a group of sows and gilts housed on dirt lots on a farm in Illinois, USA had an 8-mo history of poor signs of estrus, late-term abortions, stillbirths, and weak piglets. Two aborted Chester White fetuses and placenta from one sow were submitted for postmortem examination. Significant gross abnormalities were not identified in either fetus.
Stomach contents were submitted for bacterial culture and Brucella PCR. Kidney, liver, spleen, and placental tissues were submitted for detection of Salmonella, Leptospira, porcine reproductive and respiratory syndrome virus (PRRSV1 and 2; Arteriviridae, Betaarterivirus, Betaarterivirus suid 1 and 2), and porcine circovirus 2 and 3 (PCV2 and PCV3; Circoviridae, Circovirus) by PCR, and porcine parvovirus 1 (PPV1; Parvoviridae, Protoparvovirus, Ungulate protoparvovirus 1) by fluorescent antibody (FA) testing. The PCR and FA tests were negative. A heavy growth of pinpoint bacterial colonies resembling Mycobacterium sp. was noticed on the Columbia agar with 5% sheep blood and CNA (colistin and nalidixic acid) agar plates. Bacteria were further identified as Mycobacterium fortuitum by MALDI-TOF MS with a high confidence score of 2.12. No other suspect bacterial growth was identified through culture.
Specimens from the placenta and all major organs from both fetuses were examined histologically. There was multifocal necrosis of chorionic epithelial cells, which were sloughed and mixed with moderate numbers of neutrophils, macrophages, and scattered extracellular bacteria (Fig. 1). The subjacent connective tissues contained coalescing areas of mineralization. In the lungs of one fetus, bronchioles and alveoli were multifocally filled with viable and degenerate neutrophils, fewer macrophages, suspected intrahistiocytic bacilli, and larger clumps of extracellular bacilli (Fig. 2). Because M. fortuitum was isolated from stomach contents, Fite acid-fast stain was performed on sections of the placenta and lung. In the placenta, many clumps of acid-fast, ~0.5 × 0.8 µm bacilli were identified in the cytoplasm of inflammatory cells (Fig. 3). Foci of inflammation in the lung also contained many acid-fast, intra- or extracellular bacilli of similar size (Fig. 4). Similar, less severe changes were identified in the second fetus. Histologic findings of suppurative and histiocytic bronchopneumonia and placentitis associated with acid-fast, intrahistiocytic bacilli confirmed M. fortuitum as the cause of abortion in this sow. Other sows and gilts on the farm with decreased reproductive fitness may also have been infected, although we were unable to investigate this because additional tissues and environmental samples were not submitted.
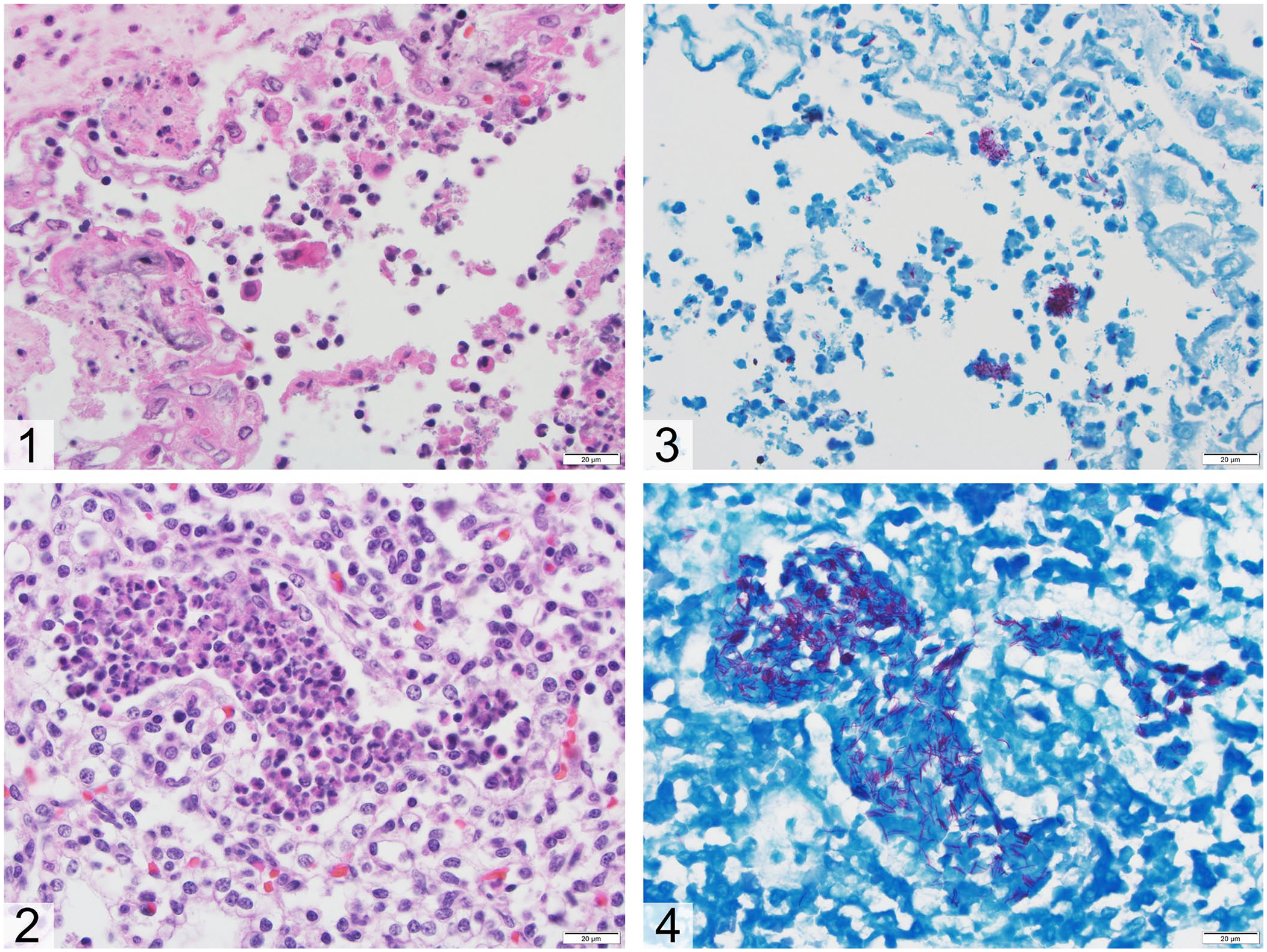
Mycobacterium fortuitum abortion in a sow. Bar = 20 µm.
