Abstract
Eighty feral swine were trapped from a herd that had been documented to be seropositive for Brucella and which had been used for Brucella abortus RB51 vaccine trials on a 7,100-hectare tract of land in South Carolina. The animals were euthanized and complete necropsies were performed. Samples were taken for histopathology, Brucella culture, and Brucella serology. Brucella was cultured from 62 (77.5%) animals. Brucella suis was isolated from 55 animals (68.8%), and all isolates were biovar 1. Brucella abortus was isolated from 28 animals (35.0%), and isolates included field strain biovar 1 (21 animals; 26.3%), vaccine strain Brucella abortus S19 (8 animals, 10.0%), and vaccine strain Brucella abortus RB51 (6 animals, 7.5%). Males were significantly more likely to be culture positive than females (92.9% vs. 60.6%). Thirty-nine animals (48.8%) were seropositive. Males also had a significantly higher seropositivity rate than females (61.9% vs. 34.2%). The relative sensitivity rates were significantly higher for the standard tube test (44.6%) and fluorescence polarization assay (42.6%) than the card agglutination test (13.1%). Lesions consistent with Brucella infection were commonly found in the animals surveyed and included inflammatory lesions of the lymph nodes, liver, kidney, and male reproductive organs, which ranged from lymphoplasmacytic to pyogranulomatous with necrosis. This is the first report of an apparent enzootic Brucella abortus infection in a feral swine herd suggesting that feral swine may serve as a reservoir of infection for Brucella abortus as well as Brucella suis for domestic livestock.
Introduction
The genus Brucella composes a group of genetically very similar, Gram-negative, facultative intracellular, pathogenic bacteria, which are known to infect over 80 domestic and wild mammalian species. 13 , 15 , 32 , 33 In animal hosts, brucellae commonly are associated with infection of reproductive tissues of the male and female and lymphoreticular tissues as well as pyogranulomatous lesions of multiple tissues. 19 Four members of the Brucella genus, Brucella melitensis, Brucella suis, Brucella abortus, and Brucella canis are zoonotic agents and produce a disease in humans referred to as undulant fever, which is characterized by recurrent pyrexia, infection of lymphoreticular tissues, bacteremia, and purulent and pyogranulomatous lesions in multiple organs in infected humans. 14 , 35 B. melitensis, B. abortus, and B. suis are also on the US Centers for Disease Control and US Department of Agriculture (USDA) Select Agents list because of their perceived potential use as biologic agents of terrorism and war. 35
The United States has had a concerted effort to eliminate both cattle and swine brucellosis from its livestock populations through cooperative programs between the states USDA Animal Plant Health Inspection Service (APHIS). 22 Both programs have been based on the principle of serologic testing and elimination of seroreactive herds. The eradication program in cattle has been aided by the implementation of calfhood vaccination with reduced virulence vaccine strains S19 and RB51. The swine program never implemented the use of a vaccine, but dramatic changes in swine management practices since the 1950s undoubtedly aided the elimination of swine brucellosis in domestic herds. With the near total eradication of brucellosis from the US livestock population, there is an increased effort to address the issue of Brucella infected wildlife populations. 7 Within the 48 contiguous states of the United States, feral swine (Sus scrofa), elk (Cervus elaphus nelsoni), and bison (Bison bison) are the major wildlife reservoirs of Brucella. 23 Today only sporadic outbreaks of brucellosis occur in both domestic swine and cattle within the United States, and most recent outbreaks have been associated with contact with Brucella infected feral swine or elk. 8
Within the United States B. abortus is maintained enzootically within elk (Cervus elaphus nelsoni) and bison (Bison bison) within the Greater Yellowstone Area of Wyoming, Montana, and Idaho. Brucellosis has been reported in the bison of Yellowstone National Park since 1917, and current studies from the Greater Yellowstone Area report seroprevalence rates of 76–90% with 46% of seropositive females being culture positive. 25 , 34 The bison herds have been managed to remain on federal land and likely have had limited contact with cattle. However, elk numbers in the area are much higher. Brucella infected elk have a much larger geographic distribution than bison, and elk often come into close contact with cattle in the area. Recent brucellosis seroprevalence rates among elk herds associated with winter feed ground has been reported to be 25–54%. 6 , 17 However, seroprevalence rates of elk herds not associated with feed ground has been reported to be 0–1%. 24
Feral swine herds have been reported in 27 of 50 states in the United States. The numbers and population distribution of feral swine have increased dramatically in recent decades partially caused by natural population dynamics but mostly because of the establishment of new populations for the purpose of hunting in areas that were previously feral-swine free. Brucellosis has been documented in feral swine populations in 14 states. 35 In addition to the infection of domestic swine with B. suis, feral swine are also responsible for infecting cattle with B. suis. 9 Because of their popularity as a game species and the widespread distribution of brucellosis within these populations, feral swine are also a significant threat for zoonotic infections. 27
Materials and methods
This study was conducted on a 7,100 hectare tract of land on a peninsula between the Atlantic Ocean and the Winyah Bay in Georgetown County, South Carolina, USA (33°20′N, 79°13′W). The study was conducted between October 2002 and February 2003. A total of 80 sexually mature feral swine (42 males and 38 females) were trapped using box traps and fermented shelled corn as bait. This population had been used to evaluate the efficacy of parenteral administration of B. abortus RB51 (RB51) in feral swine. The work was performed under the approval of the Institutional Animal Care and Use Committee of the National Animal Disease Center, Ames, IA, USA.
Necropsies. After euthanasia, 30 ml of blood was obtained via cardiac puncture. Blood was divided for serology (10 ml) and for Brucella culture (20 ml). Urine, nasal swabs, and vaginal swabs were collected for bacteriologic culture. The following tissues were harvested for bacteriologic culture and histologic examination: liver, spleen, lung, kidney, uterus, mammary tissue, testis, seminal vesicle, bulbourethral gland, prostate, and lymph nodes including prescapular, medial retropharyngeal, sternal, tracheobronchial, gastrohepatic, prefemoral, popliteal, mandibular, and parotid. All samples for bacteriologic evaluation were frozen at −70°Cuntil processed for culture. Tissues collected for histologic evaluation were immediately placed in neutral-buffered 10% formalin, processed by routine paraffin embedding techniques, cut in 4-μm-thick sections, and stained with hematoxylin and eosin (HE). Selected tissue sections were also stained by Ziehl-Neelsen (acid-fast), periodic acid-Schiff (PAS), and Hucker-Twort (silver) methods.
Serologic evaluation. After collection, blood samples were allowed to clot and serum was separated by centrifugation. Serum was divided into 1-ml aliquots, and stored at − 70°C until assays were performed. Brucella serologic status of all animals was determined by fluorescence polarization (FPA), 21 standard tube agglutination (STT), 1 and card agglutination assays 1 by previously described methods. Animals that had a positive reaction on at least 1 of the 3 serologic assays were considered positive seroreactors.
Bacteriologic culture. After thawing, tissues were individually ground in approximately 10% (w/v) sterile phosphate buffered saline (PBS, pH = 7.2) using sterile glass grinders. Aliquots (100 μl) of each tissue homogenate were plated on tryptose agar containing 5% bovine serum as well as 3 Brucella selective media including brilliant green agar (BGB132; tryptose agar base with 5% bovine serum, 0.001 μg/ml brilliant green, 25 U/ml bacitracin, 100 μg/ml cycloheximide, 100 U/L nystatin, 20 μg/ml vancomycin, 50 μg/ml trimethoprim, and 100 μg/ml EDTA), Kudzas Morse agar (KM; tryptose agar base with 5% bovine serum, 25 U/ml bacitracin, 6 U/ml polymyxin B, 100 μg/ml cycloheximide, and 1.4 μg/ml ethyl violet), and RBM agar, a selective medium for SRB51. 16 Inoculated plates were incubated at 37°C in 5% CO2 for 7 days.
