Abstract
A male Korean raccoon dog of unknown age was rescued and placed at the Daejeon Wildlife Rescue Center, Korea. Physical examination revealed severe emaciation and dehydration, as well as thick crusts and alopecia over most of the body. During medical care, the animal died and was submitted for postmortem examination. Firm, brown-red lesions of various sizes were observed on the surface of the lungs. In cross-sections of the lungs, pulmonary vessels were thickened and dilated, with white irregular papillary luminal projections. Histologically, pulmonary blood vessels were severely hyperplastic, characterized by thickened dilated walls and fibrous papillary projections covered with a single layer of endothelial cells (ECs). Hyperplastic fibrous connective tissue was confirmed by Masson trichrome staining. The ECs expressed CD31. We diagnosed the lesion as intravascular papillary endothelial hyperplasia, a unique non-neoplastic reactive process that has not been reported previously in pulmonary vessels of canids, equids, or felids, to our knowledge.
The raccoon dog (Nyctereutes procyonoides), a member of the Canidae family, is native to East Asia. There are 6 subspecies: albus, koreensis, orestes, procyonoides, ussuriensis, and viverrinus. 14 The Korean raccoon dog (N. p. koreensis) is one of the most plentiful mammals in Korea, and populations have increased, adapting to peri-urban and urban environments. 11 Korean raccoon dogs are an important reservoir and/or carrier of numerous zoonotic disease agents.14,19 However, little is known about the noninfectious diseases in this species.
A raccoon dog carcass from the Daejeon Wildlife Rescue Center in Korea was submitted to the Department of Veterinary Pathology, College of Veterinary Medicine, Chungnam National University (Daejeon, Republic of Korea) for postmortem examination. Macroscopically, firm, brown-red, 0.5–2-cm diameter lesions were observed on the visceral pleura (Fig. 1A). Throughout the pulmonary parenchyma, blood vessel walls were thickened, and the vessels were dilated, with irregular papillary projections filling most of the lumen (Fig. 1B, 1C).
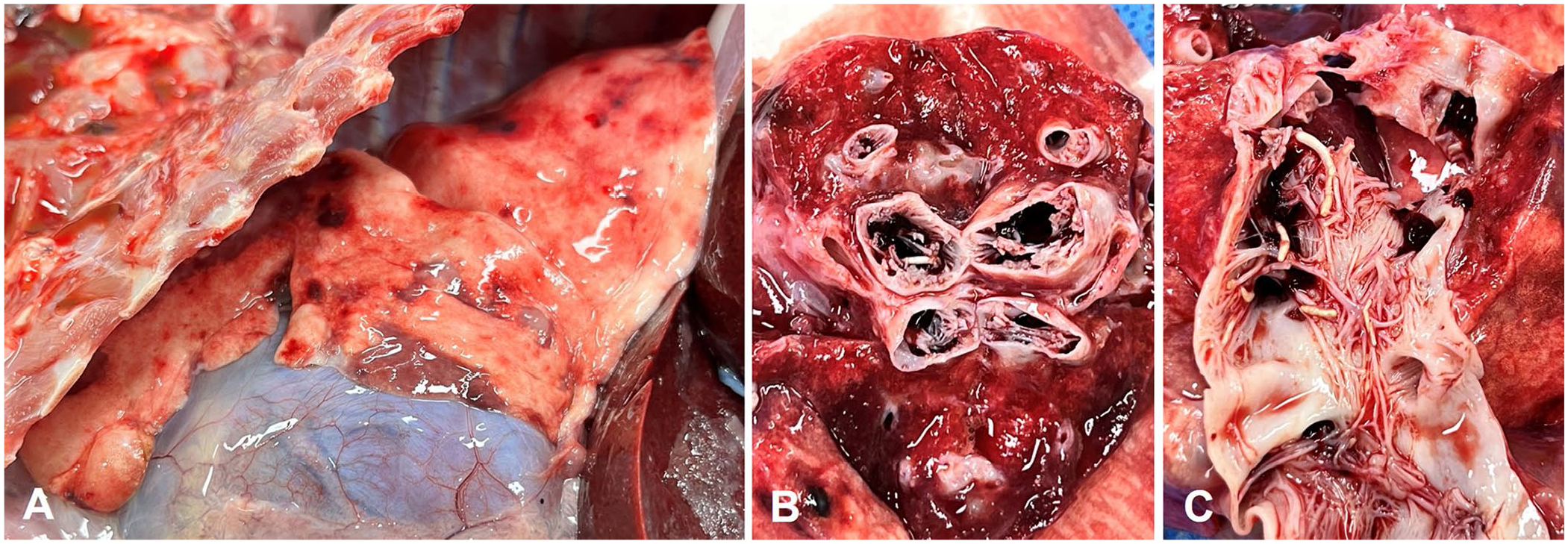
Gross findings in a case of intravascular papillary endothelial hyperplasia in the lungs of a wild Korean raccoon dog.
