Abstract
Reproductive failure in sows is one of the most important factors affecting pig breeding. Many reproductive disorders are linked to both environmental factors and infectious agents. The goal of our study was to determine the presence of pathogens that are known to cause abortion, considering a set of conditioning factors, such as seasonality and pregnancy period. A large number of aborted fetuses (1,625 fetuses from 140 farms) from a high-density breeding area in northern Italy was analyzed for a period of 3 years. The pigs were diagnosed based on direct (culture, PCR) or indirect (enzyme-linked immunosorbent assay) evidence. An infectious etiologic agent was found in 323 of 549 cases of abortion (58.8%). These included viral agents (Porcine circovirus-2, 138/323; Porcine reproductive and respiratory syndrome virus, 108/323; porcine parvovirus, 20/323; pseudorabies virus, 6/323; and Encephalomyocarditis virus, 3/323) and bacteria (Escherichia coli, 64/323; Streptococcus sp., 63/323; Staphylococcus sp., 5/323; Pasteurella sp., 3/323; Shigella sp., 1/323; and Yersinia sp., 1/323). This study describes the prevalence of infectious agents involved in reproductive failure in a high-density swine population. The data can be useful to swine breeders, practitioners, and medical specialists in monitoring animal health and in supervising the breeding process.
Reproductive failure in sows, like abortion, is a significant factor that increases the cost of pig breeding, especially within high-density breeding areas where infectious agents are transferred more easily. Because reproductive failure in pig herds significantly affects the profits of the farming business, it is important to reduce its impact.9,17,21 The causes of swine abortion can be classified into 2 categories. 28 The first is represented by agents responsible for systemic or local infection of the reproductive organs and by immune deficiency or immunologic disorders. The second category is represented by reproductive failures associated with deviation of the animal’s proper hormonal balance and normal physiologic condition. In effect, multiple deviations may interrupt gestation, determining the death of the embryo or fetus, commonly caused by toxins and environmental or nutritional stress. The first category includes a large number of infectious agents that are directly responsible for death of the fetus and/or placenta infections.8,12,16 Viruses that cause abortion include Porcine reproductive and respiratory syndrome virus (PRRSV), pseudorabies virus (PRV; Suid herpesvirus 1), porcine parvovirus (PPV), porcine enteroviruses (PEVs), Classical swine fever virus (CSFV), encephalomyocarditis virus (EMCV; Cardiovirus A), and Porcine circovirus-2 (PCV-2). Bacteria associated with abortion in pig herds include Escherichia coli, Streptococcus sp., Staphylococcus sp., Erysipelothrix rhusiopathiae, Leptospira sp., and Brucella suis.
The study of multiple agents that cause swine reproductive failure is quite rare, and requires a large bulk of data collected by observing a large number of animals for a relatively extended period.20,25 Knowledge of the most predominant and common causes of this problem can help monitor animal health and manage swine farms. The purpose of our 3-year study was to identify the presence of infectious agents likely to cause abortions, with the aim of providing a useful tool for the development of good farming practices in an area with a high density of breeding pigs. Our study focused on 1,625 fetuses, from 549 sows with an average of 3 fetuses per sow, submitted to the Diagnostic Laboratory of Animal Diseases of the Istituto Zooprofilattico Sperimentale della Lombardia ed Emilia Romagna (IZSLER) in Brescia, Italy between 2011 and 2013. Cases of reproductive failure occurred in a total of 140 different conventional farms that use various production systems (including farrow-to-finish, farrow-to-wean, and multiple-site herds).
The etiologic diagnosis was performed by direct (culture, PCR) or indirect (enzyme-linked immunosorbent assay [ELISA]) evidence. Indirect methods can support or even replace the direct methods for some agents, despite their limitations, such as the late development of fetal immunity during pregnancy. Aborted fetuses were subjected to thorough autopsies as previously described (Luppi A, Sample collection, handling and preservation in swine pathology, In: Proceedings of XXXVII SIPAS, 2011:39–47, Piacenza, Italy, March 24–25; Alborali L, et al., Diagnostic approach to sow reproductive pathology, In: Proceeding of XXXII SIPAS, 2006:71–80, Modena, Italy, March 23–24). Samples of lung, heart, liver, and brain tissue, and thoracic fluid were collected from each fetus. Organs were pooled, homogenized (10% w/v in modified Eagle medium), a and then analyzed for the presence of viruses. More specifically, PRRSV and PCV-2 were found using reverse transcription (RT)-PCR.b,c,28 Viruses such as PRV, PPV, PEVs, CSFV, and EMCV were found by infection of sensitive cell lines.a,10,32 In addition, PRV and PPV were indirectly found by serologic analysis of fetal thoracic fluid, via competitive ELISA.c,28 Bacteria were isolated and identified in brain, lung, and liver tissues from all sampled fetuses in cultures on solid agar plates.a,28 Leptospira spp. were not surveyed because of the extreme difficulty achieving a reliable identification.
