Abstract
In North America, the only endemic focus for Angiostrongylus vasorum (French heartworm) was historically thought to occur in the southeastern part of the island of Newfoundland. However, reports of A. vasorum infection in wild canids in West Virginia, USA, and Nova Scotia, Canada, suggest the introduction of the parasite to mainland North America. We screened for A. vasorum in coyotes from across southern Ontario. Additionally, we evaluated the performance of ELISAs for detection of circulating A. vasorum antigen (Ag-ELISA) and antibodies against A. vasorum (Ab-ELISA) designed for use in sera or blood of foxes for use with coyotes in this region. Autopsies were performed on 397 coyotes, and lung tissue extract prepared from each carcass was tested via both ELISAs. The sensitivity and specificity for both tests were estimated in the absence of a gold standard using a 2-test single population Bayesian model; sensitivity and specificity priors were based on the performance of the assays in foxes in Switzerland. Eight coyotes tested positive for A. vasorum antigen; no animal was antibody positive. The estimated sensitivity and specificity of the Ag-ELISA were 90.8% (95% credible interval [CrI]: 83.8–95.6%) and 95.5% (95% CrI: 93.4–97.2%), respectively. For the Ab-ELISA, the estimated sensitivity and specificity were 41.9% (95% CrI: 32.1–51.9%) and 98.0% (95% CrI: 96.3–99.0%), respectively. Based on these findings and negative postmortem data for the same animals, there is insufficient evidence to suggest the presence of A. vasorum in southern Ontario coyotes.
Angiostrongylus vasorum (French heartworm) is a metastrongyloid nematode and the causative agent of canine pulmonary angiostrongylosis. Definitive hosts of A. vasorum are typically red foxes (Vulpes vulpes) and, importantly, domestic dogs (Canis familiaris), but can also include other wild canids such as coyotes (C. latrans), 33 wolves (C. lupus), 10 golden jackals (C. aureus), 41 or crab-eating foxes (Cerdocyon thous).13,18 A. vasorum resides in the right side of the heart and pulmonary arteries of infected definitive hosts; slugs and snails act as the intermediate hosts for the parasite. 30 Infection with A. vasorum can result in cardiopulmonary disease, central nervous system disease, and coagulopathies in domestic dogs6,24,40; however, clinical manifestation of canine pulmonary angiostrongylosis can range from subclinical to fatal. Domestic dogs may be clinically normal for months to years before manifesting clinical signs, and may become chronically ill, or die acutely.3,36
A. vasorum is widely distributed in Europe, South America, and parts of Africa. 3 In North America, the endemic focus for A. vasorum was considered to occur in the southeastern portion of the island of Newfoundland in red foxes and domestic dogs9,22; however, there have been reports of A. vasorum in wildlife in mainland North America. In 2011, a red fox in West Virginia, USA, was diagnosed with A. vasorum, based on identification of the parasite postmortem. 23 In 2018, A. vasorum was detected in 4 of 284 (1.4%) coyotes tested in Nova Scotia 33 ; infection status was determined by identification of the parasite postmortem and subsequent sequencing of cytochrome c oxidase subunit 1 on the mitochondrial genome of the parasite. Such reports suggest the introduction of the parasite in mainland North America.
In Europe, approximately since the year 2000, there has been an increase in the risk of exposure to A. vasorum for domestic dogs in established endemic areas and expansion into new geographic regions. 30 Because wild canids are considered an important reservoir for the parasite, several studies have been carried out on fox populations to estimate the risk of infection for domestic dogs within the same geographic region.11,16,42 Detection of A. vasorum is most often accomplished by either recovery of adult parasites from the heart and lungs during postmortem examination,29,42 or by larval isolation from feces 25 or bronchoalveolar fluid, 20 sometimes followed by biomolecular methods for confirmation. 2 The identification of adult parasites postmortem is the most applied procedure and represents a sensitive and cost-effective method. 20 However, false-negative results, especially when animals are infected with few parasites or were only recently infected, are possible. 17 Serologic tests are increasingly being used for detection of A. vasorum in domestic dogs,28,38 and have also been applied with sera or liquid bloody tissue extracts of wild canids. For example, ELISAs for detection of circulating A. vasorum antigen (Ag-ELISA) and antibodies (Ab-ELISA) have been validated for use with sera or blood from experimentally and naturally infected red foxes, 17 and a rapid assay has been used for detection of antigen from tissue fluid of foxes. 20 Such assays are advantageous because they allow for faster and less labor-intensive mass screening of wild canids for A. vasorum than postmortem identification, and therefore are more appropriate for large-scale surveys of wild canids.16,20
Although A. vasorum has not been reported in southern Ontario, there are concerns about the spread of the parasite into the region. Therefore, our objective was to screen for A. vasorum infection in coyotes from across southern Ontario using the aforementioned Ag-ELISA and Ab-ELISA, and postmortem examination.
