Abstract
The urine protein:creatinine (UPC) ratio is considered the reference method to assess proteinuria. Its diagnostic value in ovine medicine needs further elucidation. In population monitoring and/or for research purposes, it is convenient to collect many samples simultaneously and store them for later analysis. However, analyte stability data are required to ensure reliable results. We used 15 of 90 urine samples collected from sheep to assess the effect of storage time on the UPC ratio. After centrifugation, the supernatant of each sample was divided into 6 aliquots. Urine protein and creatinine concentrations were determined immediately in one aliquot using the pyrogallol red and a modified Jaffè method, respectively. The other aliquots were stored at −18°C. Based on the absence of active sediment, alkaline urine pH, and UPC ratio ≥0.2, we included 15 samples in our study. The UPC ratio was determined in the stored aliquots 2, 7, 14, 21, and 60 d after collection. The data were analyzed with univariate ANOVA. No significant difference was observed in the urinary concentrations of protein, creatinine, and the UPC ratio (0.8 ± 0.84 in conventional units and 0.09 ± 0.095 in SI units) among different times (
The term
Tests have been developed for the detection of proteinuria, including the dipstick colorimetric test as a first-line screening test, the sulfosalicylic turbidimetric test or the Heller reaction, and the urine protein:creatinine (UPC) ratio. 6 The latter is a protein quantification method that normalizes the urine protein concentrations, adjusting the variances arising from differences in urine volume and specific gravity. The spot, or random, UPC ratio reflects 24-h protein excretion; testing more than one random sample is not deemed mandatory to obtain reasonably precise results.5,7
Regarding the use of the UPC ratio for the estimation of proteinuria in ruminants, there are 2 recent publications in cattle9,10; in sheep, an increased UPC ratio, compared to reported data on protein and creatinine excretion in heathy sheep, 4 has been associated with naturally occurring reactive systemic amyloidosis.3,11 To further explore the diagnostic value of the UPC ratio in ovine medicine, given that it is more convenient to collect many samples from the same farm at the same time and store them for later analysis, stability of protein and creatinine in urine samples should be determined to minimize the risk of erroneous results. 8 Urine protein and creatinine stability data are available for normally acidic urine in humans12,14 and dogs,15,16,18 but are lacking for the normally alkaline urine of ruminants.
Our aim was to assess the effect of storage duration at −18°C on urine protein, creatinine, and the subsequent UPC ratio in ovine samples. All procedures were carried out in accordance with the ethical standards of the Helsinki Declaration of 1975 (revised in 2000), the national law of Greece, and after receiving approval from our Institutional Animal Use Ethics Committee.
Urine samples were collected from 90 dairy ewes during lactation. The samples were obtained in the milking parlor; voiding was induced by holding the nostrils closed for a few seconds to cause transient apnea, and by rubbing the area of the vulva. 1 To reduce the risk of the samples being contaminated, midstream urine was collected into sterile plastic containers and then transferred immediately to the laboratory on ice.
Within an hour after collection, 10 mL of well-mixed urine was transferred to a clear conical centrifuge tube and centrifuged at ~250 ×
For the evaluation of urine sediment, the pellet in the conical tube was resuspended in a small volume of urine. The sediment was examined microscopically both as an unstained wet preparation and as an air-dried Giemsa-stained smear to better evaluate cellular constituents and exclude bacteriuria. 13 The number of red blood cells (RBCs) and white blood cells (WBCs) was recorded per 40× high-power fields (hpf) in 10 hpf as a range per hpf, as described previously. 21 The presence of epithelial cells and casts was assessed per 10× low-power fields (lpf) in 10 lpf as a range per lpf. A semiquantitative scale (none, few, moderate, many) was used to assess the presence and identify the type of microorganisms and crystals. The classification of sediments as active was based on the presence of one or more of the following findings: >5 RBCs or >8 WBCs per hpf, bacteria, or >10 epithelial cells per lpf. 21 Samples with none of these findings were classified as inactive.
Of the 90 samples collected, we selected 15 for our study based on the following criteria: 1) absence of active sediment, 2) alkaline pH, 3) normal, other than protein dipstick results (absence of glycosuria, ketonuria, hematuria, hemoglobinuria, bilirubinuria), and 4) UPC ratio >0.2 calculated in conventional units on the day of sampling. This UPC ratio cutoff has been used in other animal species, including ruminants. 10 Urine protein and creatinine concentrations were determined in the remaining 5 stored aliquots of the same sample 48 h after sampling and 7, 14, 21, and 60 d after collection.