M. fortuitum is an acid-fast, rapidly growing, saprophytic, nontuberculous mycobacterium (NTM) that is typically considered an opportunistic pathogen. It is found in soil and can contaminate water supplies to become aerosolized for easy spread. 8 Rapidly growing mycobacteria (RGM) can grow on a 5% sheep blood agar plate without specific additives within 7 d; in contrast, slowly growing mycobacteria may take weeks to months to form mature colonies. 1 The rapid growth paired with real-time identification by MALDI-TOF MS can aid in their detection and prompt treatment. 1 The slow-growing Mycobacterium avium complex (MAC) is the most common NTM infection in swine. 12 NTM infections generally cause no overt clinical signs, although infected pigs may have granulomatous lesions associated with lymphoid organs at postmortem examination. 12
Abortion is an uncommon manifestation of NTM infection, associated mostly with MAC, in sows,5,6 cattle, 9 and mares.4,11 In a 1979 case report, an aborted fetus from a MAC-infected sow in Iowa had granulomatous hepatitis with intralesional acid-fast bacilli; the sow had similar granulomas in multiple organs. 6 MAC infections have also been associated with abortion outbreaks in herds of pigs causing similar systemic involvement in the sow, placentitis, and fetal pneumonia with or without hepatitis.5,23 Abortion in a mare has been reported as a result of infection with Mycobacterium terrae, another slow-growing NTM. 21 In domestic animals, M. fortuitum infections are most frequently reported in cats in which the agent is introduced via breaks in the skin causing cutaneous or subcutaneous nodules, granulomatous panniculitis, or, less commonly, disseminated disease. 10 Although generally considered opportunistic, cats infected with rapidly growing NTM are typically not immunosuppressed. 15 In contrast, NTM most commonly affects the pulmonary system in humans with underlying structural disease; rare disseminated disease occurs in severely immunocompromised patients. 13
Because abortion caused by M. fortuitum has not been reported previously, the isolate from our case (IL2019) was submitted for sequencing for further characterization. DNA was extracted (DNeasy blood and tissue kit; Qiagen) followed by quantification (Qubit 3.0 fluorometer; Thermo Fisher). DNA was subjected to library preparation (Nextera XT kit, MiSeq reagent kit v2; Illumina; 500 cycles) as described previously. 22 Raw FASTQ data of the isolate were assembled (SPAdes v.3.12.0; http://cab.spbu.ru/software/spades/) and deposited into the PubMed database (SRA accession SRR14294491). The assembled contigs were analyzed for the presence of 16S ribosomal RNA sequence (Barrnap v.0.6; https://github.com/tseemann/barrnap). Software (v.7.0.26, MEGA; kSNP v.2, SourceForge) was used to analyze FASTA sequence data for phylogenetic tree analysis. BLAST search of 16S ribosomal RNA sequence showed that IL2019 was 100% similar to Mycobacterium sp. isolates VKM Ac-1817D and SY-1-26, and M. fortuitum isolates NCH E11620, DSM 46621, and CT6, and over 99% similar with Mycobacterium sp. isolates 27335 (99.4%) and DL90 (99.2%). Phylogenetic tree analysis of 16S also indicated that IL2019 clustered with those 5 mycobacterial isolates (Fig. 5A). By contrast, the whole-genome sequence–based single-nucleotide variation analysis has a higher level of discriminatory power than 16S analysis, and revealed that this isolate is more closely related to M. fortuitum CT6 isolate than to DSM 46621 and VKM Ac-1817D (Fig. 5B).