Materials and methods
Coyote carcasses were obtained as a part of a study screening for Echinococcus multilocularis in southern Ontario; details of sample collection have been described previously. 26 Briefly, from November 2015 to March 2017 inclusive, 397 coyote carcasses were collected from across southern Ontario through collaboration with licensed hunters and trappers and the Ontario Ministry of Natural Resources and Forestry; fresh and frozen carcasses were submitted for a limited postmortem examination. It was not possible to collect fresh blood or sera from these carcasses because we relied on the submission of coyote carcasses with various degrees of decomposition and clotting. As an alternative, some studies have demonstrated that applying tissue extracts or body fluids may serve as a suitable substitute for serologic analysis,21,43 including cardiopulmonary tissue fluid from foxes used for A. vasorum antigen detection. 20 Therefore, lung tissue extract (LTE) for each carcass was prepared as an alternative to fresh blood or sera using a protocol modified from a previous study. 21 A 2-cm3 section of caudal lung lobe from either lung was removed from each carcass and placed in 2 mL of phosphate-buffered saline (PBS) in a 15-mL tube. The tube containing the lung lobe and PBS was then vortexed for 2 min (fixed speed vortex mixer; Fisher) and subsequently centrifuged for 10 min at 2,500 × g. The resultant supernatant was transferred to a 2-mL tube and stored at −20°C prior to analysis.
The A. vasorum infection status of each coyote was investigated postmortem while concurrently investigating for the presence of Dirofilaria immitis, as described previously. 27 Briefly, starting at the caudal vena cava, the heart was dissected following the flow of blood through the heart; the right atrium and ventricle were dissected along the septum into the pulmonary artery. Dissection continued following major pulmonary arterial branches and terminated at the branching of the left and right lobar arteries.
Serologic tests
LTE samples were analyzed using ELISAs for the detection of circulating A. vasorum antigen and antibodies against A. vasorum. For antigen detection, a sandwich-ELISA was used that is based on an A. vasorum–specific monoclonal antibody (mAb Av 56/1/2) and polyclonal rabbit antibodies directed against A. vasorum adult excretory or secretory antigen. 37 For antibody detection, a sandwich-ELISA based on A. vasorum adult somatic antigen purified with mAbs (mAb Av 5/5) was used. 39 Data are presented as individual optical density (OD) values, and OD cutoff values used were determined previously for blood samples from wild red foxes in Switzerland. 17 As determined previously in foxes, the sensitivity and specificity of the Ag-ELISA were 91.2% (95% confidence interval (CI): 83.9–95.9%) and 89.4% (95% CI: 82.2–94.4%), respectively; the sensitivity and specificity of the Ab-ELISA were 42.2% (95% CI: 32.4–52.3%) and 92% (95% CI: 85.4–96.3%), respectively. 17
The suitability of the LTE samples for detection of antibodies was evaluated by performing an ELISA for the detection of antibodies against Toxocara canis on 20 of the wild canid LTE samples adopting a cutoff that had been determined previously based on mean plus 3 SDs of the OD values of 18 defined negative samples. 14 T. canis is, among other parasites, vertically transmitted and therefore considered to be a very common parasite in wild canids.5,17 For example, high T. canis prevalence estimates of ≥ 70% have been reported in foxes in Switzerland. 14
Statistical analyses
Confidence intervals (CI) were estimated using Agresti–Coull CIs for prevalence estimates 1 with Stata/SE v.15.1 statistical software (StataCorp; https://www.stata.com).
