Abstract
Neuroleptospirosis is a rare disease caused by pathogenic Leptospira interrogans in humans; however, it has not been fully studied in animals. A young wild raccoon dog was found convulsing in the recumbent position and died the next day. Histologic examination revealed nonsuppurative meningoencephalitis in the cerebrum, cerebellum, midbrain, and medulla oblongata. The lesions consisted of mixed infiltrates of Iba1-positive macrophages and CD3-positive T cells, with a small number of CD79α-positive B cells and myeloperoxidase-positive neutrophils. In the frontal cortex, perivascular cuffs and adjacent microglial nodules were distributed diffusely, especially in the molecular layer. Glial nodules were comprised of Iba1- and myeloperoxidase-positive activated microglia. Immunohistochemistry revealed leptospires in mononuclear cell perivascular cuffs, but not in glial nodules. Neuroleptospirosis was accompanied by Leptospira-related nonsuppurative interstitial nephritis, pulmonary edema and hemorrhage, and coronary periarteritis, as well as Toxocara tanuki in the small intestine and nonspecific foreign-body granulomas in the lungs and stomach.
Leptospirosis occurs sporadically in animals and humans worldwide, especially in North, Central, and South America, Europe, and Southeast Asia.8,12 Animals and humans are divided into maintenance (reservoir) and accidental (incidental) hosts. Reservoir hosts with asymptomatic infections can transmit Leptospira interrogans to other animals through persistent urinary shedding; incidental hosts that become infected from reservoir hosts can develop severe or fatal disease.10,16 Rodents are the main reservoir host, but other mammalian species, such as dogs, cattle, pigs, and wild animals, can also become reservoir hosts.8,10,12,16 Incidental hosts might shed pathogenic Leptospira for a short duration, but do not establish a chronic carrier state. 16 In a survey conducted in the United States using nuisance or trapped animals, wildlife, such as canids, cervids, small Indian mongooses, raccoons, and striped skunks, had >30% antibody prevalence, and these species might be reservoir hosts in the environment. 14 Acute leptospiremia in dogs as incidental hosts can follow infection by a number of serovars of L. interrogans, particularly Canicola and Icterohaemorrhagiae, as well as Autumnalis, Grippotyphosa, Pomona, and Sejroe.4,11,19 Hematogenous spread leads to interstitial nephritis, hepatitis, and pulmonary hemorrhage syndrome.4,10,16 Although the effects of Leptospira on the central nervous system, including meningitis and meningoencephalitis, have been observed in humans,13,18 –20 neurologic findings have not been fully reported in dogs. We report a raccoon dog with convulsions that had nonsuppurative meningoencephalitis and immunopositive leptospires in perivascular cuffs.
A wild, immature male, raccoon dog of unknown age (Canidae, Nyctereutes, Nyctereutes procyonoides), a canid endemic to far eastern Asia, 9 was given veterinary care in Tokyo, Japan, because the animal appeared ill and often convulsed in the recumbent position. However, he died the next day. There were no significant gross findings. The autopsy revealed dark red lungs with white spots (Suppl. Fig. 1A); excess mucus in the tracheal lumen (Suppl. Fig. 1B); nodular enlargement of costal cartilage, suggestive of rickets; pale kidneys (Suppl. Fig. 1C); congested liver (Suppl. Fig. 1D); and hyperemia and swelling of the brain (Suppl. Fig. 2). Thickened gastric mucosa (pyloric region and gastric corpus) and intussusception of the small intestine with intraluminal nematodes were found (Suppl. Fig. 3A–3C). The nematodes were morphologically identified as Toxocara tanuki (Suppl. Fig. 3D). 2 Samples of liver, lungs, kidneys, spleen, heart, stomach, small and large intestines, and lymph nodes were fixed in 10% neutral-buffered formalin, processed routinely, and stained with hematoxylin and eosin and other stains. The spinal cord was not examined. For immunohistochemistry, we used the avidin–biotin–peroxidase complex method (Suppl. Table 1). The chromogen was 3,3′-diaminobenzidine, and hema-toxylin was used for nuclear counterstaining. In negative controls, the primary antibody was replaced with non-immunized sera, and in positive controls, the appropriate reactions were confirmed in epithelial cells, mesenchymal cells, and lymphoid cells of several organs. We used samples from a second wild raccoon dog, which had been found dead in Tokyo, possibly as a result of sepsis, to confirm normal expression of E-cadherin in the liver.
