Abstract
Theileria parasites commonly infect African wild artiodactyls. In rare roan (Hippotragus equinus) and sable (H. niger) antelopes, Theileria sp. (sable)-associated calf mortalities constrain breeding programs. The pathogenicity of most leukocyte-transforming Theileria spp. originates in their invasion of and multiplication in various mononuclear leukocytes, the transformation of both infected and uninfected leukocytes, and their infiltration of multiple organs. Understanding the pathogenesis of theileriosis can be improved by the use of immunohistochemistry (IHC) to identify the localization of the parasites in tissue sections. Our aim was to develop a reproducible IHC assay to detect leukocyte-associated Theileria parasites in formalin-fixed, paraffin-embedded roan and sable tissues. Polyclonal antibodies were purified from the sera of 5 roans from an area endemic for Theileria sp. (sable) and tested for IHC reactivity in 55 infected and 39 control roan and sable antelopes, and for antigen and species cross-reactivity in an additional 58 cases. The 3 strongest antibodies consistently detected intraleukocytic theilerial antigens in known positive cases in roan and sable antelopes, and also detected other Theileria spp. in non-hippotraginid wild artiodactyl tissues. The antibodies did not cross-react with other apicomplexan protozoa, with the exception of Cryptosporidium. Given that PCR on its own cannot determine the significance of theilerial infection in wild ruminants, IHC is a useful laboratory test with which to confirm the diagnosis in these species.
Schizont-transforming theilerial species and genotypes, in the order Piroplasmida (phylum Apicomplexa), can cause life-threatening theileriosis in diverse domestic and wild ungulate species.7,27 Well-documented theilerial diseases include East Coast fever (ECF, caused by Theileria parva) 25 and tropical theileriosis (TT, caused by T. annulata) 26 in cattle, and malignant ovine theileriosis (MOT, caused by T. lestoquardi) in sheep. 10 These diseases cause high morbidity and mortality rates with associated significant yearly economic losses in tropical and subtropical regions.10,15,30 Isolated cases of acute fatal theileriosis are reported in a variety of African wild ungulate species. 7 Roan (Hippotragus equinus) and sable (H. niger) antelopes, which are rare and endangered species,21,22 are particularly susceptible to lethal infection.32,43 Given that current data are sparse and based on very few cases, 7 tests for theileriosis in these vulnerable species will enhance our understanding of the disease.
Theileriosis is characterized by schizont-induced leukocyte transformation, seen as marked atypia and proliferation of infected and uninfected mononuclear leukocytes, with multi-organ infiltration, followed by massive leukocytolysis and release of merozoites that infect erythrocytes. 7 If untreated, theileriosis causes overwhelming systemic inflammation, multiple organ dysfunction, and death.2,14,16 Erythrocytic infection with associated hemolytic anemia contributes to the pathogenesis through poorly understood mechanisms.24,25,50
Based on recent 18S rRNA phylogenies, most of the known transforming Theileria spp. occur in the T. taurotragi clade. 27 However, a few species, for example Theileria sp. (sable), which can be pathogenic in juvenile roan and sable antelopes,32,43,44 are scattered in other clades.27,34
Molecular techniques, such as reverse line blot (RLB) hybridization, conventional PCR, real-time PCR, and, most recently, DNA suspension microarrays, are used to identify Theileria species and whether animals are coinfected with different Theileria spp. and/or Babesia, Ehrlichia, and Anaplasma spp.7,27 However, molecular techniques have limitations.7,34 Most importantly, positive results are neither indicative of disease, nor can they determine the significance of coinfections. In addition, these techniques cannot identify novel species or differentiate among closely related species or variants that are cross-reactive. 7
At postmortem examination, domestic and wild ruminants that die as a result of acute theileriosis exhibit all or some of the following gross pathology lesions: anemia; icterus; parenchymal, serosal, and mucosal hemorrhages; body cavity effusions; pulmonary edema; and multiple cream-colored proliferative foci in diverse lymphoid and non-lymphoid organs.7,25,26 The observation of characteristic intraleukocytic schizonts in tissues from carcasses with the gross lesions as described is usually sufficient to diagnose acute lethal theileriosis. However, parasite identification in cytologic and histologic specimens is heavily reliant on observer skill and experience.9,20 For instance, it can be challenging to differentiate intracytoplasmic parasitic schizonts from azurophilic granules in large granular lymphocytes, or phagocytized debris in monocyte-macrophages in cytologic specimens. A significant advantage conferred by histopathology (compared to cytology) is that the location, distribution, and degree of infiltration of schizont-infected atypical leukocytes and associated lesions (e.g., parenchymal necrosis and hemorrhage) can be determined in target organs.7,13
Immunohistochemistry (IHC)23,38,48 can be used to identify specific antigens in affected tissues by adding enzyme- or fluorescent dye–linked antibodies that will bind to the sought-after antigens and elicit a visual signal at the site of binding. Immunohistochemical assays are rapid, simple, and affordable and can be performed on formalin-fixed tissues, which makes them well suited for use in resource-limited settings, where fresh material is not available or prompt processing not feasible. The major advantage of IHC is the co-localization of epitopes specific to certain cells and tissues or infectious agents with histologic lesions. In the case of infectious agents, such co-localization can imply causality between the labeled pathogen protein(s) and the associated lesions, particularly if lesions are highly characteristic for the pathogen in question, as in acute lethal theileriosis. Immunochemical techniques to detect parasitic schizonts in cytologic smear samples and formalin-fixed, paraffin-embedded (FFPE) tissues have been reported in a few (generally dated) studies on theileriosis in livestock.5,10,11,13,14,20,29 No such data are reported in African wildlife.