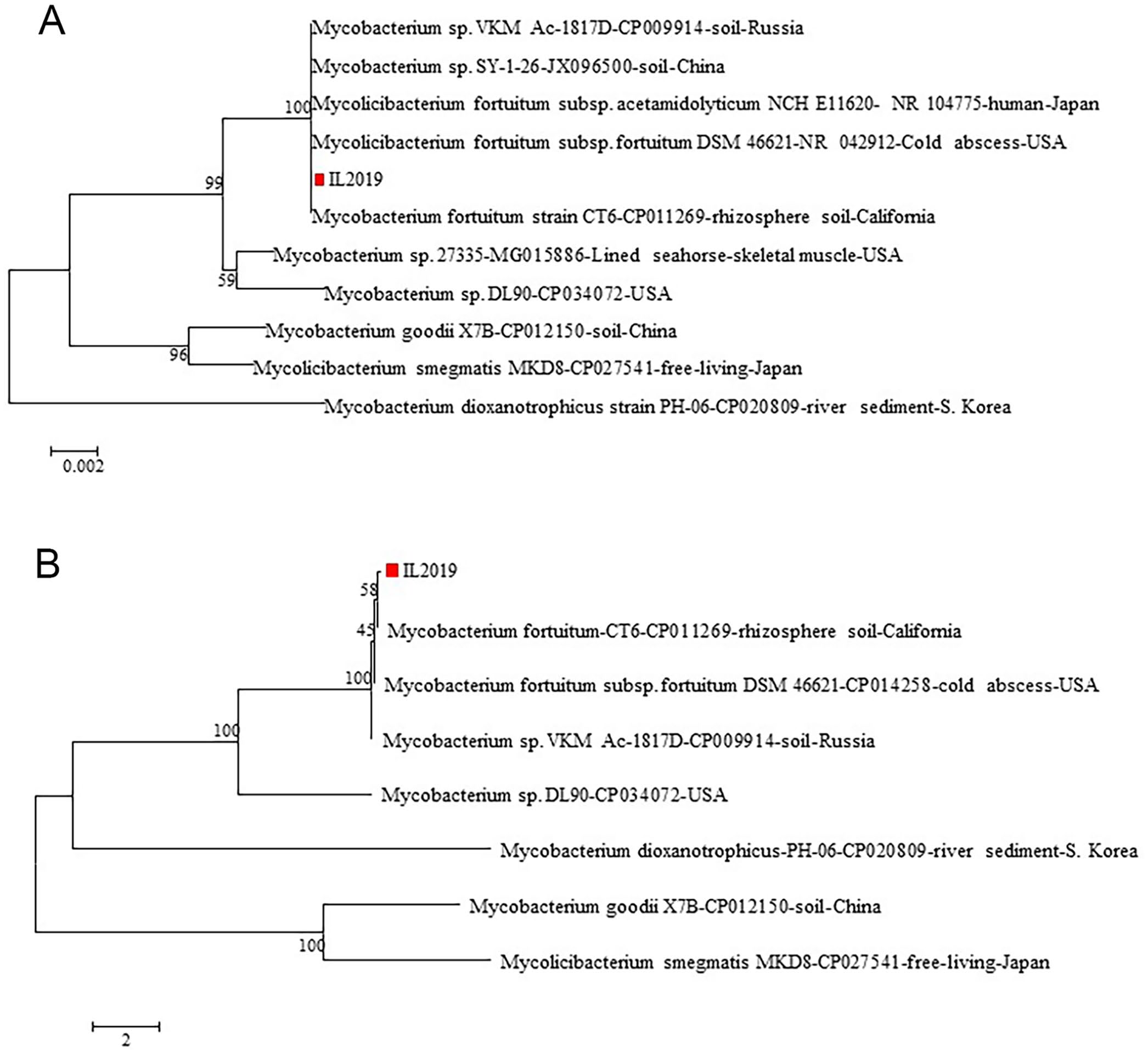
Phylogenetic tree analysis of
NTM infections in pigs are more commonly detected on postmortem examination rather than antemortem, so they are not usually treated with antibiotics. 12 In cats, antemortem testing is more common, and pradofloxacin with clarithromycin has been suggested for treatment of M. fortuitum infection in these animals. 10 Although clarithromycin has been shown to have efficacy in vitro, its in vivo application is limited to delayed or induced resistance in RGM species.2,16 Thus, macrolides should be avoided for treatment in RGM. Full-genome sequencing of the isolate from our case identified one of the macrolide resistance genes, erm(39), which encodes the ribosomal methylase, impairing the binding of macrolides to the ribosome. 17 This result is consistent with previous studies demonstrating that erm(39) is present in M. fortuitum isolates, but not erm(41), which is present uniquely in Mycobacterium abscessus.14,17 Susceptibility testing of the isolate showed the expected resistance to clarithromycin (Suppl. Table 1). In vitro studies suggest the efficacy of tetracycline and fluoroquinolones, which may be more promising and can be used in a wider variety of species. 20
Abortion in sows can be a costly issue for producers, and there are numerous etiologies to consider. Cases in which the problem is localized to the conceptus are less frequent than those that directly affect the sow. Unfortunately, maternal causes of abortion rarely leave gross lesions in the aborted litter, given that the litter is viable until expulsion. Non-infectious causes, such as nutritional deficiencies, toxins, and genetic anomalies, should be considered, but are less common than infectious etiologies. 7 Viral causes of abortion in sows are PRRSV, PCV, PPV, pseudorabies virus (PRV; Suid alphaherpesvirus 1), and classical swine fever virus (CSFV; Pestivirus C). 18 Brucella suis and Leptospira spp. are important bacterial causes of abortion in pigs, although bacteremia caused by a number of pathogens, such as Streptococcus suis, Escherichia coli, Erysipelothrix rhusiopathiae, Salmonella spp., and Staphylococcus spp. can cause placentitis resulting in abortion. 18 In a study performed in Italy, the most common agents of abortion were PRRSV and PCV, with 40–45% of the cases being coinfections with other viral or bacterial agents. 19