Culture identification. Brucella suspect cultures were identified on the basis of colony morphology, growth characteristics, and growth on selective media. 1 Isolates were identified as Brucella spp. by a polymerase chain reaction technique (PCR) using Brucella-specific primers to the omp2A region of the Brucella genome. Reactions consisted of 50 μl and contained 5 μl of suspect culture in tris-EDTA and 45 μl of reaction mixture consisting of 200 μM each of dATP, dCTP, dGTP, and dTTP a , 1 × PCR Buffer IIb, 1.5 mM MgCl2, 1.25 U AmpliTaq Gold polymerase b , and 0.2 μM of each upstream and downstream primers (Table 1) selected from the omp2A sequences of B. abortus. 12 The primers had 100% conservancy within the genomes of B. suis and B. melitensis according to BLAST analysis. Tris-EDTA and a culture of Yersinia enterocolitica O:9 served as negative controls, and B. suis strain 3B, a biovar 1 laboratory challenge strain, served as a positive control. Following a 10-min activation at 95°C, reaction preparations were cycled in a thermocycler c for 40 cycles consisting of 30 sec at 95°C, 30 sec at 44°C, and 60 sec at 72°C. Products were analyzed by electrophoresis on 1.5% agarose gels stained with ethidium bromide. Animals were considered culture positive if at least 1 sample yielded a positive Brucella culture.
Primer sequences used in PCR and sequencing assays for the identification of Brucella isolates obtained from feral swine.

Cultures that were positive by the omp2A PCR assay were run in a second multiplex PCR that could discriminate B. suis, B. abortus RB51, B. abortus S19, and B. abortus/ B. melitensis field strains. This PCR assay used RB51 specific primers targeted toward the insertion sequence 711 (IS711).4, 31 Each reaction mixture consisted of a volume of 25 μl containing 2.5 μl of suspect culture in Tris-EDTA, 1X PCR reaction buffer containing 50 mM Tris, 1.5 mM MgCl2, 10 mM KCl, 50 mM (NH4)2SO4, pH = 8.3 d , 200 μM each of dATP, dCTP, dGTP, and dTTP a ; 1X GC rich solution d ; 1.0 U of FastStart DNA polymerase e ; and 0.2 μM of the following primers: IS711 specific, RB51-3, eri multiplex forward, eri multiplex reverse, 42797 forward, and 42797 reverse (Table 1). Thermocycling consisted of a single 5-min incubation at 95°C followed by 40 cycles consisting of 15 sec at 95°C, 30 sec at 52°C, and 90 sec at 72°C. Tris-EDTA and B. suis strain 3B, SRB51, S19, and B. abortus strain 2308 served as the positive control. Products were analyzed by electrophoresis on 2.0% agarose gels stained with ethidium bromide.
To determine the degree of genetic homogeneity among the feral swine isolates found in this study and to aid in the positive identification of B. abortus vaccine strains, a variable nucleotide tandem repeat (VNTR) assay (“HOOF-Prints” assay) was employed as previously described. 2 , 3 Isolates were assayed for the number of copies of the 8 bp repeat (5′-AGGGCAGT-3′) at loci 1–8 as reported in the original paper describing the technique. 3
S19 Confirmation and sequencing. Isolates that were S19 based on the multiplex PCR were selected to determine if the deletion in the eri gene was the same as was previously reported by the reference strain of S19. 27 S19 isolates were subjected to a PCR reaction that flanked the deletion in the eri gene. 26 Each reaction consisted of 50 ml and contained 5 ml of suspect culture in tris-EDTA and 45 ml of reaction mixture consisting of 200 μM each of dATP, dCTP, dGTP, and dTTP a , 1X PCR Buffer II b , 1.5 mM MgCl2, 1.25 U AmpliTaq Gold polymerase b , and 0.2 μM of each eri sequencing forward and reverse primers (Table 1). Thermocycling consisted of a 10-min activation at 95°C followed by 40 cycles consisting of 60 sec at 95°C, 30 sec at 57°C, and 30 sec. at 72°C. Laboratory propagated S19 and B. abortus strain 2308 served as positive controls and Yersinia enterocolitica O:9 served as a negative control. Products were analyzed by electrophoresis on 1.5% agarose gels stained with ethidium bromide. PCR products were purified in a silica matrix f before sequencing. Products were quantitated using the Pico Green assay for dsDNA g . The appropriate quantity of dsDNA was labeled in both directions using Big Dye terminator chemistries and sequenced using an ABI 3100 genetic analyzer h . Primers used for sequencing were identical to those used in the primary PCR reaction and were used at a concentration of 0.13 μM. Resulting sequences were assembled and edited using Sequencher 3.0 i . Alignment of gene segments was performed with the program CLUSTALX. 30
Statistical Analysis. For all statistical analyses a level of P < 0.05 was used to determine differences between vaccinates and controls. Chi square analysis j was used to determine differences between males and females within the population for serologic positivity and serologic sensitivity, differences among serology assays for sensitivity rates, and differences between culture positive and culture negative animals for histologic lesions that were not sex specific.
Fisher's Exact Test j was used to determine differences between males and females within the population for culture positivity, the isolation of multiple Brucella species, and the isolation of specific Brucella species as well as differences between culture positive and culture negative males for sex specific histologic lesions.
Results
Necropsy findings and gross lesions. Of the 38 females that were trapped, 15 (39.5%) were gestating and 7 (18.4%) were lactating. In utero litter size ranged from 2 to 7 (mean = 4.6; median = 5). The seminal vesicles of one male were diffusely enlarged, fibrotic, and contained an abundant amount of purulent exudate. B. suis was subsequently cultured from the affected seminal vesicle. Three animals had grossly visible lymph node abscesses (2 gastrohepatic and 1 sternal). One animal had multiple pleural and mesenteric adhesions and abscesses. Five animals had multifocal, raised, firm hepatic nodules that were consistent with larval migrans tracts. Thirty-three animals had marked proliferation of the ureter and periureteral tissue which contained numerous adult nematodes consistent with Stephanurus dentatus.
Summary of histologic lesions observed in 80 feral swine (42 males, 38 females) trapped from a herd enzootically infected with Brucella spp.
Number of animals with the lesion which were culture + for Brucella.