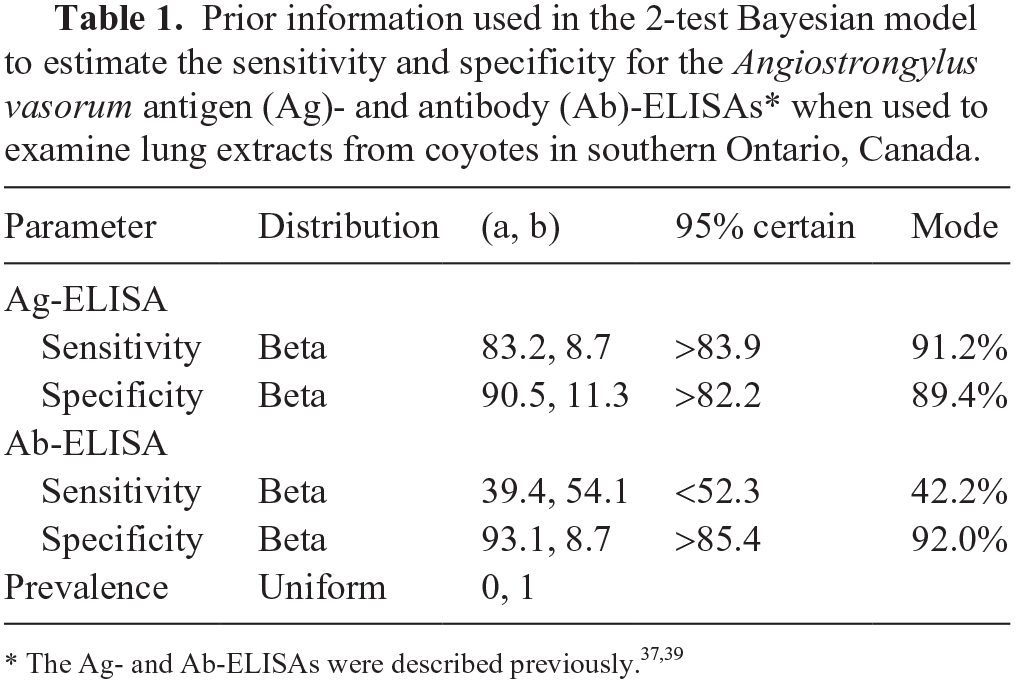
To our knowledge, the Ag- and Ab-ELISAs have not been investigated for use with LTE samples from coyotes in southern Ontario. We assessed the performance of the ELISAs for use with coyotes. However, as indicated, examination of carcasses for adult A. vasorum may lead to false-negative results and therefore may not be an appropriate gold standard. As an alternative, it has been suggested that estimation of the sensitivity and specificity of the Ag- and Ab-ELISAs may be more appropriate using Bayesian statistical inference techniques. 17 Thus, in the present work, sensitivities and specificities for the Ag- and Ab-ELISAs were estimated using a 2-test single-population Bayesian model, as described previously. 4 This model allowed for dependence between the 2 serologic tests. 12 The WinBUGS code for the 2-test, single-population Bayesian model is available in the supplemental material (Suppl. Code 2). Priors for the test sensitivities and specificities were based on sensitivity and specificity estimates derived from the aforementioned study that assessed the performance of these assays for blood samples from red foxes in Switzerland. 17 The final priors were represented by beta distributions in the format (a, b) and generated using R v.3.4.2 with R package LearnBayes 2.15.1 (https://cran.r-project.org; Suppl. Code 1). A non-informative prior represented by a uniform distribution (0, 1) was used for the prevalence parameter (Table 1).
Prior information used in the 2-test Bayesian model to estimate the sensitivity and specificity for the Angiostrongylus vasorum antigen (Ag)- and antibody (Ab)-ELISAs* when used to examine lung extracts from coyotes in southern Ontario, Canada.
