Abstract
A 25-y-old Percheron mare was admitted to the teaching hospital because of lethargy and intractable dyspnea. Thoracoabdominal ultrasound examination identified severe peritoneal effusion, mild bilateral pleural effusion, and a diffuse pulmonary nodular pattern. Cytology of peritoneal fluid revealed a hypercellular sample with clusters of neoplastic polygonal cells and admixed macrophages. Euthanasia was followed by postmortem examination; marked bi-cavitary effusion was present, and innumerable up to 4-cm diameter, round-to-floriform nodules were diffusely evident throughout serosal surfaces as well as the pulmonary and hepatic parenchyma. Disseminated adenocarcinoma, predominantly affecting lung and liver with widespread serosal implantation, was confirmed on light microscopy. Neoplastic cells had strong immunolabeling for pancytokeratin and lacked immunoreactivity to vimentin, napsin A, and Pax8. Cytokeratin 7 and thyroid transcription factor-1 were non-contributory given absent and inconsistent internal control reactivity, respectively. Such results, combined with the lack of a major mass that would indicate a primary site, were supportive of carcinoma of unknown primary site, which remains a conundrum in human oncology, and is poorly explored in veterinary medicine, mainly as a result of clinical and diagnostic limitations.
In veterinary medicine, carcinomatosis often results from ovarian, gastrointestinal, pancreatic, mammary, renal, and uterine cancers, 20 and yet is infrequently seen in equine patients. Of the few published cases, equine renal cell carcinoma (RCC),18,19 gastric squamous cell carcinoma, 17 and cholangiocellular carcinoma5,20 are the primary sites identified most frequently, primarily based on identifying a larger dominant neoplasm affecting the organ of origin. Nonetheless, a larger mass than would be expected for a primary site may not be appreciated in some instances, which is one of the hallmarks of cancer of unknown primary (CUP).
In human oncology, CUP is a cryptic entity that encompasses a heterogeneous group of metastatic tumors in which a primary site cannot be identified even after diligent searching. In addition, tumors included under this classification usually share particular features, such as early metastasis, regression or dormancy of primary site, and aggressive behavior.8,12 Extensive physical examination, computed tomography of the abdomen, thorax, and pelvis, whole-body positron emission tomography–computed tomography (PET-CT), and a series of immunohistochemistry (IHC) markers are usually required before diagnosing CUP, 12 which is considered a diagnosis by exclusion. 7 Veterinary studies are sparse, likely because of unfamiliarity with this entity and/or logistic or financial limitations for diagnostic tests. 14
A 25-y-old gray Percheron mare was presented to the teaching hospital with a history of refractory dyspnea. Upon presentation, the mare was in poor body condition (body score 2 of 9), lethargic, and dehydrated. Severe tachycardia (84 beats/min), moderate tachypnea (36 breaths/min), and increased respiratory effort were noted; rectal temperature was normal (37.9°C; 100.3°F). An enlarged lymph node was palpated at the base of the neck, and multiple cutaneous masses were detected at the base of the tail, presumably consistent with melanocytic tumors. Transrectal palpation was unremarkable.
Complete blood cell count revealed moderate-to-severe leukocytosis (25.2 × 109/L, reference interval [RI]: 6.0–12.0 × 109/L) with mature neutrophilia (22 × 109/L, RI: 3.0–6.0 × 109/L), monocytosis (2 × 109/L, RI: 0–0.6 × 109/L), and lymphopenia (1 × 109/L, RI: 1.5–5.0 × 109/L). Blood chemistry revealed moderate hypoalbuminemia (24.2 g/L, RI: 27.0–41.0 g/L), mildly increased sorbitol dehydrogenase (12.9 U/L, RI: 0.5–10.0 U/L), aspartate aminotransferase (480 U/L, RI: 144–350 U/L), and gamma-glutamyl transferase (35 U/L, RI: 2–29 U/L) activities.