Number of animals with the lesion which were culture + for Brucella in the organ with the lesion
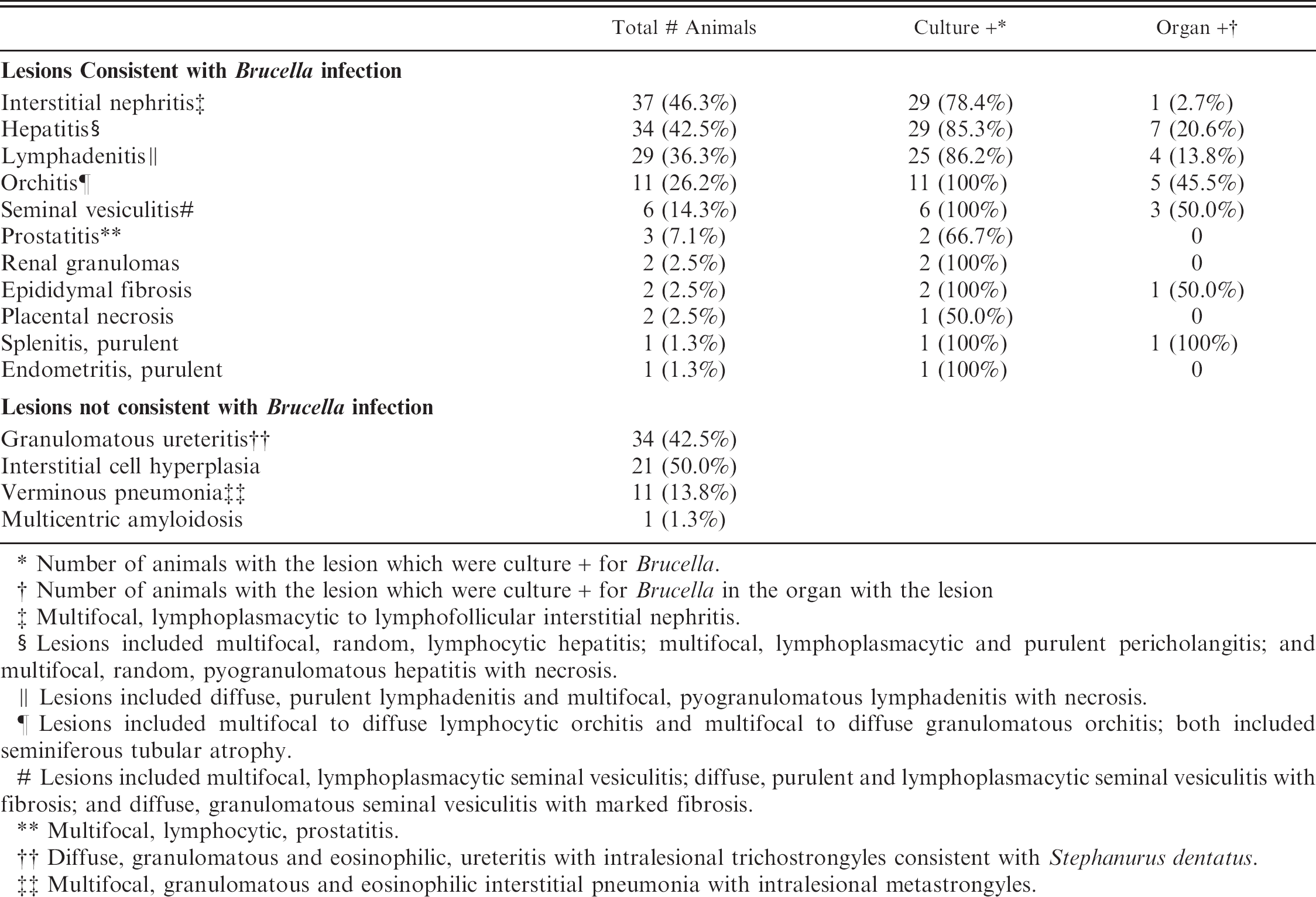
Multifocal, lymphoplasmacytic to lymphofollicular interstitial nephritis.
Lesions included multifocal, random, lymphocytic hepatitis; multifocal, lymphoplasmacytic and purulent pericholangitis; and multifocal, random, pyogranulomatous hepatitis with necrosis.
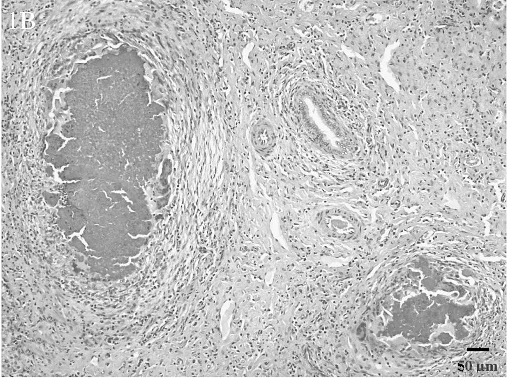
Lesions included diffuse, purulent lymphadenitis and multifocal, pyogranulomatous lymphadenitis with necrosis.
Lesions included multifocal to diffuse lymphocytic orchitis and multifocal to diffuse granulomatous orchitis; both included seminiferous tubular atrophy.
Lesions included multifocal, lymphoplasmacytic seminal vesiculitis; diffuse, purulent and lymphoplasmacytic seminal vesiculitis with fibrosis; and diffuse, granulomatous seminal vesiculitis with marked fibrosis.
Multifocal, lymphocytic, prostatitis.
Diffuse, granulomatous and eosinophilic, ureteritis with intralesional trichostrongyles consistent with Stephanurus dentatus.
Multifocal, granulomatous and eosinophilic interstitial pneumonia with intralesional metastrongyles.
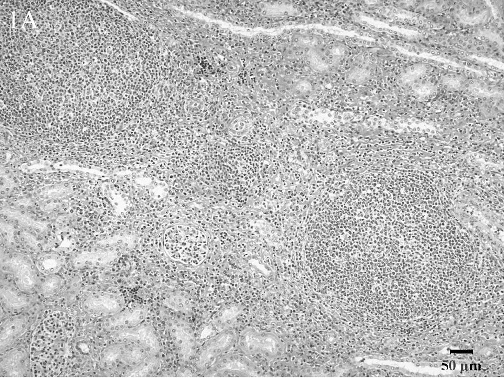
Histologic lesions. Histologic lesions are summarized in Table 2. There was no statistically significant correlation of any lesions with Brucella culture status. Five animals in the study had no significant lesions. Interstitial nephritis was the most common lesion found and was present in 37 animals. This ranged from multifocal to coalescing areas of lymphoplasmacytic infiltrates to large lymphofollicular aggregates in the renal interstitium (Fig. 1A). Hepatitis was present in 34 animals. There were 3 general forms of hepatic inflammation: multifocal, random, lymphocytic hepatitis; multifocal, lymphoplasmacytic and purulent pericholangitis; and multifocal, random, pyogranulomatous hepatitis with necrosis (Fig. 1B). Some of the pyogranulomas were associated with nematode larvae. Lymphadenitis was present in 29 animals. The lymphadenitis was often multicentric and ranged from diffuse, purulent lymphadenitis to multifocal pyogranulomas often with necrotic cores (Fig. 1C).
Histologic lesions within male reproductive organs were common findings. Orchitis was present in 11 males and was characterized by multifocal to diffuse lymphocytic (Fig. 1D) or multifocal to diffuse granulomatous inflammatory infiltrates. All cases of orchitis were associated with significant seminiferous tubular atrophy. Seminal vesiculitis was observed in 6 males and was exhibited in 3 patterns: multifocal, lymphoplasmacytic seminal vesiculitis; diffuse, purulent and lymphoplasmacytic seminal vesiculitis with fibrosis (Fig. 1E); and diffuse, granulomatous seminal vesiculitis with marked fibrosis. Multifocal lymphocytic prostatitis was also present in 3 males (Fig. 1F).
Multifocal renal granulomas were present in 2 animals. Marked epididymal fibrosis was present in 2 males. One animal exhibited multifocal-coalescing, purulent splenitis, and 1 female exhibited diffuse purulent endometritis. No pyogranulomatous or granulomatous lesions in any tissues were associated with fungi or acid-fast bacteria.
Lymphofollicular interstitial nephritis. The renal interstitium is markedly expanded owing to abundant numbers of lymphocytes which are organized into follicle-like structures. Hematoxylin and eosin (HE).