The animal had various cutaneous lesions, including thick crusts and alopecia, likely the result of ectoparasitism. Subcutaneous lymph nodes were enlarged (1 × 2–2 × 3 cm) and edematous. Small amounts of serosanguineous fluid were observed in the abdominal, pericardial, and thoracic cavities. The internal organs were pale. Small necrotic foci were apparent in the liver and kidneys. A focal hemorrhagic area was noted on the occipital region of the right cerebral hemisphere. Samples of skin, lymph node, lung, liver, kidney, cerebrum, and heart were fixed in 10% neutral-buffered formalin, processed routinely, and 4-µm sections were stained with H&E and Masson trichrome stains for histologic examination. Immunohistochemistry (IHC) was performed using the peroxidase-based avidin–biotin complex method (Vectastain ABC kit; Vector) with the following primary antibodies: CD31 and alpha–smooth muscle actin (α-SMA; Table 1).
Primary antibodies used for immunohistochemical staining in the diagnosis of intravascular papillary endothelial hyperplasia in a Korean raccoon dog.
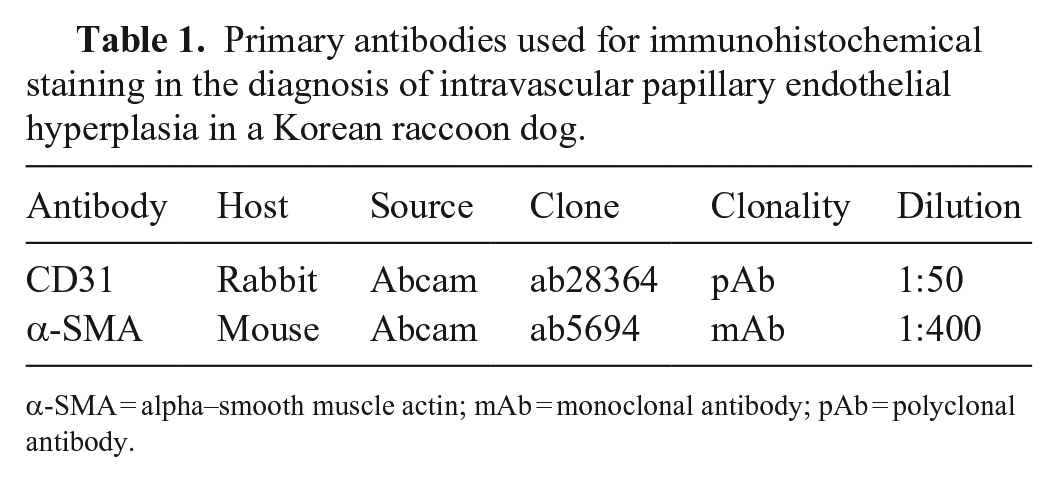
α-SMA = alpha–smooth muscle actin; mAb = monoclonal antibody; pAb = polyclonal antibody.
Histologically, most of the pulmonary blood vessel walls were thickened; luminal papillary structures were covered by a single layer of endothelium without atypia (Fig. 2A). Some papillae were attached to the vascular wall; others were free in the lumen. In some areas, clumped and fused papillae formed a loose mesh-like anastomosing network. The lesions were non-inflamed, and without mitoses, nuclear and cellular pleomorphism, or invasion of adjacent tissues. The cores of papillae consisted of well-differentiated fibrous connective tissue, which also effaced the architecture of vessel walls (Fig. 2A, 2B). CD31 (a vascular marker) was expressed strongly and specifically in endothelial cells covering proliferative collagenous tissues (Fig. 2C). The proliferative fibrous tissues were negative for α-SMA; hence, pulmonary medial hypertrophy and hyperplasia, which occur commonly in cats 16 and rats, 15 were ruled out. We diagnosed this pulmonary lesion as intravascular papillary endothelial hyperplasia (IPEH), 1 which is a novel case in pulmonary vessels from animals. We retrieved no cases of pulmonary IPEH in a search of Google, PubMed, CAB Direct, Web of Science, and Scopus, using the search term “intravascular papillary endothelial hyperplasia,” suggesting that this condition has not been reported in lungs of non-human species.
