Abstract
Two follow-up studies of a positive methicillin-resistant Staphylococcus aureus, (MRSA) finding in the 2008 European Union baseline survey on MRSA in pig herds were performed to gain more knowledge about the epidemiology of the particular MRSA type, a known human type (ST8/t008), among pigs. Two persons on a Norwegian farm in the study were found to be MRSA carriers, and human-to-animal transmission was suspected. In the first follow-up study, all pigs (n = 346) were sampled by taking nasal swabs. A pooled sample from 5 individual pigs housed together in a single pen, and a dust sample from the equipment in the same room, were positive. Dust samples from a building housing MRSA-negative animals were negative. The MRSA was not detected in the second follow-up, after removing positive animals from the farm and cleaning and disinfecting. A low MRSA occurrence among the animals was found, suggesting that MRSA ST8/t008 may be less able to colonize and persist in pig holdings compared with more host-adapted S. aureus strains.
Keywords
In 2008, the European Union-initiated baseline survey on the prevalence of Salmonella spp. in herds of breeding pigs in the European countries also included a survey of methicillin-resistant Staphylococcus aureus (MRSA) in the same holdings. 3 The investigations were initiated as recent reports had documented high prevalence of MRSA in pig holdings. The emergence of MRSA among pigs was first described from the Netherlands, 8 and typing studies have shown that most MRSA isolates from pigs are related and belong to sequence type 398 (ST398). 1,8 Recent studies have shown that MRSA ST398, also referred to as livestock-associated MRSA, occurs with high prevalence in pig holdings in several European countries. 10 Investigations have also documented MRSA ST398 among pigs in Canada and China. 6,9 MRSA ST398 is presumably an animal-adapted MRSA type that seems to have a strong ability to colonize and persist among livestock animals, particularly pigs.
In Norway, a total of 252 holdings were included in the 2008 MRSA baseline survey. 3 Standardized sampling protocols and laboratory methods were used by all participating countries, and the material investigated was dust samples from the herds as instructed by European Food Safety Authority (EFSA). 3 Only 1 dust sample from a farrow-to-finish pig herd was positive for MRSA, yielding a prevalence of 0.40% (95% confidence interval: 0.01–2.19%). The isolate was further genotyped at the Norwegian Reference Laboratory for MRSA at St. Olavs Hospital (Trondheim, Norway), as well as at the EU Reference Laboratory for antibiotic resistance in Denmark (www.crl-ar.eu). Genotyping using multilocus sequence typing a–c , 2 showed that the isolate belonged to ST8 (allelic profile 3–3–1–1–4–4–3); spa typing a–c , 4 showed spa type t008 (spa-repeats: 11–19–12–21–17–34–24–34–22–25). The strain contained a SCCmecIV element a , 7 and was Panton-Valentine leukocidin (PVL) negative. a Susceptibility testing using a broth microdilution method d and break points applied by the Norwegian monitoring program for antimicrobial resistance in the veterinary sector (NORM/NORM-VET: 2008, Usage of antimicrobial agents and occurrence of antimicrobial resistance in Norway. Available at http://www.vetinst.no/eng/Research/Publications/Norm-Norm-Vet-Report/Norm-Norm-Vet-report-2008. Accessed on November 17, 2010) showed that the isolate was resistant to fusidic acid in addition to β-lactams. Resistance to tetracycline was not observed, in contrast to most MRSA ST398 from pigs. 10
The MRSA prevalence among human beings in Norway is low; 0.7% of S. aureus isolates from wounds and bloodstream infections are classified as MRSA (NORM/NORM-VET: 2008, Usage of antimicrobial agents and occurrence of antimicrobial resistance in Norway). MRSA belonging to spa type t008 is the second most common genotype isolated from human beings with MRSA infections in Norway (NORM/NORM-VET: 2008, Usage of antimicrobial agents and occurrence of antimicrobial resistance in Norway). Because Norway has a low prevalence of human MRSA infections, an active search and destroy policy is practiced. In connection with the MRSA baseline survey, the authorities decided that follow-up testing of human beings associated with MRSA-positive farms should be performed to identify persons at risk of being MRSA positive. This action plan was initiated to prevent introduction of MRSA into hospitals and nursing homes.