The model was fitted using WinBUGS software (Imperial College & MRC, UK; https://www.mrc-bsu.cam.ac.uk/software/bugs/the-bugs-project-winbugs). Posterior estimates were generated using Markov chain Monte Carlo (MCMC) sampling via the Gibbs sampler. Model convergence was assessed visually by examining traces, histories, and Monte Carlo (MC) errors. Convergence Diagnostic and Output Analysis software (CODA) outputs were generated from WinBUGS, and convergence of the model was evaluated using the CODA package 0.19-3 in R.32,34 Geweke, Raftery & Lewis, and Heidelberger & Welch diagnostics were used.15,19,35 For MCMC sampling, 50,000 iterations were run for each analysis with the burn-in phase of 5,000 iterations deleted. The final estimates for the performance of the 2 tests were based on their posterior medians and 95% credible intervals (CrI) from MCMC sampling.
A sensitivity analysis was performed to assess the influence of the priors on the test performance estimates. The prior distributions for the sensitivity and specificity of the Ag- and Ab-ELISAs were replaced one at a time with non-informative prior information represented by beta distributions (1, 1).
Results
Adult A. vasorum were not detected during postmortem examination of 397 coyote carcasses.
Angiostrongylus vasorum antigen and antibody detection
Overall, 8 of 397 (2.0%; 95% CI: 1.0–4.0%) coyotes had LTE samples with an OD ≥ 0.260 by Ag-ELISA and were considered positive (Suppl. Table 1). Zero of the 397 (0%; 95% CI: 0–1.2%) coyotes had LTE samples with an OD ≥ 0.144 when tested via the A. vasorum Ab-ELISA. The suitability of LTE samples for detection of antibodies was supported by detection of antibodies to T. canis in 19 of the 20 coyotes tested (Suppl. Table 2).
Statistical analyses
The estimated specificities of the Ag- and Ab-ELISAs exceeded 95%, but the estimated sensitivity of the Ag-ELISA was substantially higher than the Ab-ELISA (Table 2). Inspection of the trace plots, histories, and MC errors of all nodes suggested model convergence. This was further corroborated when the Geweke, Raftery & Lewis, and Heidelberger & Welch diagnostics were performed.
Performance estimates of the Angiostrongylus vasorum antigen (Ag)- and antibody (Ab)-ELISAs* for lung extract samples from 397 coyotes in southern Ontario, Canada.

95% credible interval in parentheses.
There was little appreciable effect (change < 2% of the median value) in the posterior estimates of the specificity of both tests when non-informative distributions were used as priors for any parameter. In contrast, a larger change in the posterior estimates for the Ag-ELISA sensitivity (−48%) and the Ab-ELISA sensitivity (−12%) was observed when non-informative priors were applied, suggesting a stronger effect for these priors on their respective parameters.
Discussion
Previous work has demonstrated that combined examination of fox sera and/or whole blood samples for A. vasorum antigen and antibody provides a suitable method for epidemiologic screening of foxes for A. vasorum infection.16,17 We found 8 of 397 coyote LTE samples positive for A. vasorum antigen using the same Ag-ELISA. However, no animals were antibody positive using the Ab-ELISA.
Results from the Bayesian analysis suggest that the 2 serologic tests perform similarly for LTE from coyotes in southern Ontario compared to the reported performance of the assays for blood from red foxes in Switzerland. The estimated specificities of the Ag- and Ab-ELISAs were 95.5% and 98.0% when applied to coyote LTE samples, respectively. In comparison, the reported specificities for the Ag- and Ab-ELISAs when used with sera or blood from foxes were 89.4% (95% CI: 82.2–94.4%) and 92.0% (95% CI: 85.42–96.29%), respectively. 17 Notably, based on the estimated specificity of the Ag-ELISA (95.5%) in our study, one would expect ~18 of 397 false positives (95% CrI: 12–26) for A. vasorum antigen. Therefore, it cannot be ruled out that the aforementioned 8 positive results in our study were false-positive results. On the other hand, no animal tested positive for A. vasorum antibody.