Despite the pivotal role played by IHC in diagnostic and experimental investigative studies in human and veterinary pathology, protocols for the validation of IHC assays are extremely variable1,48 and particularly scarce in wildlife. 3 To date, there are no minimum validation standards for IHC assays, but there is a wealth of guidelines for IHC test validation in both human and veterinary pathology.1,3,12,23,37,38,48 Validation of IHC assays is multifaceted, and there is currently a strong drive to improve the quality (specificity) of IHC assays in pathology.1,48 However, by striving to address all contingencies, modern test validation requirements can be impractical. In fact, even in sophisticated laboratories that use laboratory animal models and established cell lines, there is a scarcity of properly designed IHC studies that have characterized the performance of even commercial monoclonal antibodies (mAbs), especially under physiologic conditions in which proteins are usually not expressed at levels nearly as high as in engineered cell lines. 1 Properly validated IHC tests are even less common in non-laboratory animals, let alone wildlife. Importantly, it cannot be assumed that test protocols that have been validated in domestic species can be applied equally in related wildlife species. 3
At the very least, the diagnostic sensitivity (DSe) and specificity (DSp) of IHC should be estimated by scoring assay performance in target tissues from known infected and uninfected host species sourced from clearly defined populations. Pertaining to laboratory tests for infectious diseases, DSe refers to the probability that an infected animal will test positive; DSp refers to the probability that an uninfected animal will test negative. The use of a variety of assays, including western blots, is advocated to corroborate antibody specificity for target proteins in specified host species.18,48 In addition, cross-reactivity with the target organism in different host species (referred to as species cross-reactivity) and with closely related organisms and unrelated organisms that produce similar lesions (referred to as antigen cross-reactivity) should be assessed.37,38,48
Our main objective was to validate an immunoperoxidase assay, using polyclonal anti–Theileria sp. (sable) IgG antibodies purified from roan antelope sera for the detection of theilerial schizonts in roan and sable FFPE tissues. We investigated the species cross-reactivity of the purified antibodies with transforming theilerial parasite antigens in non-hippotraginid wild artiodactyl tissues. We also assessed antigen cross-reactivity with non-theilerial apicomplexan protozoa, unrelated rickettsial bacteria, and leukocytes in lesions that resemble theileriosis histologically.
Materials and methods
We characterized and purified antibodies from 5 different roan sera; tested them on archived FFPE tissues from hippotraginids and non-hippotraginids with fatal theileriosis, and tissues from diverse host species with related coccidian parasites, rickettsial bacteria, or histologically similar lesions; and calculated and compared the DSe and DSp and host and antigen cross-reactivity of the IHC assay (using the 5 different antibodies) to establish the repeatability of the method.
Antibody characterization and purification
Serum and EDTA blood samples and blood smears were obtained from the cephalic vein of 5 healthy free-ranging roan antelopes (3 yearlings, 2 calves 1–3 mo old) that were chemically immobilized for routine management purposes on a private game ranch in the Northern Cape province of South Africa. The blood smears were stained (Diff-Quik; Kyron Laboratories) within a few hours of collection. South African Veterinary Council–registered veterinarians performed the immobilization and then collected and transported the blood smears and blood samples according to prescribed methods (Department of Agriculture, Land Reform and Rural Development Permit Reference 12/11/1/1/8). The University of Pretoria Animal Ethics Committee approved the study (protocol V028-18).
DNA was extracted from EDTA blood samples (PureLink genomic DNA mini kit; Invitrogen) according to the manufacturer’s instructions. DNA was eluted in 100 µL of elution buffer. The RLB hybridization technique was used to screen the DNA samples for the simultaneous detection of Theileria, Babesia, Anaplasma, and Ehrlichia species.31,32 For the parasite 16S rRNA, primers Ehr-F (5′-GGAATTCAGAGTTGGATCMTGGYTCAG-3′) 42 and Ehr-R (5′-Biotin-CGGGATCCCGAGTTTGCCGGGACTTYTTCT-3′) 4 were used to amplify the V1 hypervariable region, and for the 18S rRNA forward primer, RLB F2 (5′-GACACAGGGAGGTAGTGACAAG-3′) and the reverse-biotin-labeled primer RLB R2 (5′-Biotin-CTAAGAATTTCACCTCTGACAGT-3′) 31 were used to amplify the V4 hypervariable region. Anaplasma centrale and Babesia bigemina DNA, extracted from the A. centrale and B. bigemina vaccines (Onderstepoort Biological Products) were used as positive controls, and water was used as the negative control. For DNA amplification, the touchdown PCR thermocycler program was used. 32 Various Ehrlichia, Anaplasma, Theileria, and Babesia genus- and species-specific oligonucleotide probes were used including Ehrlichia/Anaplasma catch-all, 4 Anaplasma bovis, 4 Anaplasma platys (A.M. Nijhof, Freie Universität Berlin, unpublished), Anaplasma sp. ‘Omatjenne’, 4 Theileria/Babesia catch-all, 17 Theileria catch-all (Nijhof, unpublished), T. bicornis, 31 Theileria sp. (sable), 32 and T. separata. 40
For each serum sample, the presence of antibodies against Theileria sp. (sable) proteins (from roan lymph node–derived subcultures) 51 was tested on western blots and compared to nonspecific cross-reactions with cultured T. parva–infected leukocytes as well as uninfected bovine lymphocytes. Cells were washed with phosphate-buffered saline (PBS) and resuspended in RIPA lysis buffer (150 mM NaCl, 5 mM EDTA, 50 mM Tris, 1% NP-40, 0.5% sodium deoxycholate, 0.1% sodium dodecyl sulfate [SDS], pH 8) and 25 units of Benzonase nuclease (Novagen; MilliporeSigma) per mL of RIPA buffer. The mixture was incubated on ice for 20 min or until complete digestion of nucleic acids and the solution was non-viscous. An equal volume of protein sample buffer (1 M Tris [pH 6.8], 2% SDS, 10% glycerol, 5% 2-mercaptoethanol, 0.01% bromophenol blue) was added, vortexed, boiled for 5 min, and vortexed again. Total cell lysate equivalent to 2 × 104 cells per lane was loaded on 4–15% precast gels (Mini-protean TGX; Bio-Rad) and separated in the presence of TGS buffer (Tris-glycine-SDS; Bio-Rad). The separated proteins were transferred to PVDF membranes (Novex; Thermo Fisher) using Towbin buffer (25 mM Tris, 192 mM glycine). After transfer, the membranes were blocked in 2% MP-PBS (milk protein–PBS) at room temperature (RT) for 1 h. The roan serum was diluted 1:100 in MP-PBST (0.05% Tween) and incubated for ≥1 h at RT. After 3 washes with PBST, protein A/G-HRP (Thermo Scientific) diluted 1:10,000 in MP-PBST was added and incubated for 1 h at RT. After the final 3 washes, TMB (3,3′,5,5′-tetramethylbenzidine) substrate for blots (Life Technologies) was added and left for 10 min.