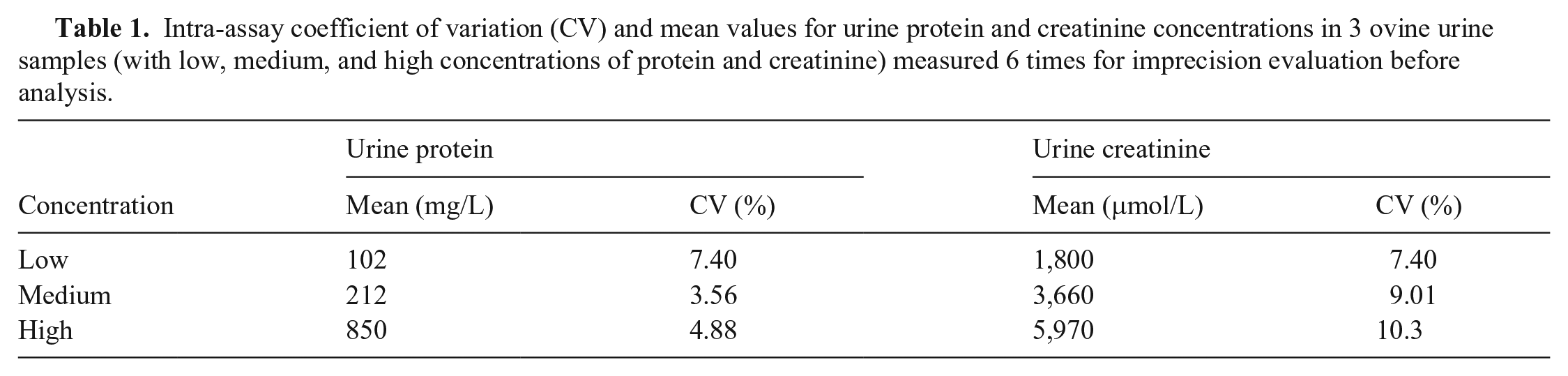
Urine protein concentrations were determined with the pyrogallol red method from the supernatant of the urine without prior dilution; urine creatinine concentrations were determined with a modified Jaffè method from the prediluted to 1:20 supernatant of urine samples using an automated analyzer (Vitalab Flexor E; Vital Scientific) and commercial kits. The intra-assay coefficient of variation (CV) of each analyte was evaluated by repeating urine sample measurements 6 times at 3 different concentrations for each analyte prior to the onset of our study (Table 1). The UPC ratio was calculated employing the following formulae: UPC = urine protein (mg/L)/urine creatinine (μmol/L) in SI units, and UPC = urine protein (mg/dL)/urine creatinine (mg/dL) in conventional units.
Intra-assay coefficient of variation (CV) and mean values for urine protein and creatinine concentrations in 3 ovine urine samples (with low, medium, and high concentrations of protein and creatinine) measured 6 times for imprecision evaluation before analysis.