To our knowledge, M. fortuitum has not been documented previously as a cause of abortion in any species. The sow of the aborted fetus and others in the group did not undergo postmortem examination given that they remained asymptomatic. Tuberculin skin tests can be considered for antemortem detection of NTM in pigs, although results can be variable. 23 The presumptive mode of infection was through the soil given that these pigs were housed on a dirt lot with subsequent vertical transmission to the fetus. M. fortuitum, like other NTM, is relatively resistant to chlorine disinfectant; however, previous research has shown it is rapidly neutralized by alkaline glutaraldehyde. 3 Preventive measures can include individual or small group housing on flooring that can be sanitized easily with appropriate disinfectants. The number of pigs in the group was not provided, likewise vaccination status of the group and morbidity data over the 8-mo period were not available. Without additional history or samples from other pigs or the environment, we were unable to determine if this was a sporadic event or the cause of reproductive decline in the entire group. However, the producer of these pigs has since filled in the lot with concrete and has not experienced additional reproductive losses.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387211042289 – Supplemental material for Mycobacterium fortuitum abortion in a sow
Supplemental material, sj-pdf-1-vdi-10.1177_10406387211042289 for Mycobacterium fortuitum abortion in a sow by Allysa L. Cole, Natalie M. Kirk, Leyi Wang, Chien-Che Hung and Jonathan P. Samuelson in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the histology, microbiology, and molecular laboratories at the University of Illinois Veterinary Diagnostic Laboratory for their work on this case.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