For the purpose of IHC labeling, IgG was purified from the 5 roan sera (Protein G HP SpinTrap; GE Healthcare) according to the manufacturer’s instructions. The purified IgG was quantified by spectrophotometry. For each sample, IgG was purified from 800 µL of serum in a final volume of 6.5 mL, to enable using the same effective titer for IHC.
For each of the 5 roans from which antibodies were purified (roan 1 = antibody A; roan 2 = antibody B; roan 3 = antibody C; roan 4 = antibody D; roan 5 = antibody E), the Theileria piroplasm parasitemia (%) was estimated in blood smears by counting the number of Theileria piroplasm–infected erythrocytes in 1,050 erythrocytes. Theilerial piroplasms were identified based on their small size and pleomorphic appearance7,8; they frequently occurred singly within erythrocytes and had neutral-staining cytoplasm.
Chromogenic IHC technique
A manual indirect immunoperoxidase technique was performed on 4-μm thick recent histologic slices of FFPE specimens. Although formalin fixation time was not known for the majority of cases, all cases were submitted for diagnostic purposes, and many of the animals were valuable, therefore tissues were unlikely to have been fixed for >1–2 mo. Method optimization was performed according to prescribed protocols and included application of different antigen retrieval (AR) methods (including no AR), and different detection methods including a variety of polymer-based systems as well as the avidin–biotin complex detection system (Vector Laboratories). The following method is the one that worked best on selected positive and negative tissue controls in our IHC laboratory.
Briefly, after routine deparaffinization, rehydration, and incubation with 3% hydrogen peroxide in methanol for 15 min at RT to block endogenous tissue peroxidases, tissue sections were pretreated by microwave heating (96°C) in citrate buffer (pH 6) for 14 min. A 2% solution of skimmed milk powder in distilled water was applied to the sections for 15 min at RT to block hydrophobic background staining.6,36 Five roan polyclonal IgG antibodies (A–E) were applied separately to the tissue sections at a dilution of 1:25 for 2 h at RT. Tissues were subsequently incubated with a universal antibody detection reagent (Pierce peroxidase-conjugated recombinant protein A/G; Thermo Scientific) for 30 min at RT, followed by a polymer detection system (Mouse and rabbit specific HRP/DAB IHC micro-polymer detection kit; Abcam) according to the manufacturer’s instructions. To visualize the reaction product, sections were subjected to chromogen treatment with NovaRED (Vector Laboratories) for 1–2 min, followed by counterstaining in Mayer hematoxylin for 10 s, rinsing, and routine dehydration through increasing alcohol concentrations and xylene. Slides were mounted with Entellan (MilliporeSigma) and coverslipped. To ensure assay performance, positive and negative tissue controls were included in every IHC run.
Two different positive tissue controls were used per lot. First, we used cytospins made from 5 × 104 Theileria sp. (sable)-infected roan leukocytes harvested from cell culture. 51 The same cell culture was the source of the Theileria sp. (sable) proteins used in the western blots so that we could compare Theileria sp. (sable) antibody reactivity in 2 different assays using the same theilerial proteins. Cytospins were fixed in 4% neutral-buffered formalin for 5 h. This was followed by incubation with 3% hydrogen peroxide in methanol for 5 min at RT. The rest of the assay technique was the same as for the FFPE tissue sections.
In addition to the cytospin positive controls, we used FFPE tissues with clear histologic evidence of intraleukocytic parasitic schizonts from a freshly dead Theileria sp. (sable)-positive roan and a sable antelope, based on RLB results, for positive tissue control purposes. Negative tissue controls included lymphoid and non-lymphoid FFPE tissues (similar to those affected by transforming Theileria parasites) 7 from 1 roan and 1 sable antelope that died as a result of cerebellar cortical degeneration (compounded by aspiration pneumonia and exertional myopathy) and hemonchosis, respectively. In neither case were gross or histologic lesions remotely suggestive of theileriosis, and no hemoparasites or rickettsial bacteria were identified in blood smears. During IHC optimization, to check for nonspecific binding of the universal antibody, the primary polyclonal roan IgG antibodies were omitted altogether during the labeling of positive and negative tissue controls.
Antibody validation
Case selection
Five purified Theileria sp. (sable) polyclonal antibodies (pAbs) were tested on 55 Theileria-positive roan (n = 31) and sable (n = 24) cases derived from private game ranches across South Africa from 1996–2019 (50 cases were from 2011–2019) that were selected from the archives at the Section of Pathology, Department of Paraclinical Sciences, Faculty of Veterinary Science, University of Pretoria. In all cases, the cause of death was acute theileriosis based on the observation of characteristic gross lesions, blood smear findings, and typical histologic lesions. 7 The level of autolysis was no more than mild in all selected cases, with no loss of tissue architecture and, in all cases, the erythrocytes were still intact and visible. RLB analyses on 10 positive cases (6 roans and 4 sables) confirmed infection with at least Theileria sp. (sable); 2 roans and 2 sables were coinfected with T. separata, and 1 of these 2 roans also with T. buffeli.
Similarly, 39 mildly autolyzed negative control cases (19 roan; 20 sable) were selected from the same archives from 2013–2019. These animals had died for reasons other than acute theileriosis, based on the case history, gross pathology, histopathology in a variety of organs, and, where pertinent, bacterial culture results (Suppl. Table 1). Parasitic schizonts were not identified in routine blood smears from any of these cases, but no PCR or RLB results were available.