Abortion occurred mainly in the fall (28.2%) and winter (35.5%). The highest number of cases (72) was reported in February, but no significant difference in the prevalence of infectious agents was found over the year. This suggests the possible involvement of different factors, such as environmental or nutritional stressors, which have not been investigated in this study.
Most of the cases of abortion occurred in the second half of pregnancy (71%), as shown in Figure 1. This is likely a consequence of the difficulty finding small fetuses aborted in the first half of gestation in the sow’s bedding, or of the possible occurrence of fetal resorption, which could be mistaken as a fertility problem, as a result of the sow returning to the estrus or anestrus stage. For a comprehensive assessment of sow reproductive problems, sampling should include vaginal swabs and boar semen, not only in order to identify infections agents, but also to assess the fertile quality of sperm. 23
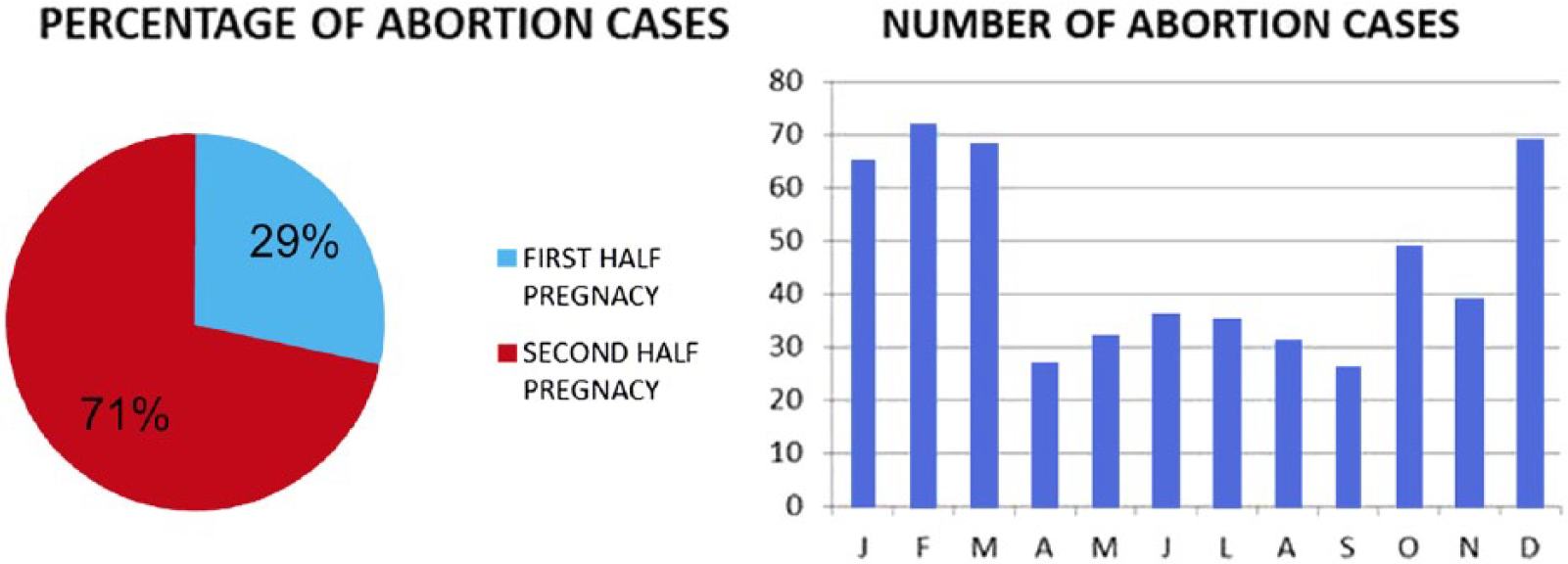
The pie chart represents the percentage of abortion in the first and second stage of pregnancy, respectively. The bar chart represents the seasonality of abortions.