Data were analyzed using SPSS Statistics v.25 (IBM). Normality of data distribution was assessed with the Kolmogorov–Smirnov test, and homogeneity of variances was evaluated with the Levene test. Repeated-measures ANOVA was run to evaluate the effect of storage time on the parameters tested. Bonferroni test was used for the adjustment of confidence interval. A value of
No significant differences (
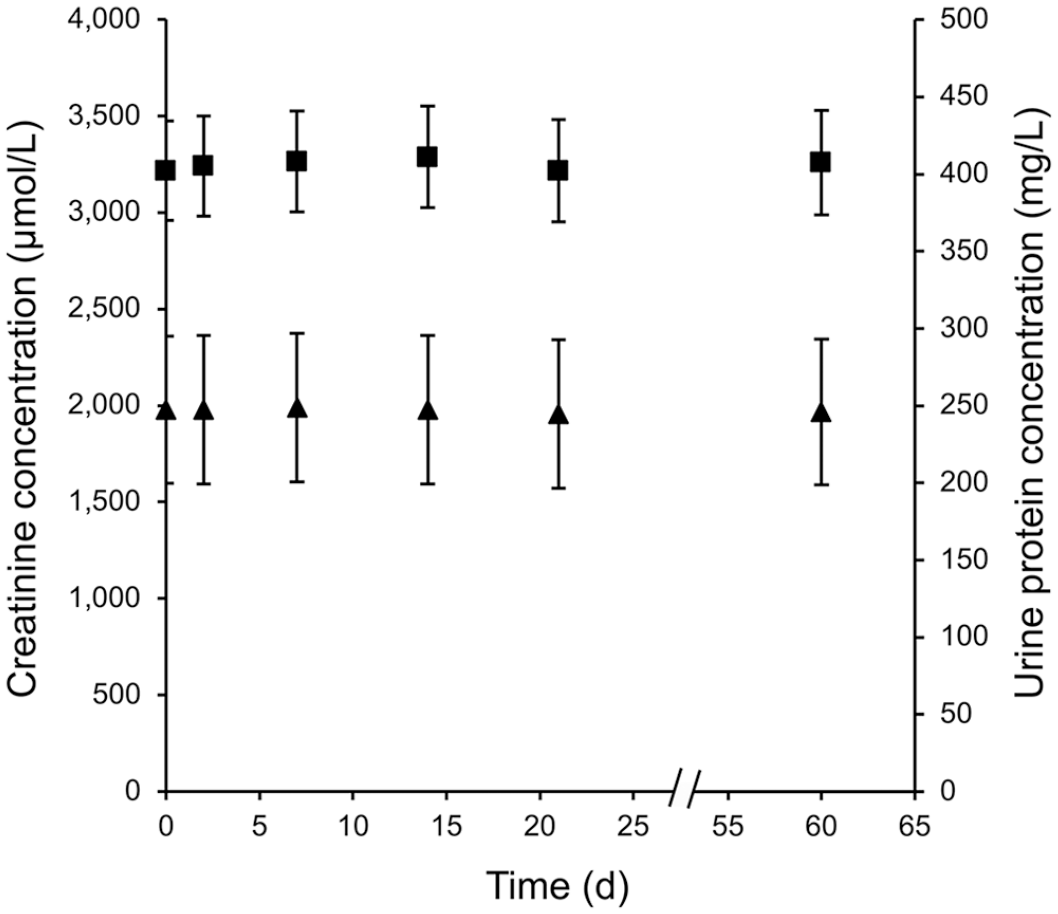
Mean concentrations of urine protein (▲) and creatinine (■) and standard error (bars) in 15 ovine urine samples 2, 7, 14, 21, and 60 d after sampling. No significant difference was detected among the times within each parameter based on repeated-measures ANOVA.
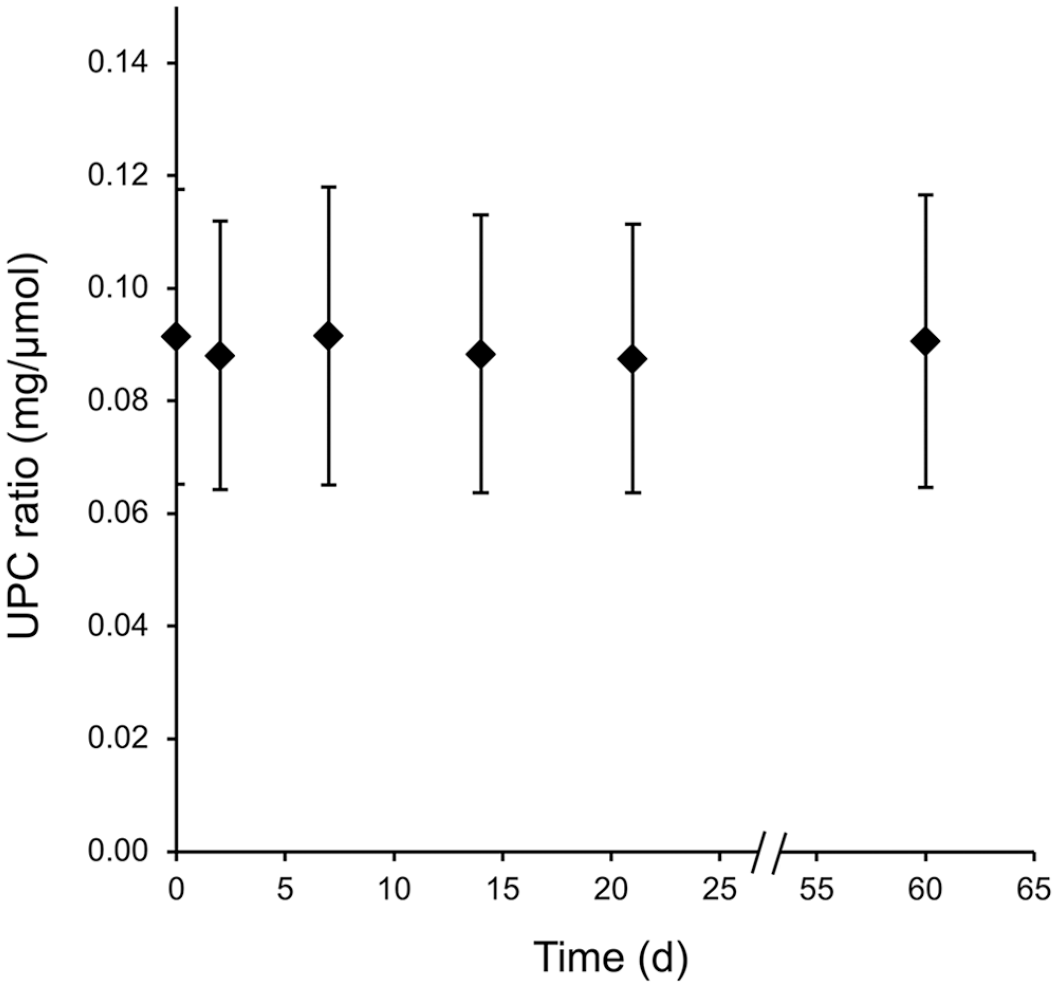
Mean urine protein:creatinine (UPC) ratio in SI units (mg/µmol; ♦) and standard error (bars) in 15 ovine urine samples 2, 7, 14, 21, and 60 d after sampling. No significant difference was detected among the times based on repeated-measures ANOVA.