Target tissues
For each positive test case, liver and 1 or 2 of the most severely affected of lung, kidney, adrenal gland, spleen, lymph node, and/or heart were selected for IHC. Organ selection concurred with previously reported target organs in African wild artiodactyls with acute lethal theileriosis. 7 The same organs were selected for the negative test cases.
Host species and antigen cross-reactivity
FFPE tissues were selected from 58 archived cases from 1992–2020 (49 cases were from 2011–2020), to test the cross-reactivity of the 5 Theileria sp. (sable) antibodies with the same and different Theileria spp. in distantly related host species and with related and morphologically similar microorganisms capable of infecting ruminants, as well as those causing similar lesions in the tissues of interest. Cases were excluded if tissues were more than mildly autolyzed. In addition, apart from the Theileria-positive non-hippotraginid wild bovid cases that were selected, cases were further excluded if there was any evidence of theilerial infection in blood smears, tissue sections, and/or if there were Theileria-positive RLB results.
Ten of the 58 cases included target tissues from non-hippotraginid wild artiodactyls that died of theileriosis with characteristic gross and histologic lesions, including visible intraleukocytic schizonts in diverse tissues; RLB results were available for 6 of these cases (Suppl. Table 2). The 10 cases included 1 American bison (Bison bison), 2 eland (Tragelaphus oryx), 1 gray/common duiker (Sylvicapra grimmia), 1 lechwe (Kobus leche), 1 tsessebe (Damaliscus lunatus), and 4 waterbuck (Kobus ellipsiprymnus). The bison, lechwe, and 2 of the waterbuck tested positive for Theileria sp. (sable) and T. separata, and both eland were infected with T. taurotragi. In addition, one of the eland also had T. bicornis infection.
Thirty-three of the 58 cases incorporated tissues from diverse mammalian species infected with non-theilerial apicomplexan coccidian parasites that were identified on histopathology, IHC, and/or transmission electron microscopy (TEM). The cases included 5 Besnoitia besnoiti cases in cattle; 5 cases with intestinal eimeriid coccidian parasites (1 African buffalo, Syncerus caffer; 2 impala, Aepyceros melampus; 1 sable; 1 springbuck, Antidorcas marsupialis); 5 Cryptosporidium cases (2 cattle; 1 sheep; 1 nyala, Tragelaphus angasii; 1 impala); 6 Neospora caninum cases in domestic dogs; 5 Sarcocystis cases (1 buffalo; 1 greater kudu, Tragelaphus strepciseros; 2 impala; 1 springbuck); and 7 Toxoplasma gondii cases (4 ring-tailed lemurs, Lemur catta; 3 domestic cats).
We also tested antibody cross-reactivity with activated and transformed mononuclear leukocytes in ruminants with malignant catarrhal fever (MCF, caused by wildebeest-associated alcelaphine herpesvirus 1, AlHV1 [Alcelaphine gammaherpesvirus 1] or ovine herpesvirus 2, OvHV2 [Ovine gammaherpesvirus 2]) and enzootic bovine leukosis (EBL), respectively, in which histologic lesions in diverse lymphoid and non-lymphoid tissues resemble those of acute theileriosis. To this end, 5 MCF cases (3 AlHV1-positive African buffalo, 1 AlHV1-positive cow, 1 OvHV2-positive springbuck) and 5 EBL cases in cattle with positive histopathology and serology results were selected for IHC.
We further tested antibody cross-reactivity with intracytoplasmic Ehrlichia ruminantium bacterial colonies in tissues from 5 springbuck that died of heartwater, given that the roan blood samples from which the Theileria sp. (sable) antibodies A–D were purified and then hybridized with the Anaplasma/Ehrlichia catch-all probe (Suppl. Table 3). In all of the springbuck cases, the Ehrlichia colonies were identified readily in vascular endothelial cells in hematoxylin and eosin (H&E)-stained brain sections, and heartwater infection was confirmed in Diff-Quik–stained brain smears and with IHC.
Scoring histopathology and IHC
An independent person randomized the Theileria-positive and -negative cases as well as all of the cases selected to check antibody cross-reactivity prior to their evaluation by the principal investigator (PI). In addition, the PI did not know which antibody (A–E) was derived from which roan until data collection was completed. Each case was comprised of 1 H&E- and 1 IHC-stained tissue section. Following randomization, each case was scored positive (score 1) or negative (score 0) based on the respective presence or absence of lesions characteristic of theileriosis in the H&E-stained slides. This was followed by scoring the presence (score 1) or absence (score 0) of IHC-labeled intraleukocytic theilerial parasites (as well as cross-reactive microorganisms) in consecutive tissue sections. Cases were scored IHC-positive if clearly labeled intraleukocytic parasites were detected in association with typical lesions in at least one of the tissues per case or if there was obvious labeling of microorganisms that were not necessarily associated with the expected lesions (i.e., cross-reactivity). Staining intensity was not scored because of the inherent variability associated with manual staining. However, it was noted if intraleukocytic parasites remained pale after repeated staining.
Statistical analysis
We calculated the DSe and DSp for all 5 antibodies (A–E). The DSe was calculated as the proportion of confirmed positive cases that stained positive with IHC. The 95% confidence intervals (CIs) were calculated using the R binom.test. 35 The DSp was calculated by dividing the observed negative cases by the total negative cases.
Antibody cross-reactivity was established for all of the antibodies. Cross-reactivity was positively identified when there was strong crisp or granular positive labeling of selected microorganisms and/or leukocyte paranuclear cytoplasm in FFPE tissue sections of an intensity clearly above nonspecific background staining.
We also investigated whether the histologic diagnosis concurred with the IHC diagnosis, although these tests should not be considered independent when used to diagnose diseases with characteristic histologic lesions. To do this, we counted the number of times both, either, or neither test suggested theilerial infection in the 94 positive and negative roan and sable cases, and then applied a χ2 test to the 2 × 2 contingency table to test for independence. We also compared labeling results for the 5 antibodies by counting the number of times they gave similar results in the 94 cases.