Microscopically, in the cerebrum, nonsuppurative meningitis was characterized by marked subarachnoid infiltrates of lymphocytes, plasma cells, and macrophages, particularly around arterioles (Fig. 1A). Inflammatory cell infiltration was not observed in the choroid plexus or lateral ventricle. In the frontal cortex, there was perivascular lymphohistiocytic cuffing, and microglial nodules were found in the molecular layer (Fig. 1A). Similar nonsuppurative meningitis with infiltrated mononuclear cells and periarteritis was seen in the cerebellum, midbrain, and medulla oblongata. The meninges and parenchyma were congested; the meninges were more congested than the parenchyma. There were no neuronal changes in the gray matter including the cortex, corpus striatum, thalamus, and hippocampus, nor demyelination in the white matter, including the subcortical area and corpus callosum. A small granuloma with multinucleate giant cells was observed in the fimbria of the hippocampus, but parasitic migration with eosinophil infiltration was not confirmed. Furthermore, no bacteria were observed with the Giemsa or Ziehl–Neelsen stains, and no fungi or protozoa were observed with the periodic acid–Schiff reaction or the Grocott methenamine silver stain. No viral inclusion bodies were identified in neurons or glial cells. Immunohistochemistry (IHC) revealed that the major cells in the subarachnoid space were ionized calcium-binding adapter molecule 1 (Iba1)-positive macrophages (Fig. 1B) and CD3-positive T cells (Fig. 1C); the remaining cells were CD79α-positive or IgG-positive B cells or plasma cells and myeloperoxidase (MPO)-positive neutrophils. Glial nodules consisted of Iba1-positive and MPO-positive microglia (Fig. 1D; Suppl. Fig. 4A). CD3-positive T cells and Iba1-positive macrophages were also observed in perivascular cuffs. Leptospires were detected in perivascular cuffs (Fig. 1D, inset), but not in the glial nodules. Glial fibrillary acidic protein (GFAP)-positive astrocytes were scattered in the brain sections (Suppl. Fig. 4B), but not related to the pathologic findings.
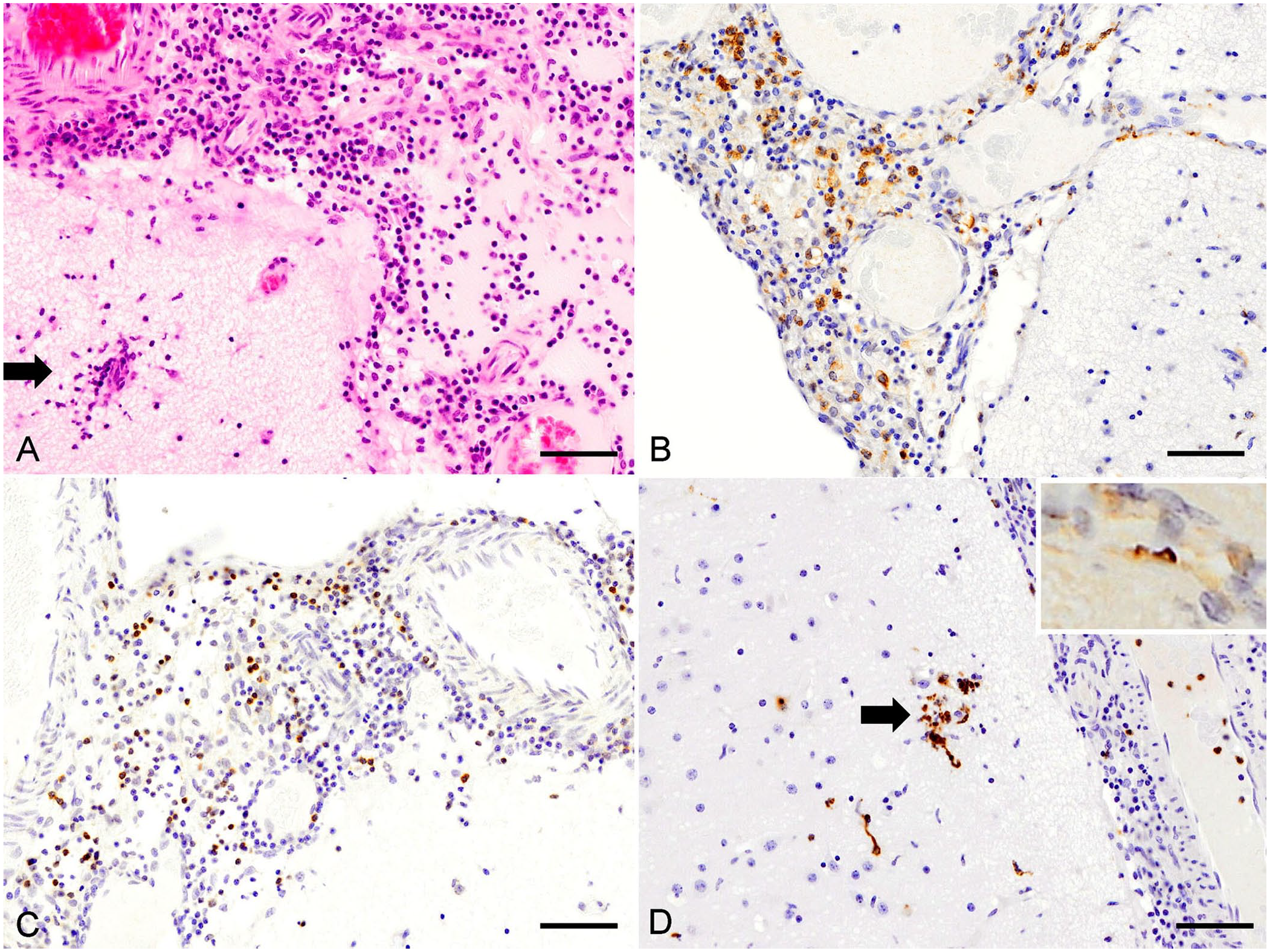
Nonsuppurative Leptospira-related meningoencephalitis in a raccoon dog.