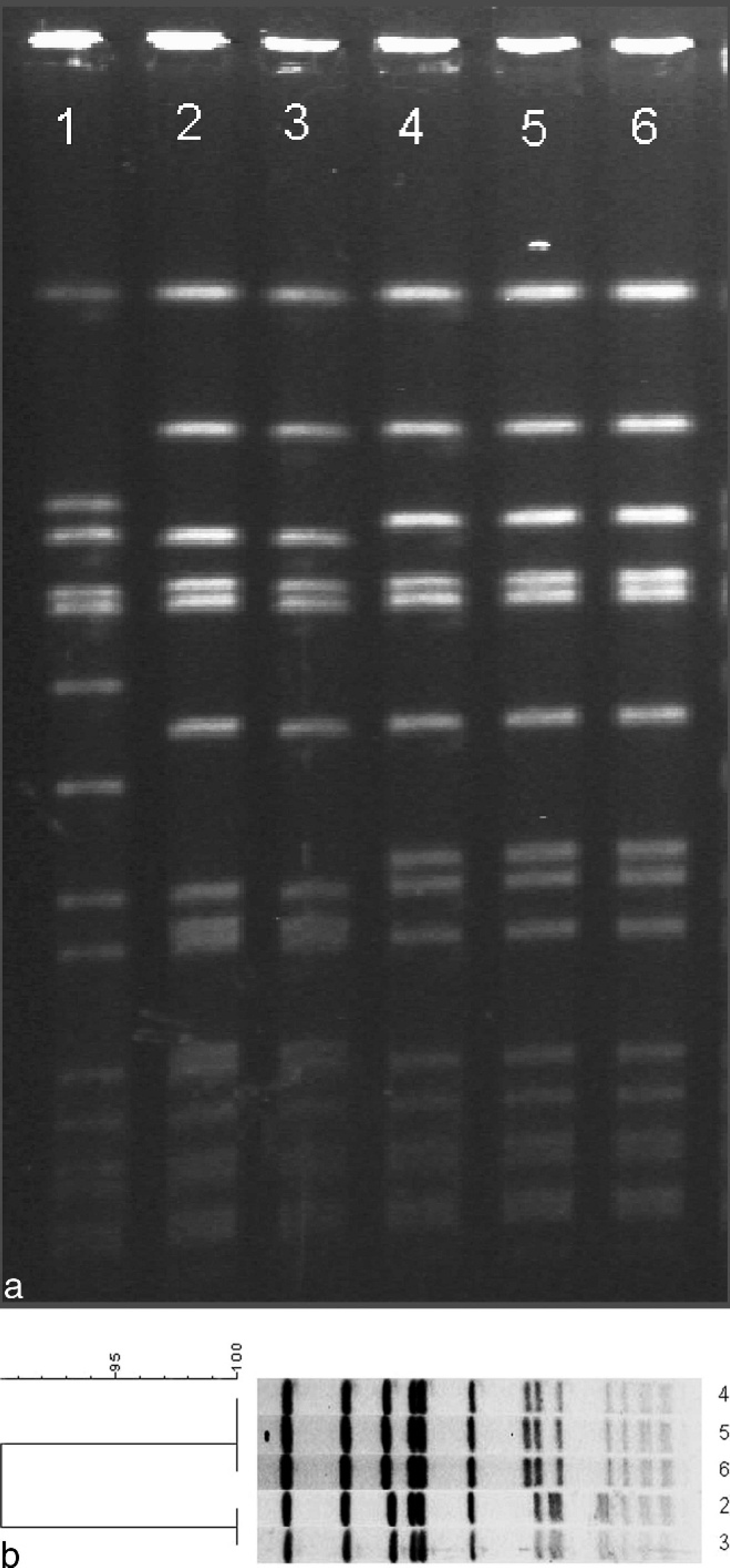
Pulsed-field gel electrophoresis (
MRSA screening of all family members on the MRSA-positive farm showed that 2 persons were positive. Both MRSA carriers had regular contact with the pigs.
Genotyping of both isolates showed the same MRSA type as detected from the dust sample analyzed in the baseline study. Treatment to eradicate MRSA carriage in the 2 MRSA-positive human beings was initiated, but the risk of MRSA recontamination from the pigs was considered as a possible threat. However, MRSA colonization status of the pigs was uncertain, as the samples investigated in the baseline survey were dust samples, and not samples from the animals.