Results
The 3 roan yearlings (roans 1, 2, 4), from which antibodies A, B, and D were purified, respectively, tested positive for Theileria sp. (sable) using RLB (Suppl. Table 3), and their sera reacted strongly with Theileria sp. (sable) proteins on western blots (Suppl. Fig. 1). Each of these sera reacted with at least 15 Theileria sp. (sable) proteins with different patterns but have the proteins of 34, 45, 54, a 70–75 doublet, and 90 kDa in common. Piroplasms were observed, albeit at low frequencies, in blood smears from all 3 roans (Suppl. Table 3). Even though the PCR products from the 2 roan calves (roans 3, 5) did not hybridize with Theileria sp. (sable) (Suppl. Table 3), their sera (C, E) reacted weakly with Theileria sp. (sable) proteins on western blots (Suppl. Fig. 1). Sera C and E reacted with the same proteins, and the prominent proteins recognized were 45 and 70–75 kDa. No Theileria piroplasms were observed in blood smears from these 2 roans (Suppl. Table 3). Pertaining to all of the antibodies, low-level cross-reactions with a few T. parva and bovine lymphocyte proteins were observed in western blots.
The pattern of immunochemical labeling of intraleukocytic theilerial parasites in formalin-preserved roan and sable tissues mirrored the parasite morphology in H&E- and Wright-Giemsa–stained tissue sections and cytospin preparations (Fig. 1A–C; Suppl. Fig. 2A–C). Positively labeled schizonts comprised clusters of finely stippled red-brown granules, usually in a paranuclear location in the cytoplasm of mono- or multinucleate target leukocytes (Fig. 1B, 1C; Suppl. Fig. 2A, 2C). In contrast, the Theileria-negative roan and sable tissues showed no specific labeling (Fig. 1D; Suppl. Fig. 2A, 2C, bottom insets).
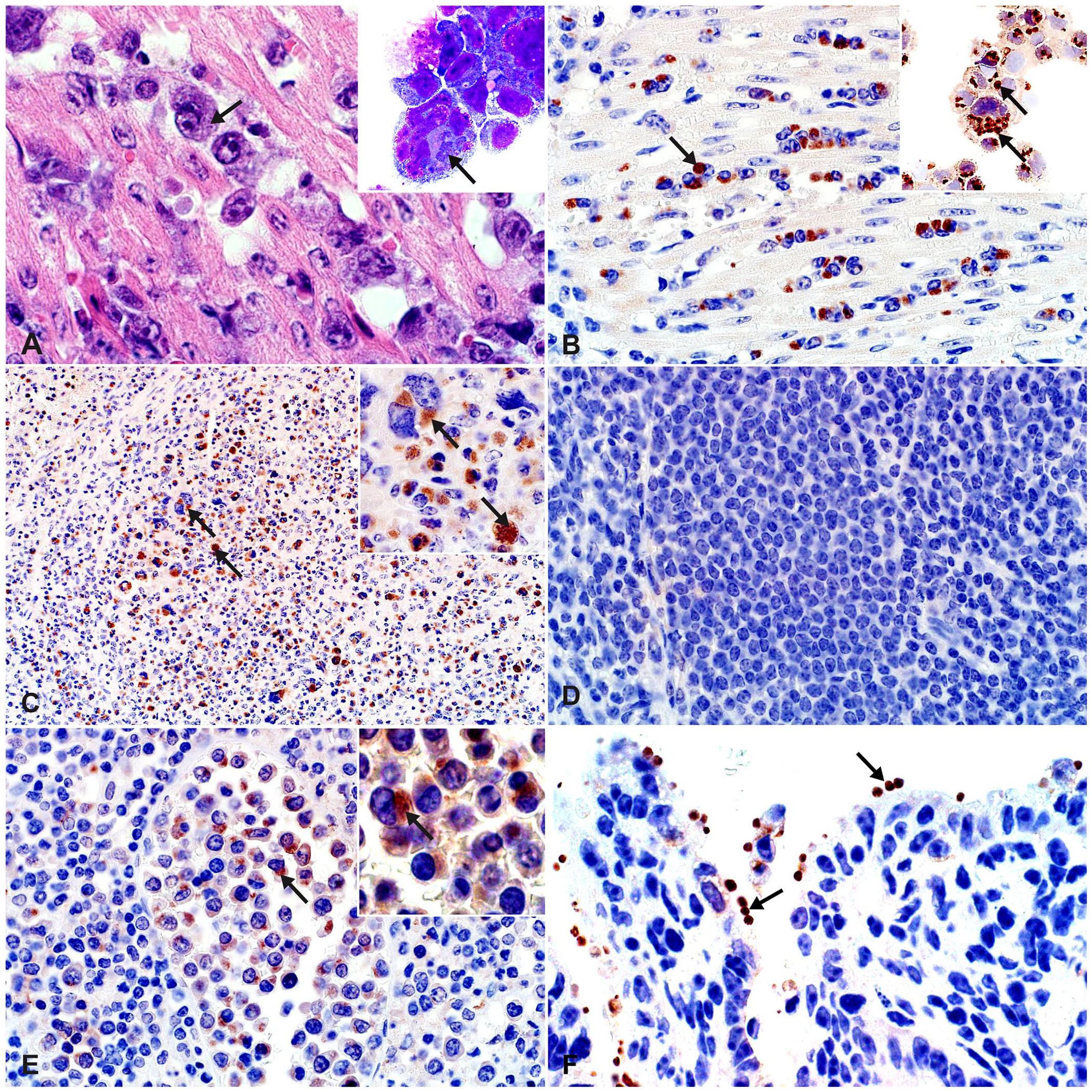
Efficacy of immunohistochemistry (IHC) and immunocytochemistry (ICC) in the detection of theilerial schizonts in African antelopes.