Autopsy findings in our study, such as systemic visceral congestion or the presence of blood-tinged fluid in body cavities, are common nonspecific alterations in aborted fetuses that cannot be associated with any specific pathogen. 12
Infectious etiologic agents were found in 323 of 549 cases (58.8%). None were found in 226 (41.2%) of 549 fetuses submitted. The detection rate in aborted swine fetuses was consistent with that of previously conducted studies.5,24,25 Viral agents were identified in most of the cases: PCV-2 (138/323), PRRSV (108/323), PPV (20/323), PRV (6/323), and EMCV (3/323). No traces of CSFV and PEVs were found.
PRV and PRRSV are widespread in European pig farms, causing systemic diseases and abortion. In unvaccinated pregnant sows, the viruses can trigger hyperthermia, and abortion is a consequence of viremia without infection of the fetuses. These viruses are chiefly found in the female genital tract and in fetal tissues in later pregnancy. 31 In particular, PRRSV transmission by the mother to the fetus and virus replication in the endometrium and placenta are confined to advanced gestation. 13
In our study, the low number of detected cases of PRV (6/323) reflects the situation of the investigated area, where pigs are vaccinated against this virus. Conversely, PRRSV prevalence was high (108/323) caused by the ability of virus to spread within the pig population. This high mutation frequency of PRRSV makes it difficult to limit the disease by vaccination. 23 PPV showed low prevalence (20/323) and was associated with abortion only in pregnant naïve sows, and was asymptomatic in the other cases. 23 Vaccination against PPV is normally effective to reduce the prevalence of abortion. 3 EMCV is generally associated with systemic infections and myocardial necrosis in pigs. Particularly during pregnancy, EMCV can reproduce in the fetal heart and cause abortion or stillbirth. 14 The prevalence of symptomatic infection depends on the serotype involved and the age of the pigs. 22 EMCV is endemic in the investigated area and was found in 3 of 323 cases of abortion. Vaccination is available, but generally not applied. PCV-2 was found in 138 of 323 cases. PCV-2 is involved in multiple diseases in swine, including systemic, enteric, and respiratory disorders, and porcine dermatitis and nephropathy syndrome. The virus can also infect the placenta and fetus, causing reproductive failure.6,27,29 Vaccination is available and widely used.19,26 It is normally administered to piglets and only rarely to sows in the farms involved in this study.
The bacteria found in our study were Escherichia coli (64/323), Streptococcus sp. (63/323), Staphylococcus sp. (5/323), Pasteurella sp. (excluding P. multocida; 3/323), Shigella sp. (1/323), and Yersinia sp. (1/323), with varying prevalence. Brucella suis was not investigated because the study area is officially free of this bacterium. Bacteria such as Staphylococcus sp., Pasteurella sp., Yersinia sp., and Shigella sp., considered opportunistic agents in our study, are occasionally observed in the genital tract of sows and in aborted fetuses. 31 In addition, other bacteria, such as E. coli and Streptococcus sp., are widely detected. E. coli is a typical enteric pathogen, causing diarrhea in piglets. 7 Streptococcus sp. is the most important agent of septicemia in piglets and post-weaning animals. 30 Abortion caused by these bacteria is as a result of retrograde infections from the open cervix and rarely a result of hematogenous placental infection. Antimicrobials are commonly used in farm practice to control these bacterial infections.
The prevalence of infectious agents, reported in Table 1, points to an association of multiple pathogens in different percentages (depending on the pathogen involved). Although viruses, such as PRRSV and PCV-2, and bacteria, such as E. coli and Streptococcus sp., are sufficient to induce abortion on their own, the data collected in our study show that they are often associated.13,20,27,31 In our study, we cannot speculate about the evidence of this multi-agent action, as the autopsy findings do not suggest specific infections, and no histopathology was conducted. In conclusion, we can infer that the main infectious pathogens involved in reproductive failure in sows in the investigated area are related to a small number of common agents.