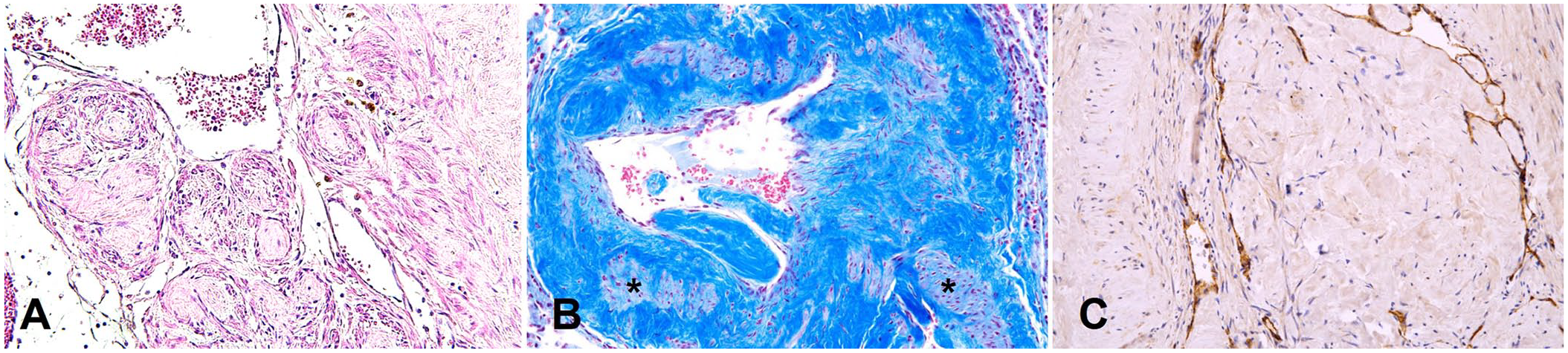
Histologic findings in a case of intravascular papillary endothelial hyperplasia in the lungs of a wild Korean raccoon dog.
Bronchi and bronchioles had normal architecture. Alveolar walls appeared thickened and/or collapsed, and some alveoli were filled with eosinophilic material. The generalized cutaneous scale crust, comprised of serous exudate, neutrophils, eosinophils, and lymphocytes, with multifocal ulceration or erosion secondary to excoriation, was the result of a heavy Sarcoptes sp. infection. Incidental focal necrotic lesions with inflammatory cell infiltration were observed in the liver, kidney, brain, heart, and lymph nodes.
IPEH, also known as Masson tumor, which can mimic angiosarcoma, 2 is a rare non-neoplastic reactive lesion that has been reported primarily in humans,1–3,10 with only 2 cases reported in the lungs. 10 In animals, a single case of IPEH was reported from the conjunctiva of a horse, 9 and 10 cases in dogs with vascular neoplastic lesion in the skin. 6 Two cases of extravascular PEH have been reported in cats; one from an intramuscular mass with inguinal lymphadenopathy, and one from subcutaneous masses on the thorax. 7 Three clinical types of IPEH have been described. 8 A primary or “pure” form arises within a normal blood vessel. The secondary lesion or “mixed” form arises in a preexisting vascular malformation. The rarest type, an extravascular hemangioma, usually arises from an organizing hematoma. Our case is in the first category given that it originated in pulmonary blood vessels in the absence of a secondary lesion.
In humans, IPEH usually occurs as well-defined, oval, small solitary nodules, and frequently develops in blood vessels in the extremities. IPEH lesions can also be found in the oral and lingual mucosae, heart, lung, conjunctiva, and small intestine. 4 IPEH arises more commonly within a dilated vein than in an artery; however, it can also arise in varices, hemorrhoids, existing vascular tumors, and in hematomas. 18 IPEH consists of an intravascular proliferation of numerous papillae with a core of connective tissue and an endothelial surface within distended veins. 13 Our case had similar histologic features in both pulmonary arteries and veins.
The pathogenesis of IPEH remains poorly understood. IPEH is closely associated with thrombus formation; however, the basis for the development of the lesion in the thrombus has not been fully determined. Some researchers have proposed that the developing thrombus serves as a matrix for the ingrowth of papillary structures. 17 Similar to thrombus organization and recanalization, the development of IPEH lesions occurs through several steps. 5 In cases of vascular injury resulting in thrombus formation, typically within veins, endothelial cell proliferation can be mediated by inflammation and vascular stasis. 12 Embedding of endot-helial cells within the thrombus characterizes early-stage lesions. 1 Subsequently, proliferating endothelial cells separate the collagenase-digested thrombus into irregular clumps, which then develop into papillary structures. 1 Finally, these papillae combine to form anastomosing intravascular structures. 1 Heartworm infection can cause pulmonary vascular wall proliferation with subintimal myofibrosis and villous projections into the pulmonary arterial lumen with extensive vasculitis and periarteritis. 16 However, we did not observe thrombus formation, vasculitis in pulmonary vessels, and/or heartworm infection in our case. Further studies are warranted to elucidate the pathogenesis of IPEH.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our study was supported by Chungnam National University and the National Institute of Wildlife Disease Control and Prevention (NIWDC), funded by the Ministry of Environment (MOE) of the Republic of Korea (NIWDC-2021-SP-2022-010).