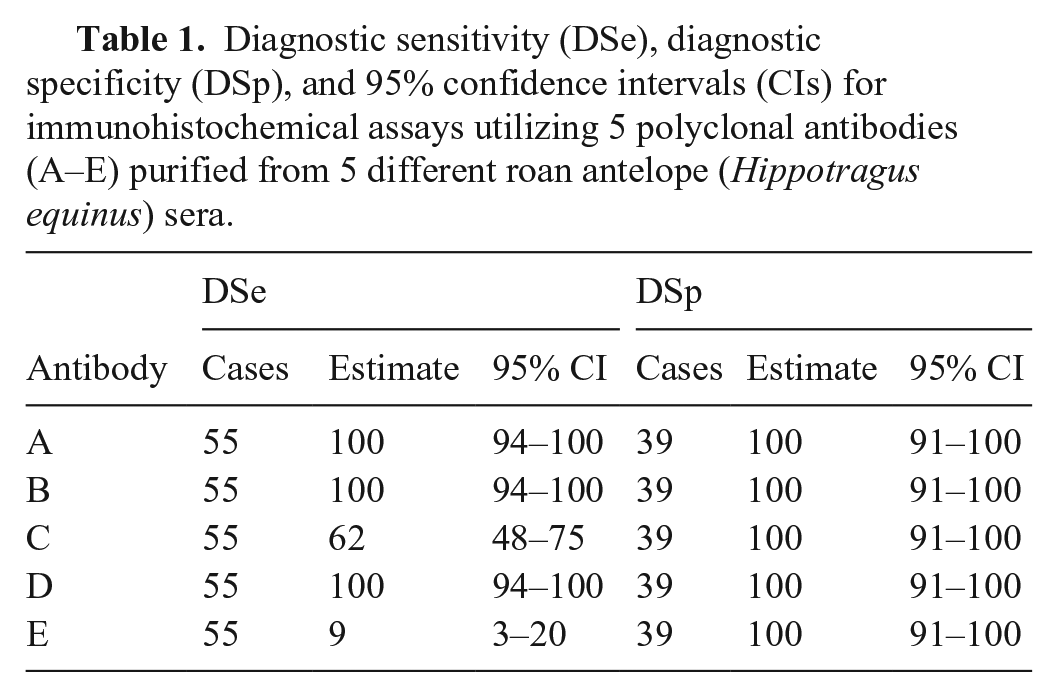
In our laboratory, the IHC assays utilizing antibodies A, B, and D had a DSe of 100% (roan and sable n = 55 [95% CI: 94–100%]; Table 1; roan n = 31, DSe = 100% [95% CI: 89–100%]; sable n = 24, DSe = 100% [95% CI: 86–100%]), and parasitic schizonts were usually clearly identifiable in all tissues with typical lesions (Fig. 1B, 1C; Suppl. Fig. 2A, 2C). In contrast, the IHC assays that used antibodies C and E detected theilerial schizonts in roan and sable tissues at much lower rates (Table 1), and the labeling intensity was generally weaker and more variable across tissues than was seen with the other antibodies (Suppl. Fig. 2B, 2D). The DSp of all antibodies was 100% (roan and sable n = 39 [95% CI: 91–100%]; Table 1; roan n = 19, DSp = 100% [95% CI: 82–100%]; sable n = 20, DSp = 100% [95% CI: 83–100%]). For the assays using antibodies A–D, there was occasional mild-to-moderate nonspecific (background) labeling of tissue parenchyma (e.g., hepatic, bronchial, renal tubular and adrenocortical epithelium, and cardiomyocytes; Suppl. Fig. 2E). Pulmonary edema and intra-alveolar fibrin deposits did sometimes also label nonspecifically in both negative (Suppl. Fig. 2F) and positive test cases, but never to the extent that Theileria-specific positive labeling was obscured in the latter (Suppl. Fig. 2G).
Diagnostic sensitivity (DSe), diagnostic specificity (DSp), and 95% confidence intervals (CIs) for immunohistochemical assays utilizing 5 polyclonal antibodies (A–E) purified from 5 different roan antelope (Hippotragus equinus) sera.
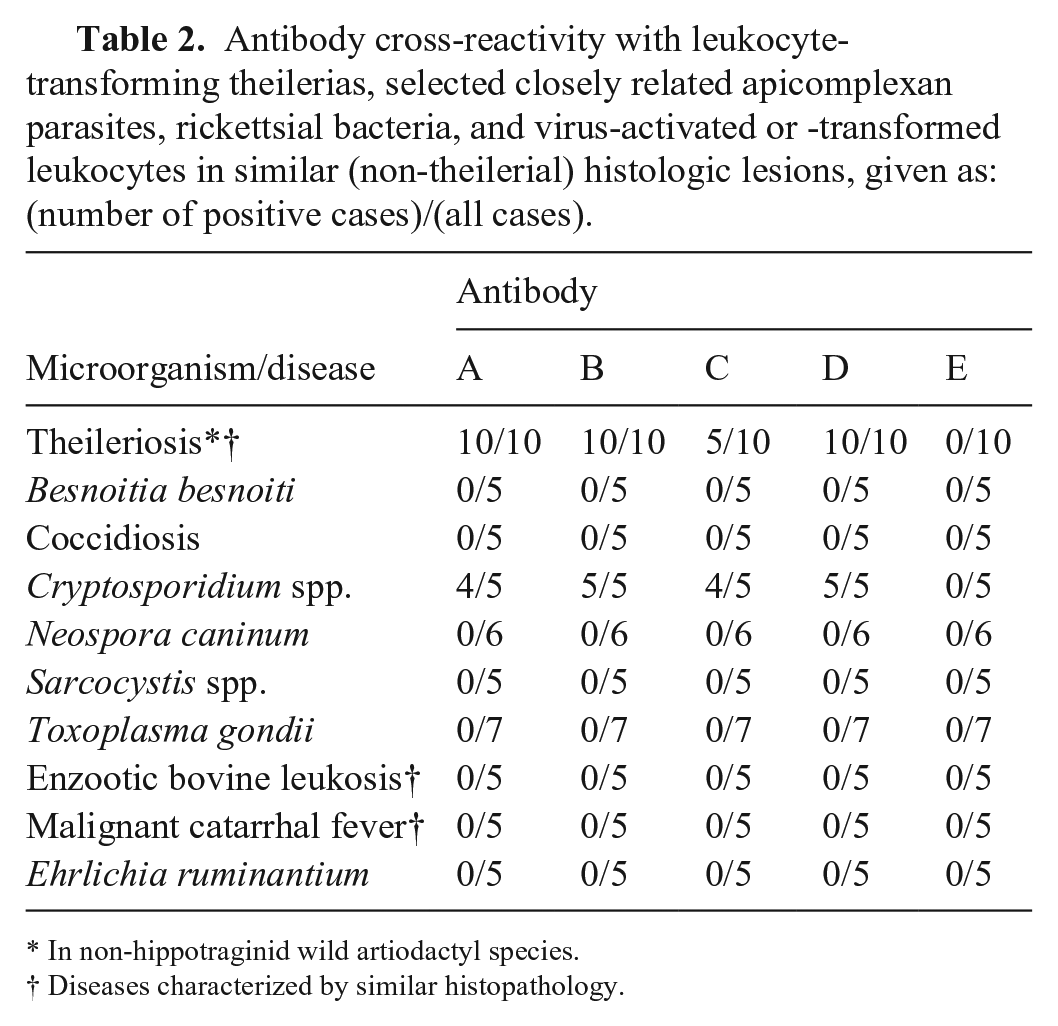
Assays using antibodies A, B, and D detected intraleukocytic Theileria sp. (sable) and other transforming theilerias in tissues from all 10 non-hippotraginid artiodactyls (Fig. 1E). Assays using antibody C stained only half of these cases, and those using antibody E stained none (Table 2). Antibody C specifically labeled Theileria sp. (sable) schizonts in tissues from 1 lechwe and 1 of 2 waterbuck (case T5) with Theileria sp. (sable)-positive RLB results (Suppl. Table 2). This antibody also positively identified theilerial parasites in another waterbuck with no PCR results (viz. tissues were not submitted for PCR) and cross-reacted with theilerial antigens in tissues from both eland that tested positive for T. taurotragi on RLB (Suppl. Table 2).
Antibody cross-reactivity with leukocyte-transforming theilerias, selected closely related apicomplexan parasites, rickettsial bacteria, and virus-activated or -transformed leukocytes in similar (non-theilerial) histologic lesions, given as: (number of positive cases)/(all cases).
In non-hippotraginid wild artiodactyl species.
Diseases characterized by similar histopathology.
Of the 9 potentially cross-reactive apicomplexan and rickettsial species and diseases with histologic lesions similar to theileriosis, only Cryptosporidium parasites stained consistently with antibodies A–D (Table 2; Fig. 1F). No other cross-reactivity was recorded (Table 2).