Liver microgranulomas. Hepatic parenchyma is replaced by multiple granulomas with necrotic centers. The area between the microgranulomas contains abundant collagen as well as scattered lymphocytes and plasma cells. HE.
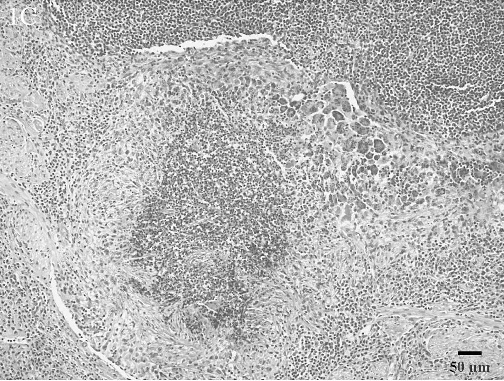
Lymph node, poorly organized pyogranuloma. Abundant neutrophils are surrounded by macrophages and lymphocytes as well as loosely organized fibrous tissue. Numerous multinucleated giant cells also segmentally surround the pyogranuloma. HE.
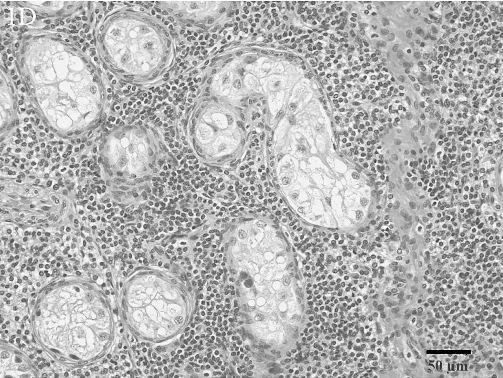
Lymphocytic orchitis with atrophy of the seminiferous tubules. Seminiferous tubules are greatly separated from one another because of abundant numbers of small lymphocytes. The remaining seminiferous tubules are completely devoid of maturing spermatogonia; only Sertoli cells remain within the seminiferous tubules. HE.
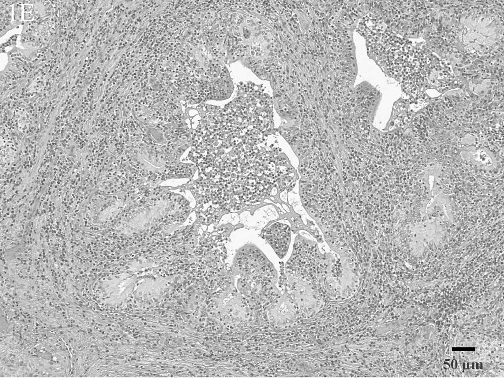
Purulent and lymphoplasmacytic seminal vesiculitis with fibrosis. Periglandular interstitial areas are expanded due to abundant numbers of lymphocytes and plasma cells as well as fibrous tissue. The glandular lumena contain numerous neutrophils and are lacking normal secretory fluid. HE.
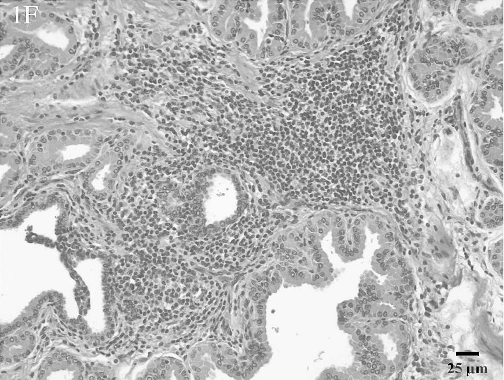
Lymphocytic prostatitis. Periglandular interstitial areas contain abundant numbers of small lymphocytes. HE.
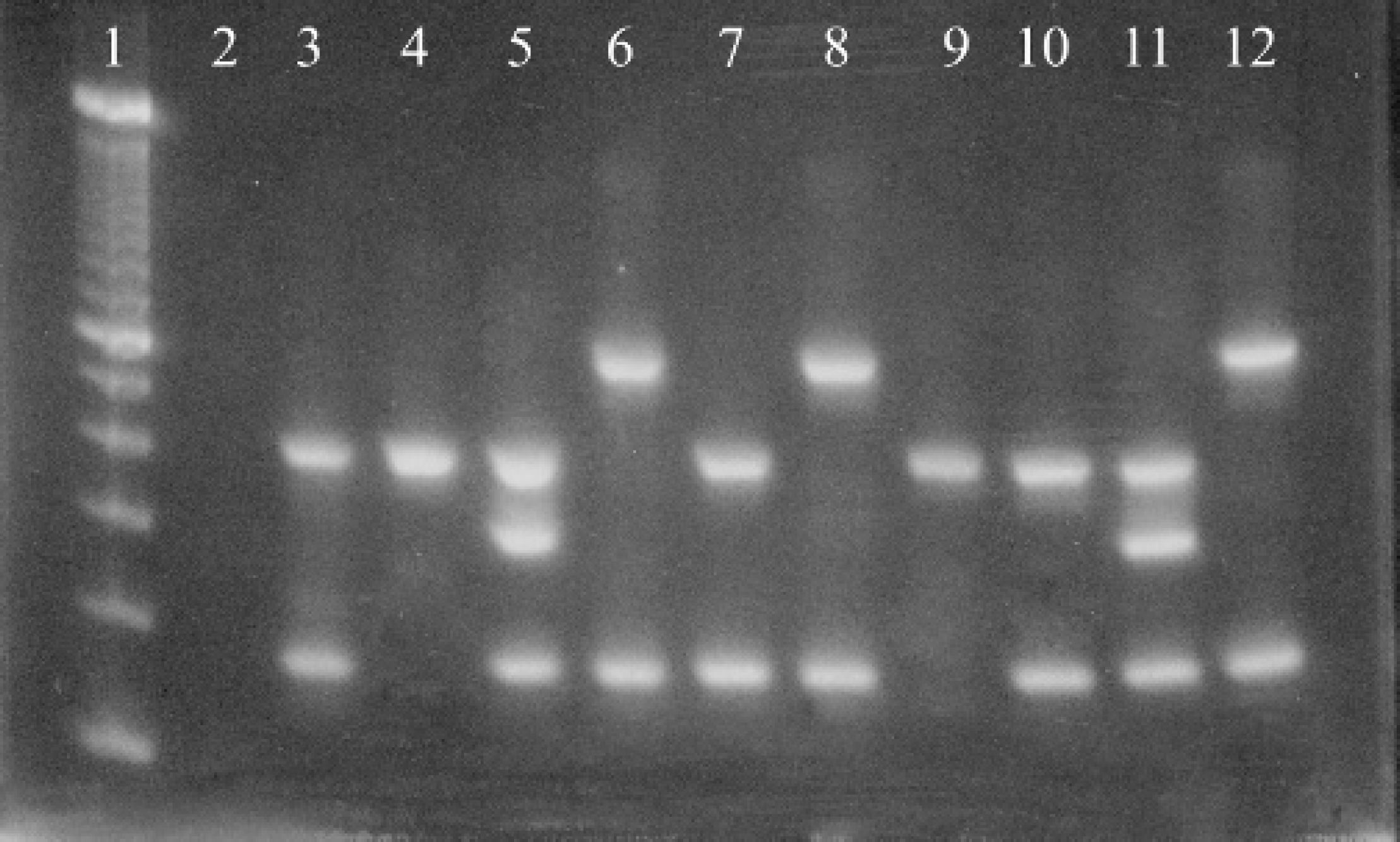
A 2% agarose ethidium bromide stained gel containing products from the multiplex PCR reactions from feral swine Brucella isolates. The lanes contain the following: 1) 100 bp ladder, 2) Negative control, 3–8I Feral swine Brucella isolates, 9) S19 positive control, 10) B. abortus 2308 positive control, 11) RB51 positive control, 12) B. suis 3B positive control.