Diffuse lymphoplasmacytic interstitial nephritis was observed in the cortex and outer medulla of both kidneys (Suppl. Fig. 5A). Interstitial infiltrates consisted mainly of IgG-positive and CD79α-positive B cells or plasma cells (Suppl. Fig. 5B), Iba1-positive macrophages (Suppl. Fig. 5C), and occasional MPO-positive neutrophils. Intratubular cell debris, hyaline casts, and tubule cell necrosis and hyperplasia with nuclear expression of proliferating cell nuclear antigen (PCNA) were often observed. Leptospires were detected in renal tubules with Warthin–Starry stain (Suppl. Fig. 5D) and IHC (Suppl. Fig. 5D, inset). Ureteritis was detected with intraepithelial infiltrates of CD3-positive T cells and Iba1-positive macrophages and a slight increase in the nuclear expression of PCNA in the ureteral epithelium.
In the liver, centrilobular congestion with a reduction of intercellular E-cadherin were observed (Suppl. Fig. 6A, 6B), as well as focal hepatocellular necrosis. However, hepatocellular dissociation was not evident. Many leptospires were detected in the hepatic cords with Warthin–Starry stain (Suppl. Fig. 6D, inset). Kupffer cell activation was detected by prominent expression of Iba1 in the sinusoidal spaces (Suppl. Fig. 6C); MPO-positive neutrophils were scattered (Suppl. Fig. 6D). In the periportal areas, hyperplasia of bile duct cells, with prominent intercellular expression of E-cadherin (Suppl. Fig. 6B) and mild nuclear expression of PCNA, were observed, occasionally with dispersed single epithelial cell necrosis and peribiliary mononuclear infiltration.
In the heart, pericarditis and periarteritis (Suppl. Fig. 7A) were observed with infiltrated CD3-positive T cells and Iba1-positive macrophages. In the lungs, edema, hemorrhage, and multiple foreign-body granulomas were observed, but not interstitial pneumonia (Suppl. Fig. 7B). A few leptospires were detected in intact alveolar walls with IHC (Suppl. Fig. 7B, inset). Moreover, bronchitis and tracheitis with neutrophilic infiltration in the tracheal glands was noted. Diffuse depression of paracortical CD3-positive T cells was observed only in the popliteal lymph nodes, along with sinusoidal fluid retention. The splenic white pulp was atrophic, and hemosiderin was deposited in the red pulp. In the stomach, foreign-body granulomas with central food debris were observed in the submucosal tissue and smooth muscle layer (Suppl. Fig. 7C). In the small intestine, intraluminal nematodes were associated with submucosal granulomas (Suppl. Fig. 7D), which contained CD3-positive T cells and Iba1-positive macrophages. Periarteritis with CD3-positive T cells and Iba1-positive macrophages was observed in the serosa of the stomach and small intestine. The osteoid deposition in ribs, with prominent proliferation of osteoclasts, was consistent with rickets.
We isolated DNA from paraffin sections of kidney and cerebrum (NucleoSpin DNA FFPE XS; Macherey-Nagel) to detect Leptospira spp. using PCR. 11 Specific sequences were not demonstrated.
A third wild raccoon dog without convulsions injured concurrently in a traffic accident in Tokyo, Japan, was examined using serum microscopic agglutination tests for L. interrogans serovars Icterohaemorrhagiae (strain RGA), Canicola (strain Hu-IV), Autumnalis (strain Akiyami-A), and Australis (strain Ballico), and Leptospira alexanderi ser. Hebdomadis (strain Hebdomadis);11,14 titers were 1,024; 64; 128; 2; and 8, respectively. We concluded that the third raccoon dog was infected with L. interrogans ser. Icterohaemorrhagiae.