Histopathology alone and IHC assays (with antibodies A, B, and D) gave identical scoring for all 94 cases (χ2 = 89.926, df = 1, p << 0.001). The concordance among antibodies A, B, and D labeling was 100%, with all 55 positive roan and sable cases labeling positive, and all 39 negative roan and sable cases failing to label specifically. In all 39 Theileria-negative cases, IHC assays that utilized antibodies C and E were also negative. In the 5 Theileria-positive cases in which antibody E labeled intraleukocytic parasitic antigens, all of the other antibodies also detected theilerial antigens. However, in the remaining 50 Theileria-positive cases that labeled positive with antibodies A, B, and D, antibody C labeled 29 cases positive, but failed to label parasitic antigens in 21 cases.
Discussion
We developed 3 serum-purified pAbs that detected theilerial schizonts in tissues from roan and sable antelopes with lethal theileriosis with 100% sensitivity and specificity. Four of 5 antibodies cross-reacted with other Theileria spp. proteins in non-hippotraginid wild artiodactyl tissues, but there was minimal cross-reactivity with related coccidian parasites. The host species and theilerial antigen cross-reactivity observed in our study indicates that similarly made antibodies could be applied more widely to tissue specimens from diverse wild artiodactyl species with suspected acute theileriosis. On the other hand, the lack of cross-reactivity with diverse related (non-theilerial) coccidian parasites, selected rickettsial bacteria, and potentially confusing histologic lesions of importance in ruminants suggests that false-positive labeling is highly unlikely using similarly purified antibodies in the future.
The IHC test validation procedure that we followed, although preliminary, was detailed and extensive, using specifically developed antibodies. As recommended, 38 our antibody performance was gauged by comparing the IHC results with western blots. Demonstrating antibody binding with at least 2 completely different assays is advisable because immunoreactivity in one assay cannot necessarily be used to predict immunoreactivity in another. We established that the 3 best-performing antibodies (A, B, and D) were capable of detecting theilerial proteins that were either altered by formalin fixation (in cytospins and FFPE tissues used for IHC) or denatured in western blots.
A significant limitation to pAbs, which are purified from the blood of animals, is that they are produced in finite quantities, and antibody titers and quality can vary quite considerably between batches. 38 This may necessitate repeated optimization and validation for each batch of antibodies. However, the exact concordance among antibodies A, B, and D in our study, all purified from healthy roan yearlings with RLB-verified Theileria sp. (sable) DNA that yielded strong signals in western blots, suggests that similarly selected and purified polyclonal Theileria antibodies are likely to deliver reproducible results in FFPE roan and sable tissues. Also, given that roan and sable antelopes in southern Africa are commonly infected with Theileria sp. (sable) and/or closely related Theileria sp. (sable-like) species or genotypes,27,34 and that chemical immobilization is routinely performed for management purposes, obtaining blood samples for PCR and Theileria antibody purification is feasible for future studies. Furthermore, the antibody purification technique that we used was relatively simple and inexpensive, both important considerations in low and lower-middle-income regions including sub-Saharan Africa.