Lesions that were not consistent with swine brucellosis were also present. Thirty-four animals had eosinophilic and granulomatous ureteritis and periureteritis with intralesional trichostrongyles consistent with Stephanurus dentatus. Moderate to marked interstitial cell hyperplasia was present in the testes of 21 males. Eleven animals exhibited eosinophilic and granulomatous interstitial pneumonia with intralesional metastrongyles, and 1 animal had amyloidosis of the liver, kidney, and spleen.
Bacteriologic culture. Brucella was isolated from a total of 62 (77.5%) feral swine in this study. A multiplex PCR, which could distinguish among B. suis, B. abortus field strains, S19, and RB51, was used in these studies because of the history of using RB51 in this population for vaccine studies. The multiplex PCR assay revealed that not only was B. suis and RB51 present in the population but also B. abortus field strains and S19 were present (Fig. 2). The results of the multiplex PCR assay correlated 100% with traditional Brucella biotyping methods. All B. suis isolates were biovar 1 and all B. abortus field strain isolates were also biovar 1. Table 3 lists the species of Brucella isolated on a per animal basis. Isolation of B. suis only was the most common finding; however, multiple species were isolated from 21 animals (26.3% of the total population; 33.9% of the culture positive animals). B. suis was isolated from 55 animals (68.8% of the total population; 88.7% of the culture positive animals). B. abortus was isolated from 28 animals (35.0% of the total population; 45.2% of the culture positive population). Of these 28 B. abortus positive animals, biovar 1 field strains were isolated from 21 animals (26.3% of the total population; 33.9% of the culture positive population); S19 was isolated from 8 animals (10.0% of the total population; 12.9% of the culture positive population); and RB51 was isolated from 6 animals (7.5% of the total population; 9.7% of the culture positive population). Table 4 lists the number of isolates made for the various species on a per tissue basis. In general there tended to be a dominant species in each tissue even in animals that were infected with multiple species. However, isolation of multiple species of Brucella in particular tissues was achieved.
Bacteriologic culture results. The species of Brucella obtained from 80 feral swine is given on a per animal basis.
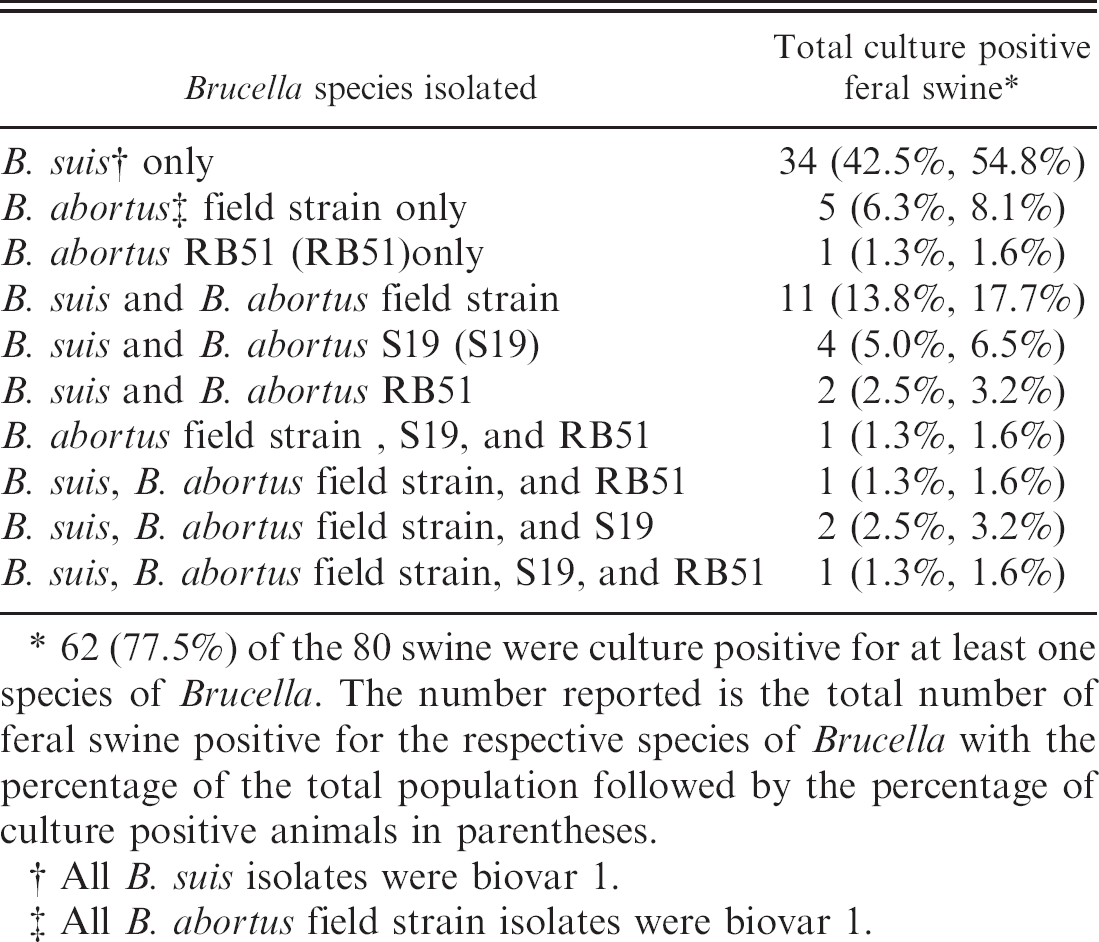
62 (77.5%) of the 80 swine were culture positive for at least one species of Brucella. The number reported is the total number of feral swine positive for the respective species of Brucella with the percentage of the total population followed by the percentage of culture positive animals in parentheses.
All B. suis isolates were biovar 1.
All B. abortus field strain isolates were biovar 1.
Males were significantly more likely to be Brucella culture positive than females (P < 0.001). The culture positivity rate was 92.9% (39/42) for males and 60.5% (23/38) for females. There was no statistically significant difference between males and females in the culture positivity rates for any individual species of Brucella found in this study when the results were compared among culture positive animals only or among the entire population. Among culture positive animals, females were more often infected with multiple Brucella species than males (60.9% for females vs. 20.5% for males; P < 0.01); however, there was no significant difference when the data was compared among the entire population.
Brucella culture results by tissue. The results of bacteriologic culture of tissues from 80 feral swine is shown on a per tissue basis as well as the number of each species isolated for each given tissue.