Neuroleptospirosis is a rare neurologic manifestation of infection by pathogenic L. interrogans in humans. 13 Leptospira organisms are found in systemic organs a few days post-infection, and reach the cerebrospinal fluid (CSF) and brain. 13 Clinical manifestations are subdivided into early leptospiremic and later immune phases, with neurologic signs of acute influenza-like illness and meningitis, respectively.13,18 Acute influenza-like illness, such as fever and headache, was observed in 80–100% of patients; leptospiral meningitis was diagnosed only in a small subset of patients (~20%).1,20 Altered behavior and seizures, but not convulsions, were reported in patients with leptospiral meningitis. 18 As well as CSF pleocytosis, Leptospira spp. and/or antibodies in blood and/or CSF were detected to establish the diagnosis of meningitis. 20 We did not examine the CSF in our case.
The source of infection and the pathogenesis of meningitis in our case are unknown. Rodents inhabiting urban and rural areas are the main reservoirs of Leptospira infections; leptospires are transmitted to other animals and humans via urine, given that the natural location of pathogenic leptospires is the renal proximal convoluted tubules.4,8,10,12 In our case, the raccoon dog with malnutrition, immunodeficiency, and rickets might have been infected with organisms through the injured mucosa of the gastrointestinal tract. It has been reported that T cells and macrophages invaded the arachnoid space in a 14-y-old human patient with combined severe immunodeficiency and neuroleptospirosis. 19 Cellular immunity might have played a role in the pathogenesis of meningoencephalitis in our case, given that the major infiltrating cells were T cells and macrophages in the arachnoid space with perivascular predilection. We detected leptospires in several perivascular cuffs, but not in glial nodules. Glial nodules were restricted to the molecular layer of the cortex and occasionally were close to small vessels. Invading leptospires might be destroyed by activated or amoeboid microglia, as described in bacterial meningitis. 17 The role of MPO in microglial phagocytosis is not fully understood; however, activated microglia might release reactive oxygen species via MPO and kill organisms.15,17 A small granuloma with multinucleate giant cells in the fimbria of the hippocampus suggested a cellular reaction against migrating parasites. We cannot eliminate the possibility of visceral larval migrans; however, we did not detect migrating nematodes. Several studies of neuroleptospirosis in humans demonstrated perivascular lymphocytic cuffing and demyelination in the white matter of the cerebrum, cerebellum, and medulla oblongata.3,13 However, we did not observe lesions in the white matter in our case.
The spectrum of clinical signs might be associated with the severity of Leptospira-related pathologic changes. In humans, anicteric leptospirosis including meningitis is either subclinical or of mild severity; icteric leptospirosis, including acute renal failure, pulmonary hemorrhage, and cardiac involvement, is more severe or rapidly progressive. 10 This concept might also apply to dogs. 16 Typical Leptospira-related nonsuppurative interstitial nephritis was prominent in our case.3 –5 This was concordant with the findings in raccoons with leptospirosis. 7 Interendothelial cell migration of Leptospira can damage endothelial cells, release inflammatory cytokines, and localize in the renal interstitium, then enter tubules, with a final damaging predilection for proximal convoluted tubule epithelium.4,5 We also observed pulmonary edema and hemorrhage, as reported previously3,4,10,16; myocardial periarteritis was a Leptospira-related lesion; cardiac aortitis with marked infiltration of lymphocytes in the adventitia has been reported in human patients with leptospirosis.3,6,10 Leptospira can reduce the expression of E-cadherin, contributing to hepatocyte dissociation and individualization; 5 however, hepatocyte dissociation was not evident in our case, even with a reduction of interhepatocellular E-cadherin. Prognosis in neuroleptospirosis cases is generally favorable in patients when treated with antibiotics such as penicillin;13,18,20 however, the untreated raccoon dog in our case may have died as a result of a severe form of leptospirosis with renal, pulmonary, and cardiac involvement.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387211033583 – Supplemental material for Leptospiral meningoencephalitis in a raccoon dog
Supplemental material, sj-pdf-1-vdi-10.1177_10406387211033583 for Leptospiral meningoencephalitis in a raccoon dog by Risako Yamashita, Toshinori Yoshida, Mio Kobayashi, Suzuka Uomoto, Saori Shimizu, Keisuke Takesue, Natsuno Maeda, Erika Hara, Kanami Ohshima, Wen Zeng, Yasunori Takahashi, Aoi Ikeuchi, Emi Okamoto, Takutoshi Sugiyama, Hiroki Nagakubo, Madoka Ichikawa-Seki and Makoto Shibutani in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declared that they received no financial support for their research and/or authorship of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