Polyclonal antibodies are more likely to be immunologically promiscuous than cell culture–derived mAbs that are targeted against a single epitope.37,38 Thus, pAbs are far more likely to cross-react with multiple molecules, both related and unrelated to the sought-after antigens/epitopes, leading to false-positive results with reduced specificity of the assay.37,38 In our study, all 5 roan antibodies cross-reacted to some extent with related (T. parva) and unrelated (bovine lymphocyte) proteins in western blots. Pertaining to the IHC assay, the 3 strongest polyclonal Theileria sp. (sable) antibodies (A, B, and D) detected Theileria sp. (sable) in a variety of non-hippotraginid wild artiodactyl species and also cross-reacted with T. taurotragi in 2 eland, but there was no nonspecific labeling of leukocytes in diverse FFPE tissues.
Although 4 of the 5 pAbs labeled Cryptosporidium parasites in the small intestines of diverse ungulate species, this will not lead to misdiagnosis of either disease because the clinical manifestations and lesions are completely different. 46 The cross-reactivity with Cryptosporidium was puzzling given that the genus is not phylogenetically more closely related to Theileria than the other coccidian parasites that we tested for antigen cross-reactivity. 41 The most likely explanation is that antibodies to Cryptosporidium were present in the roan polyclonal IgG antibodies. Cryptosporidium is an emerging pathogen in both the developed and developing world. 33 Oocysts have been identified in fecal samples from wild ungulates, specifically African buffalo and impala in game reserves in South Africa. 39 The parasite is known to be transmitted between livestock and wildlife, 49 and its significance in roan antelopes may warrant further investigation.
For positive tissue control purposes, we used a Theileria sp. (sable) schizont–infected continuous culture, which was derived from a lymph node biopsy from an infected roan antelope in South Africa. 51 This method could be used to develop mAbs in the future. However, although mAbs are likely to bind more specifically given their homogeneity, and can be produced in much larger quantities, 38 it is well known that the isolation and purification of intracellular proteins (e.g., theilerial schizonts) from cultured mammalian cells is difficult.19,45 Typically, complex laboratory techniques are required to extract a sufficient number of intact parasitic schizonts that have retained their antigenicity and are uncontaminated by host cell components. 45 The necessity for expensive laboratory equipment and technical expertise is a significant constraint in resource-poor regions, where theileriosis most commonly occurs in domestic and wild ungulates. More crude techniques with which to generate pure intracellular proteins from cultured cells 19 are worthy of consideration in the future.
Given that the sera from which antibodies C and E were purified were collected from 1- to 3-mo-old calves, the weaker Theileria sp. (sable) western blot signals (compared to the other antibodies) may indicate that these antibodies were maternally derived. Maternal transfer of theilerial antibodies via the colostrum has been established in several studies in cattle in East and sub-Saharan Africa, and these maternally derived antibodies reportedly persist in calves up to 5 mo old.28,47
Although 3 of the 5 antibodies reliably detected leukocyte-associated parasitic antigens in roan and sable tissues, there were some important study limitations. First, the DSe and DSp calculations were based on fewer samples than is optimal. 38 This is because roan and sable antelopes are scarce and valuable, and, as is common with wildlife fatalities, tissues were often at least moderately autolyzed at the time of collection, rendering them unusable for our study. Furthermore, variables that can affect the IHC reaction could not be investigated. For instance, there was no record of the time lapse between death and transfer of tissues to formalin. This is often the case in wildlife given the inaccessibility of carcasses, particularly in extensive camps. Although it is unlikely that the formalin fixation time exceeded 1–2 mo in our study, the elapsed time was not recorded for most cases. Both prolonged time to fixation and fixation time in formalin can hamper antigen accessibility to antibodies, resulting in loss of immunoreactivity.37,38 However, the consistent performance of the 3 best antibodies indicates that the assay is robust.
The lack of significant cross-reactivity with non-theilerial antigens, coupled with highly characteristic histologic lesions, makes IHC utilizing similarly purified pAbs an important consideration for future studies investigating tissue and cell tropism in hippotraginids with acute theileriosis. Ideally, however, histopathology and IHC should be combined with molecular techniques such as next-generation sequencing 34 to establish which theilerial species or genotypes are most commonly associated with a fatal outcome in these antelopes.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387211033272 – Supplemental material for Polyclonal antibody–based immunohistochemical detection of intraleukocytic Theileria parasites in roan and sable antelopes
Supplemental material, sj-pdf-1-vdi-10.1177_10406387211033272 for Polyclonal antibody–based immunohistochemical detection of intraleukocytic Theileria parasites in roan and sable antelopes by Sarah J. Clift, Bernat Martí-Garcia, Rephima M. Phaswane, Emily P. Mitchell, Antoinette I. Josemans, Ilse Vorster, Katja N. Koeppel and Jeanni Fehrsen in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
Special thanks to veterinarians Drs. Anndri and Greg Garrett for procuring the roan blood samples. We also thank the technical staff, particularly Ms. Naomi Timmerman, of the Histopathology and Immunohistochemistry Laboratories in the Section of Pathology, Department of Paraclinical Sciences (DPS), Faculty of Veterinary Science (FVS), University of Pretoria (UP) for excellent technical assistance. Thanks to the faculty pathologists and residents, especially Drs. Johan Steyl and June Williams who contributed FFPE tissue samples to our study. We extend our appreciation to Dr. John Lawrence, extraordinary professor (DPS, FVS, UP) for his editorial assistance, and to Ms. Caitlin Holyoake, veterinary student, for her invaluable assistance with data curation. We are also grateful to Dr. Kgomotso Sibeko-Matjila, Department of Veterinary Tropical Diseases, FVS, UP, and Mr. Christo Troskie of the Agricultural Research Council–Onderstepoort Veterinary Research Campus for their generous donations of cultured T. parva–infected leukocytes and uninfected bovine lymphocytes, respectively, for the western blots. We thank Estelle Mayhew in the Department for Education Innovation, FVS, UP for her help with the figures.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our study was supported by the Department of Paraclinical Sciences at the Faculty of Veterinary Science, University of Pretoria in South Africa, and the Health and Welfare Sector Education and Training Authority in South Africa.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