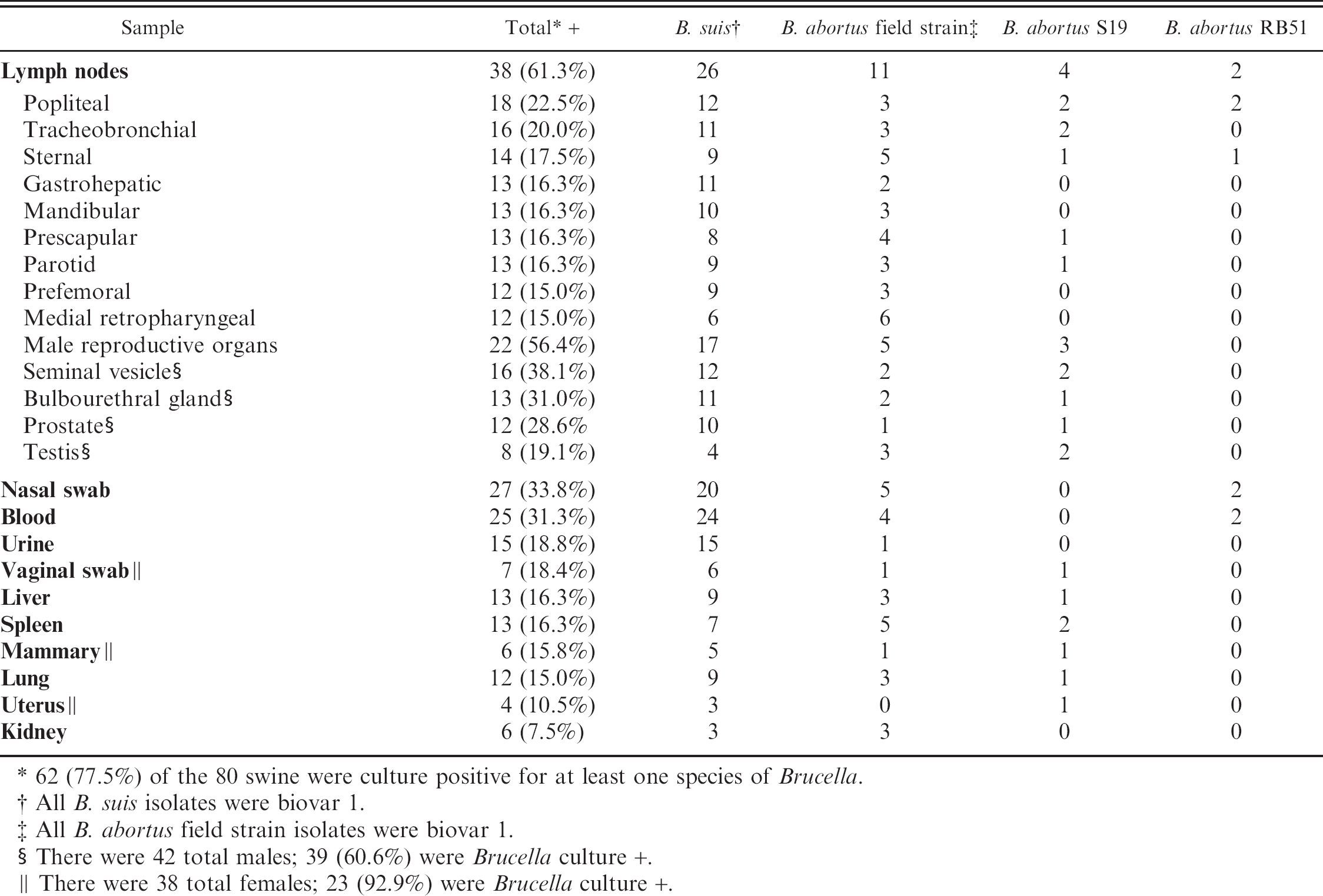
62 (77.5%) of the 80 swine were culture positive for at least one species of Brucella.
All B. suis isolates were biovar 1.
All B. abortus field strain isolates were biovar 1.
There were 42 total males; 39 (60.6%) were Brucella culture +.
There were 38 total females; 23 (92.9%) were Brucella culture +.
VNTR patterns of selected B. suis biovar 1 isolates from feral swine. Isolate or strain designation.
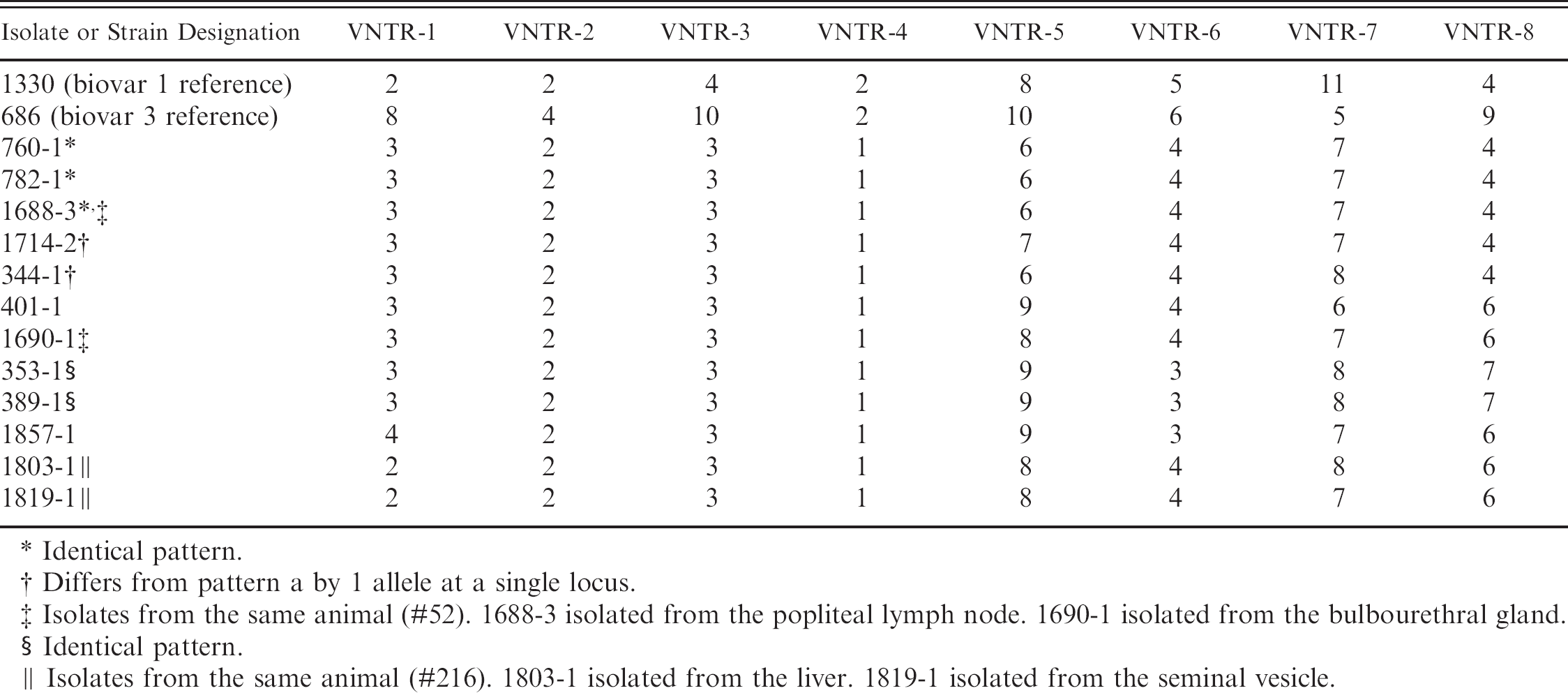
Identical pattern.
Differs from pattern a by 1 allele at a single locus.
Isolates from the same animal (#52). 1688-3 isolated from the popliteal lymph node. 1690–1 isolated from the bulbourethral gland.
Identical pattern.
Isolates from the same animal (#216). 1803–1 isolated from the liver. 1819–1 isolated from the seminal vesicle.
VNTR patterns of Selected B. abortus isolates from feral swine. Isolate or strain designation.