Knowledge about colonization and persistence of known human MRSA types among pigs is limited. To gain more knowledge about the MRSA type involved and its eventual occurrence among animals on the pig farm, follow-up investigations were carried out. In the first follow-up, which was performed approximately 2 months after the positive finding in the baseline survey, swabs from the nostrils of all pigs and dust samples representing dust from all pens were taken. There were 2 separate buildings housing swine on the farm, 1 of them housing fattening pigs only. At the first follow-up sampling, the herd had 346 pigs (51 sows, 1 boar, 183 piglets, and 111 fatteners), and the fattening pigs were housed in a single compartment with a continuous flow of pigs. There were 10–12 pigs per pen, 1.2 m 2 per pig, and a low-pressure ventilation system with automated regulation of the temperature. The pigs were restrained with a snout rope during sampling, and the swab was placed directly into the nostril. The laboratory method used for investigation of MRSA was the same as used in the baseline survey, 3 except that the swabs were pooled up to 6 together in the initial broth. The initial broth was a Mueller-Hinton broth supplemented with 6.5% NaCl. After overnight incubation 1 ml was inoculated into Trypticase Soy Broth with 3.5 mg/l cefoxitin and 75 mg/l aztreonam and incubated 16–20 hr. The culture was then plated out on Brilliance MRSA agar plates. e Laboratory testing was started on the day of sample collection. Persons involved in the sampling were screened for MRSA before and after visiting the farm, and were found to be negative on both occasions.
The MRSA was detected from 1 pooled sample containing 5 swabs from 5 individual fattening pigs housed together in the same pen. The MRSA was not detected from other pigs in the same pen (4 animals). One dust sample from the building housing the MRSA-positive pigs was also positive. The dust samples from the building housing MRSA-negative animals were negative. Multilocus sequence typing and spa typing showed the same sequence type and spa type as previously detected in the baseline survey and from human beings at the farm. All 5 MRSA isolates (2 of human origin, 1 from fattening pigs, and 2 isolates from dust) were subjected to pulsed-field gel electrophoresis (PFGE) using smaI and a standardized protocol. f–h Two slightly different banding patterns were obtained. The isolate from one of the human beings and the isolate from pigs produced identical banding pattern. The other 3 isolates, 1 from human and 2 from dust, produced identical banding pattern when compared with each other, but this pattern was slightly different from the banding pattern from pigs/human being. The similarities between the 2 patterns obtained suggested that all strains were closely related and had a common origin. All strains also exhibited the same resistance profile, were PVL negative, and contained a SCCmecIV element. The PFGE banding patterns and dendrogram are shown in Figure 1. The MRSA-positive animals and pigs in the neighboring pens were removed from the farm within 2 weeks, and all pens in the fattening unit were cleaned and disinfected with 2% Virkon S. i Three months later, a second follow-up testing was performed. Dust samples from all pens in both buildings were tested. A total of 20 dust cloths were taken (1 cloth per 2 pens), more than the number prescribed by EFSA for the baseline study. 3 Cloths with dust from the ventilator fan were also included. Laboratory testing yielded negative MRSA results on all samples. Follow-up samples from human beings after eradication also yielded negative results, indicating that eradication was successful.
In the present study, a low MRSA prevalence was found in the pigs, in contrast to what has been reported for MRSA ST398 among pigs. 10 An explanation may be that MRSA ST8/t008 is adapted to human beings, without the same ability to colonize and persist among pigs as more host-adapted S. aureus strains, like S. aureus belonging to ST398 or ST9. 5,10 The small herd size with a relative low density of pigs may have also been of importance as high animal density is suspected to be associated with higher prevalence of MRSA.
One possible explanation for the MRSA occurrence in this pig holding may be a human-to-animal transmission. Transmission of MRSA between animals and human beings has usually involved companion animals and horses and their owners and handlers. The close contact between such animals and human beings could facilitate MRSA transmission in both directions. The present results also showed that fattening pigs, their production environment, and human beings harbored the same human MSRA type, indicating that human MRSA variants can be exchanged within production units for livestock.
Footnotes
a.
AmpliTaq Gold, Applied Biosystems, Foster City, CA.
b.
ExoSAP-IT®, USB Corp., Cleveland, OH.
c.
BigDye® Terminator v.3.1 Cycle-Sequencing-Kit, Applied Biosystems, Foster City, CA.
d.
VetMIC™, National Veterinary Institute, Uppsala, Sweden.
e.
Brilliance™ MRSA agar plates, Oxoid Ltd., Cambridge, United Kingdom.
f.
SmaI restriction enzyme, New England Biolabs, Ipswich, MA.
g.
Low Melting Point Agarose, Invitrogen Corp., Paisley, United Kingdom.
h.
Pulsed Field Certified Agarose, Bio-Rad Laboratories, Hercules, CA.
i.
Virkon S, Antac International-a Du Pont Company, Suffolk, England.