Performing urinalysis as soon as possible after collection is recommended to minimize artefactual changes. 8 However, in experimental studies, especially those conducted in farm animals, it is more convenient to collect samples and store them for later analysis. Our results indicate that urine protein and creatinine concentrations and, consequently, UPC ratios are not affected significantly by 60 d of storage at −18°C. The relatively higher CV observed for creatinine compared to protein, especially for the high values, is probably attributed to the Jaffè creatinine methodology used 2 and the dilution of the sample required. However, this is the most commonly used method for urine creatinine determination given its simplicity and its low cost.
In a study of the stability of albumin in human urine, storage at −20°C for 6–8 mo showed underestimation of protein, but not creatinine. 17 However, in that study, both the analyte determined and the method employed were different than the ones that we used: albumin instead of total protein, and ELISA instead of biochemical analysis. Evidence in the literature regarding canine urine samples suggests that storage may cause specific alterations in the protein fraction. Storage at −20°C for >15 d resulted in the appearance or strengthening of protein bands with a molecular mass ≥ 150 kDa. 18 In the same study, a mild though significant decrease was detected in the UPC ratio in canine urine samples stored at −20°C for 30–180 d, mainly as a result of the decrease in protein concentration. On the contrary, in another study, 16 the UPC ratio increased after storage for 12 h at 20°C as a result of an increase of urine protein concentration, rather than to a decrease of urine creatinine concentration, and remained stable for a further 3 mo.
Apart from the interlaboratory variability 15 that could account, at least partially, for the differences observed among the results of the 2 aforementioned studies and our study, the magnitude of proteinuria seems to affect the stability of proteins. In non-proteinuric dogs, the UPC ratio significantly decreased with time after >15 d of storage, whereas this period was more prolonged to 30–180 d in proteinuric dogs. 18 A cutoff for the UPC ratio for defining proteinuria in ovine urine samples is still to be determined. A UPC ratio range of 0.21–1.34 has been reported in healthy sheep 4 and, these are, to the best of our knowledge, the only references for the UPC ratio in ovine urine. However, the kidneys of those healthy sheep were not without lesions given that histologic examination revealed mild glomerulonephritis. Based on this evidence and on previous reports for cattle, which also have alkaline urine,9,10 we selected the UPC ratio of 0.2 for defining proteinuria, and this was one of the inclusion criteria for the samples evaluated to be considered as proteinuric.
Most UPC ratio stability studies have been performed in species with acidic urine. It has been reported that the mean albumin value in voided acidic urine decreased 10 wk after storage at −20°C, whereas in neutral pH-adjusted samples it remained fairly stable. 14 Although albumin is only a fraction of the proteins determined in our study, the effect of storage on the samples with normal alkaline pH of sheep seems to be minimal. Regarding the effect of pH on the stability of creatinine, it is not substantial, at least within a pH range of 5.4 to 8.2, 12 which is in accordance with our study as well as with the results of other studies performed in species with normally acidic urine.16,18
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387211031517 – Supplemental material for Effect of storage time on the urine protein: creatinine ratio in alkaline ovine urine
Supplemental material, sj-pdf-1-vdi-10.1177_10406387211031517 for Effect of storage time on the urine protein: creatinine ratio in alkaline ovine urine by Labrini V. Athanasiou, Panagiotis D. Katsoulos, Victoria M. Spanou, Aikaterini T. Pazarakioti, Eleni G. Katsogiannou, Ioanna Iliadi, Rania Baka and Zoe S. Polizopoulou in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