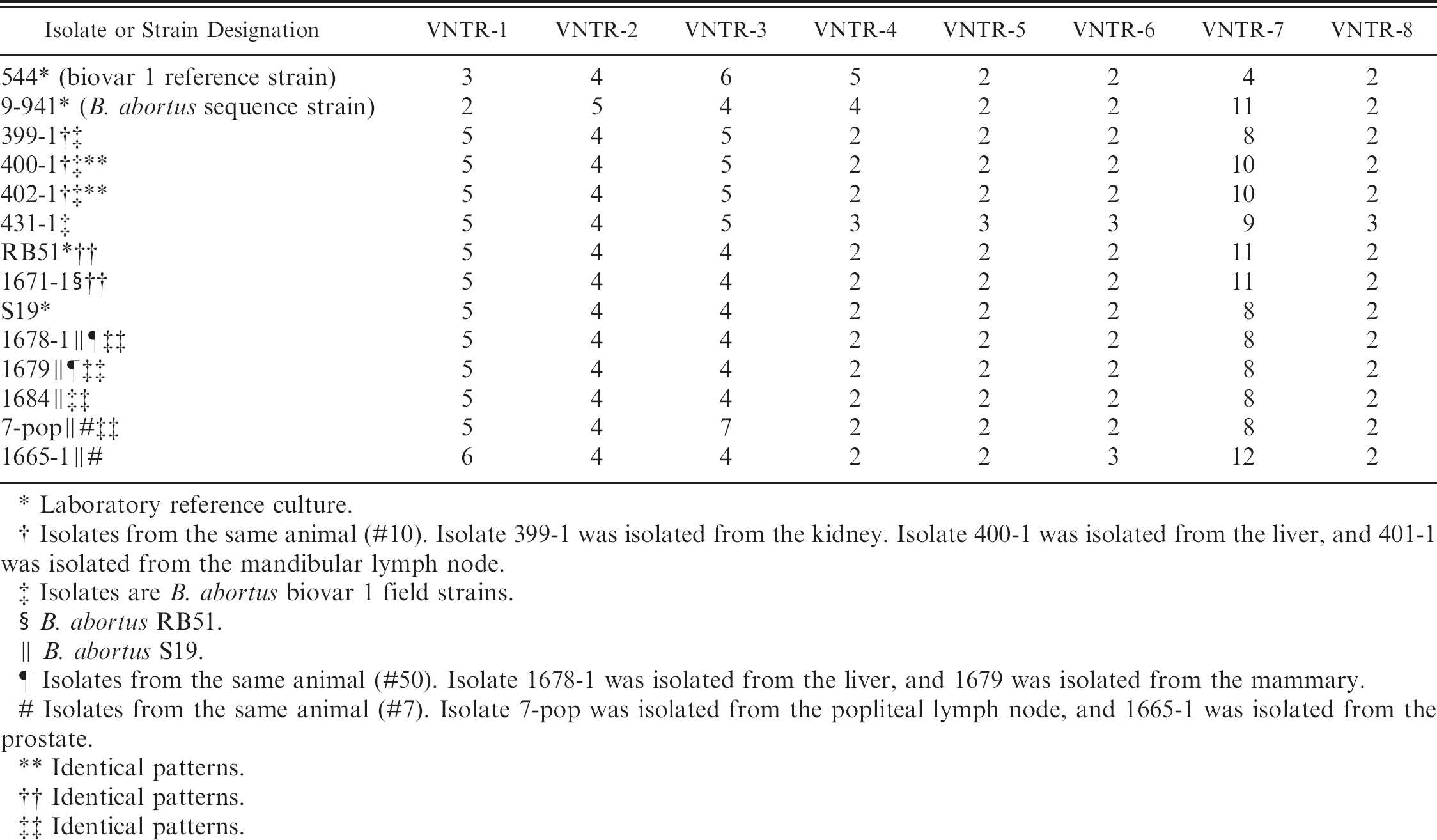
Laboratory reference culture.
Isolates from the same animal (#10). Isolate 399-1 was isolated from the kidney. Isolate 400-1 was isolated from the liver, and 401-1 was isolated from the mandibular lymph node.
Isolates are B. abortus biovar 1 field strains.
B. abortus RB51.
B. abortus S19.
Isolates from the same animal (#50). Isolate 1678-1 was isolated from the liver, and 1679 was isolated from the mammary.
Isolates from the same animal (#7). Isolate 7-pop was isolated from the popliteal lymph node, and 1665-1 was isolated from the prostate.
Identical patterns.
Identical patterns.
Identical patterns.
S19 sequencing. Four isolates that typed as S19 by the multiplex PCR assay and by traditional biotyping were selected for sequencing of the eri gene to determine if the deletion within the eri gene matched the laboratory reference strain of S19. All 4 isolates and the S19 laboratory strain yielded the expected PCR product of approximately 361 bp. The resulting sequences from all 4 of the feral swine isolates were 100% homologous to the sequence obtained from the S19 laboratory strain.
Serology. The results of the serologic assays are listed in Table 7. The seropositivity rates were 48.8% for the entire population, 61.9% for males, and 34.2% for females. Males were significantly more likely to be seropositive among the entire population (P < 0.05); however, there was no difference in seropositivity rates between males and females among culture positive animals.
Relative sensitivity rates for each assay and a combination of the 3 assays are also listed in Table 7. To estimate the sensitivity the number of seropositive animals from the culture positive group was divided by the number of culture positive animals. One animal yielded only RB51 on bacteriologic culture, and that animal was excluded from the sensitivity estimates as RB51, being O-polysaccharide deficient, does not induce detectable immune responses by the 3 assays used in this study. 21 , 29 The sensitivity rates of both the STT (P < 0.0001) and FPA (P < 0.005) assays were significantly higher than the Card Test but did not differ from each other.
Serologic results. Results of serologic assays from necropsy sera from 80 feral swine.
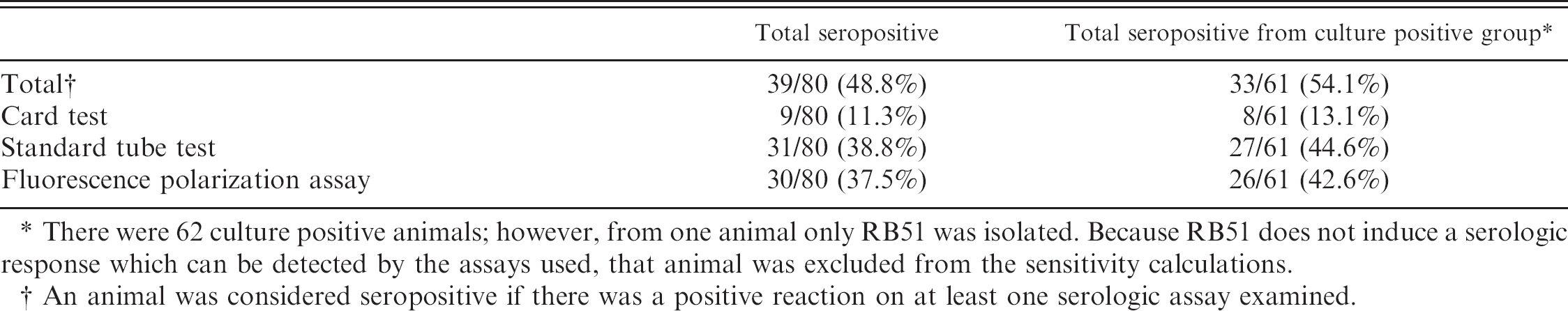
There were 62 culture positive animals; however, from one animal only RB51 was isolated. Because RB51 does not induce a serologic response which can be detected by the assays used, that animal was excluded from the sensitivity calculations.