Prevalence of infectious agents: number and percentage of pure and associated cases.*
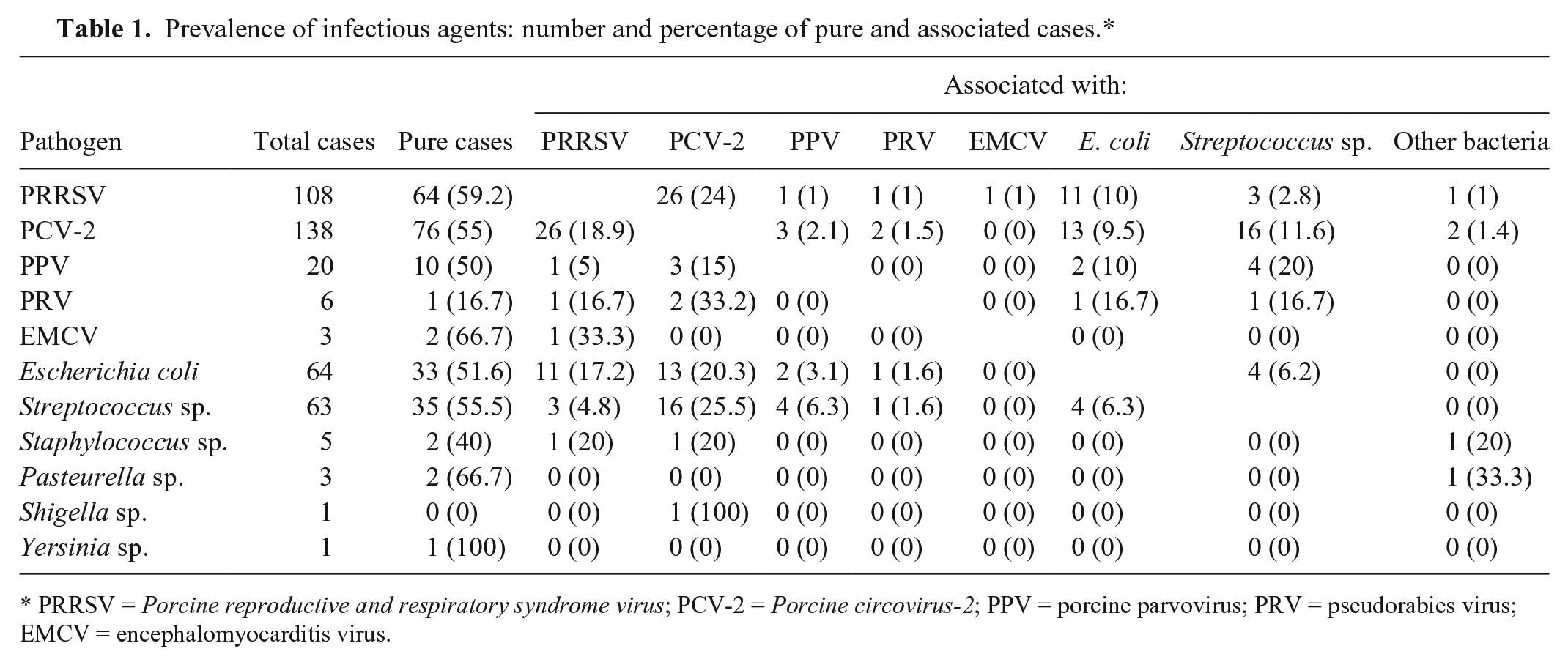
PRRSV = Porcine reproductive and respiratory syndrome virus; PCV-2 = Porcine circovirus-2; PPV = porcine parvovirus; PRV = pseudorabies virus; EMCV = encephalomyocarditis virus.
Considering that gross lesions are infrequently observed in swine fetuses and, when observed, are often not specific to a particular etiology, it is important for practitioners or medical specialists to apply a consistent protocol of collecting and sampling fetuses to examine possible infectious agents involved in abortion. This can give us a better overall sense of the health of pigs on each farm.11,15 Finally, health surveillance can be useful to support farmers in the provision, application, and improvement of biosecurity protocols, good management practices, vaccination, and antibiotic therapy in the control of reproductive failure on pig farms.1,2,4,11,15,18
Footnotes
Acknowledgements
We thank the project manager of Scienze & Tecnologie Studio Moretto Group Srl, Italy for assistance in drafting the English version.
Authors’ contributions
C Salogni and GL Alborali contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; and critically revised the manuscript. M Lazzaro and MB Boniotti contributed to design of the study; contributed to acquisition and analysis of data; drafted the manuscript; and critically revised the manuscript. E Giacomini contributed to conception of the study and contributed to acquisition of data. S Giovannini, M Zanoni, P Pozzi, and P Pasquali contributed to conception and design of the study; contributed to analysis and interpretation of data; drafted the manuscript; and critically revised the manuscript. M Giuliani contributed to design of the study and contributed to acquisition of data. J Ruggeri contributed to conception of the study; contributed to interpretation of data; drafted the manuscript; and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Diagnostic Lab, Istituto Zooprofilattico Sperimentale della Lombardia e dell’Emilia Romagna “Bruno Ubertini,” Brescia, Italy.
b.
One-step RT-PCR kit, Qiagen GmbH, Hilden, Germany.
c.
PCV2 DNeasy blood and tissue kit, Qiagen GmbH, Hilden, Germany.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