It is unlikely that LTE is inappropriate for antibody detection given that we demonstrated that the sample type is fundamentally suitable; T. canis antibodies were detected in 19 of 20 of the same coyotes. It is of interest to note that the reported sensitivity of the Ab-ELISA, when used with blood from foxes, is low (42.2%; 95% CI: 32.4–52.3%), which is likely the result of the highly variable antibody response in foxes. Decreasing antibody levels after ~7 wk post-infection were demonstrated in experimentally infected foxes in Denmark despite never receiving anthelmintic treatment, and adult A. vasorum specimens were confirmed at postmortem examination. 17 It is likely that foxes need reinfections to remain antibody positive. 17 Perhaps a similar antibody response exists in coyotes, which could explain why coyotes in our study were antigen-positive but antibody-negative; however, to our knowledge, the A. vasorum antibody response has not been investigated in this species. Overall, in light of the Ag-ELISA results and the negative postmortem findings, there is insufficient evidence to suggest the presence of A. vasorum in the coyote population in southern Ontario.
There are several limitations that should be considered for our study. First, Bayesian analysis allows for the incorporation of previous knowledge in the form of prior distributions. This not only can help derive posterior estimates of the parameters but can also have a strong influence on the results when the amount of data is scarce or not robust. Notably, our sensitivity analysis indicated that the posterior estimates for the Ag-ELISA and Ab-ELISA sensitivities were influenced by the prior distributions of their respective parameters. This likely is the result of the low effective sample sizes of antigen-positive and antibody-positive coyotes. Second, our postmortem examination of the coyote carcasses for the presence of A. vasorum parasites was limited to dissection of the heart and major pulmonary vessels, terminating at the left and right lobar arteries. Notably, after death, it is possible for adult A. vasorum parasites to be found in the periphery of the lung lobes; flushing the lungs in addition to fine dissection of all heart and pulmonary vessels is recommended for surveys for A. vasorum.17,31 This combination was found to be an effective way of detecting A. vasorum that may otherwise be missed by coarse dissection.
Although we did not find sufficient evidence to suggest the presence of A. vasorum in southern Ontario, the recent detection of the parasite in Nova Scotia, Canada, and West Virginia, USA, raises concerns about the spread of the parasite across mainland North America.23,33 In particular, it has been suggested that a biological indicator of a region conducive to the establishment of A. vasorum is the fox lungworm (Crenosoma vulpis), which shares the same lifecycle requirements (e.g., wild canid and terrestrial gastropod hosts) as A. vasorum. 7 Although we did not investigate the presence of C. vulpis in the coyotes examined for our study, C. vulpis does occur in Ontario. 8 Thus, the possibility of the emergence of A. vasorum in Ontario should be re-evaluated.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387211011990 – Supplemental material for Investigation of the occurrence of Angiostrongylus vasorum in coyotes in southern Ontario, Canada
Supplemental material, sj-pdf-1-vdi-10.1177_10406387211011990 for Investigation of the occurrence of Angiostrongylus vasorum in coyotes in southern Ontario, Canada by Jonathon D. Kotwa, Manuela Schnyder, Claire M. Jardine, Peter Deplazes, David L. Pearl, Olaf Berke, Nicola Mercer and Andrew S. Peregrine in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the southern Ontario hunters and trappers, Aylmer District Stakeholders Committee, Nuisance Wildlife Control Inc., and the Ontario Ministry of Natural Resources and Forestry for coyote submissions. The Canadian Wildlife Health Cooperative contributed invaluable resources and support for the processing of wild canid samples. Many thanks to Dr. G. Douglas Campbell for his assistance and guidance during the autopsy process. Dr. Tamara Hofstede, Bayer Animal Health Canada, is thanked for facilitating testing of samples, and Nataliya Miroshnikova for support in testing samples for A. vasorum antigen and antibody detection. Many thanks to Dr. Cathy Bauman for guidance on the Bayesian analysis.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by Bayer Animal Health (Canada), the National Center for Veterinary Parasitology (USA), Natural Sciences and Engineering Research Council of Canada, Ontario Veterinary College, Ontario Animal Health Network, and Burroughs-Wellcome Fund.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