An animal was considered seropositive if there was a positive reaction on at least one serologic assay examined.
Discussion
In this study 80 feral swine from an area of enzootic feral swine brucellosis on the Atlantic coast of South Carolina were trapped, euthanized, and necropsied to determine Brucella culture, serologic, and lesion profiles. It was found that not only were the feral swine in this herd infected with biovar 1 of B. suis but also with biovar 1 field strains of B. abortus and the vaccine strains B. abortus S19 and B. abortus RB51. This is the first report of B. abortus in feral swine and the first report of a wildlife reservoir of B. abortus outside the Greater Yellowstone National Park Area of Wyoming, Montana, and Idaho.
The finding of well established B. abortus infection in this feral swine herd was surprising not only because it had never been reported but also because of experimental evidence that shows that swine become only briefly infected with B. abortus when experimentally challenged with B. abortus. 29 Previous studies in domestic swine have shown that RB51 is cleared quickly and persists only to 4 weeks in lymph nodes draining the site of infection. 29 In the current study, however, B. abortus was found in a variety of tissues including whole blood suggesting that bacteremia and systemic distribution of the B. abortus occurs in feral swine.
The introduction of B. abortus S19 and biovar 1 field strains in this feral swine herd was possibly caused by scavenging of aborted fetuses of dead, B. abortus infected cattle at a time when cattle were kept on the same property or in the vicinity. The introduction of RB51 is easily explained by the use of this herd in experiments evaluating the potential use of this vaccine in feral swine. However, no such experiments were ever conducted in this herd with the S19 vaccine strain. Examination of property records suggests a longstanding B. abortus infection in this feral swine herd. No cattle have been kept in the area of the feral swine since at least 1970. The property is bordered by the Atlantic Ocean, the Winyah Bay, and a coastal housing development and country club, which greatly limit the migration of feral swine into and out of the area. The VNTR data supported long standing infections of B. suis, B. abortus biovar 1 field strain, and B. abortus S19 because 3 isolates had allelic differences at multiple loci. However, all RB51 isolates examined showed the same pattern as the reference strain of RB51.
The Brucella culture positivity rate in this feral swine herd was high, which reflects the tendency of swine to develop chronic Brucella infections. The serologic sensitivity rates were much lower than those previously reported for the assays used in this study. The results of this study serve as an example for the lack of sensitivity of serologic assays for diagnosing brucellosis in individual feral swine. When necropsy culture results were compared to necropsy serology results, the sensitivity of a combination of the 3 serologic assays that were used was 54.1%. Previously published sensitivity rates for these assays in domestic swine were 20–67% for the card test, 83–100% for the standard tube test, and 80–94% for the FPA test. 13 , 18 , 21 The sensitivity rates for individual assays calculated in this study, however, were 13.1% for the card test, 44.6% for the standard tube test, and 42.6% for the fluorescence polarization assay. Antigens used in these assays are from B. abortus, which may at least partially account for the lack of sensitivity when evaluating swine presumably infected with B. suis. The lack of sensitivity may also be reflected in the chronicity of infection in the animals in this study. Samples used to determine sensitivity rates in previously published reports were obtained from acute outbreaks of swine brucellosis. The relative sensitivity rates of these serologic assays have not been previously investigated in enzootically infected herds. Antibody decay may occur despite the persistence of culturable bacteria in tissues.
This study used a multiplex PCR assay for the identification of Brucella species. The assay could definitively identify B. suis, B. abortus RB51, and B. abortus S19. The assay could also identify Brucella species other than B. suis and the 2 B. abortus vaccine strains; however, it could not differentiate among the other species. Within this assay, a single primer pair was used to differentiate between B. suis and other Brucella species (B. abortus and B. melitensis). These primers (42797 forward and reverse) were targeted to a location on chromosome II at which B. suis has a 189 bp insertion. This size difference allowed easy discrimination of B. suis from the other Brucella species on agarose gels. Bricker has previously reported on the use of multiplex PCR (AMOS assay) to discriminate Brucella species. 4 , 5 The AMOS assay used species differences in the IS711 insertion sequence and required individual primers for species identification. 4 Fayazi and colleagues have also reported on the use of a PCR assay with a single primer pair that can distinguish B. suis from B. abortus. 10 The primer pairs used by Fayazi were unsuccessful in identifying either Brucella species in the studies reported in this paper. Upon examination of the genetic sequence used to design Fayazi's primers, it was found that the sequence was an assembly of 4 unique, nonlinked MboI fragments. The corresponding sequences are as follows: 1) Nucleotides 1–149 of the Fayazi sequence align to nucleotides 1595585–1595734 of chromosome I of B. abortus 9–941; 2) nucleotides 146–200 of the Fayazi sequence align to nucleotides 225993–225939 of chromosome I of B. abortus; 3) Nucleotides 195–330 of the Fayazi sequence align to nucleotides 582796–582668 of Chromosome II B. abortus; and 4) Nucleotides 336–482 of the Fayazi sequence align to nucleotides 53279–538430 of chromosome II of B. abortus. 15 The forward primer used in Fayazi's assay corresponds to positions 1595587–1595611 of chromosome I of B. abortus, and the reverse primer corresponds to positions 538397–538421 of chromosome II of B. abortus. In light of the sequence data, it is easily explained that the primers used in the Fayazi PCR assay could not identify either Brucella species in the current study. Therefore, it appears that the current study is the first report of a single primer pair that can distinguish B. suis from B. abortus and B. melitensis.
Animals within this herd had histologic lesions in multiple organs that were consistent with swine brucellosis 19 ; however, none of these lesions could be significantly correlated to Brucella infection within the animal or within the tissue containing the lesion. Despite a high culture positivity rate, the number of animals with reproductive lesions was surprisingly low. Of the 39 culture positive males in this study, only 14 had lesions in reproductive tissues, and of the 23 culture positive females in this study, only 1 exhibited endometritis and 2 exhibited placental necrosis. In the face of a high level of Brucella within this population, these feral swine manage to maintain a level of reproductive efficiency that supports significant population growth and warrants regular population control strategies to control the amount of environmental damage imposed by the feral swine herd.
The results of this study have identified a feral swine herd that is enzootically infected with B. suis as well as multiple strains of B. abortus. This study shows that within an enzootically infected feral swine population, the seroprevalance rates may be significantly lower than the true infection rate. The results of this study also reiterate the possibility of feral swine to serve as reservoirs of Brucella spp. for both domestic livestock and human infections.
Acknowledgements
We would like to thank Eliza Albrecht, Jimmy Bessinger, Deb Buffington, Jeremy Carpenter, George Chastain, Jean Donald, Travis Duit, Clay Gallup, Aileen Hudspith, Darl Pringle, Rachel Renshaw, and Sharla Van Roeckel for their technical assistance with this project.
Names are necessary to report factually on available data. Mention of trade names or commercial products in the article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture.
Footnotes
a.
Boehringer Mannheim, Indianapolis, IN.
b.
Perkin Elmer, Branchburg, NJ.
c.
MJ Research Inc., Watertown, MA.
d.
Roche Molecular Biochemicals, Indianapolis, IN.
e.
Roche Diagnostics, Basel, Switzerland.
f.
Geneclean II kit, Q-Biogene, Irvine, CA.
g.
Molecular Probes, Eugene, OR.
h.
Applied Biosystems Inc., Foster City, CA.
i.
Gene Codes Corp., Ann Arbor, MI.
j.
PROC FREQ CHISQ, SAS Statistical Software, SAS Institute, Inc., Cary, NC.