Transcutaneous thoracoabdominal ultrasound examination revealed severe peritoneal effusion, severe diffuse bilateral lung consolidation with a diffuse nodular pattern, and mild pleural effusion. Peritoneal fluid collected via abdominocentesis was submitted for cytology and had 20–30 × 109/L cellularity, 75% being large polygonal neoplastic cells with vacuolated cytoplasm arranged in cohesive clusters (Fig. 1), with interspersed small lymphocytes and macrophages, interpreted as a neoplastic effusion. Mesothelioma, carcinomatosis, and histiocytic sarcoma were listed as differential diagnoses. Based on the critical clinical condition and the poor prognosis, the mare was euthanized, and autopsy was followed by histopathology.
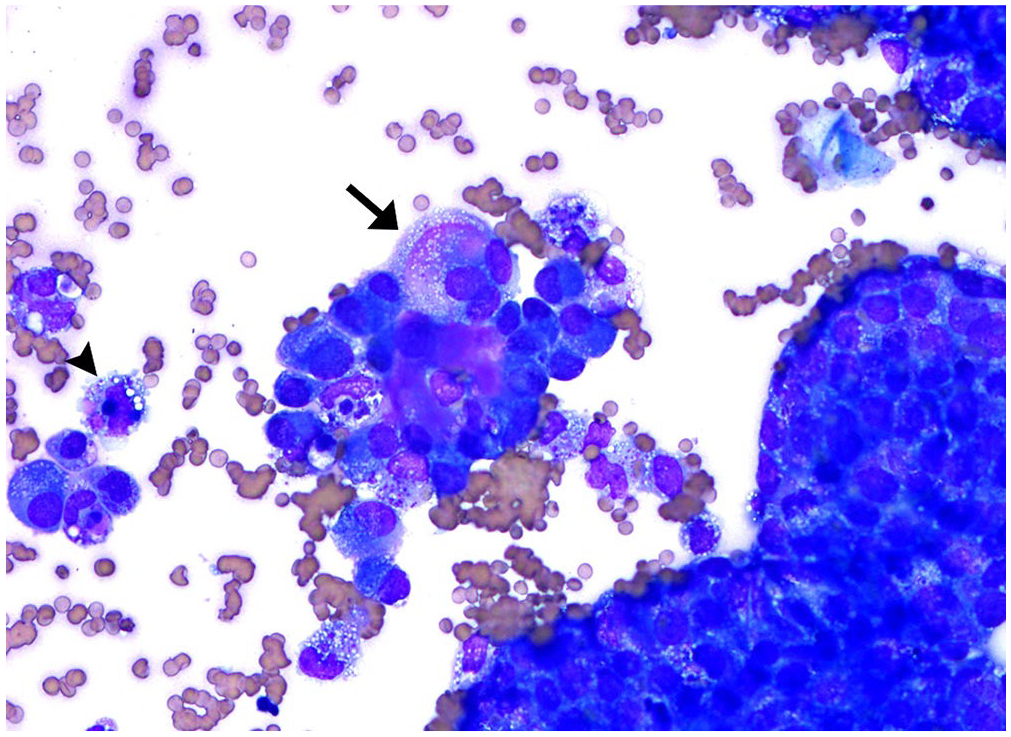
Cytopathology of peritoneal fluid of a mare containing clusters of round-to-polygonal neoplastic cells with variable amounts of vacuolated cytoplasm. Anisocytosis and anisokaryosis are moderate with some multinucleation (arrow). Erythrocyte- and hemosiderin-laden macrophages are scattered throughout (arrowhead). Romanowsky stain.
Postmortem examination revealed 25 L of serosanguineous peritoneal effusion and ~3 L of similar effusion in the pleural space. Innumerable pinpoint to 4-cm diameter white-tan, firm, round-to-floriform nodules covered the serosal, peritoneal, retroperitoneal, and pleural surfaces (Fig. 2A) and extended to the cut surface of the liver (Fig. 2B) and lung. All remaining viscera were similarly affected, with masses distributed throughout their serosa and no apparent dominant mass. Many of these masses had a white, chalky, necrotic core. The reproductive system, including the mammary gland, was unremarkable.
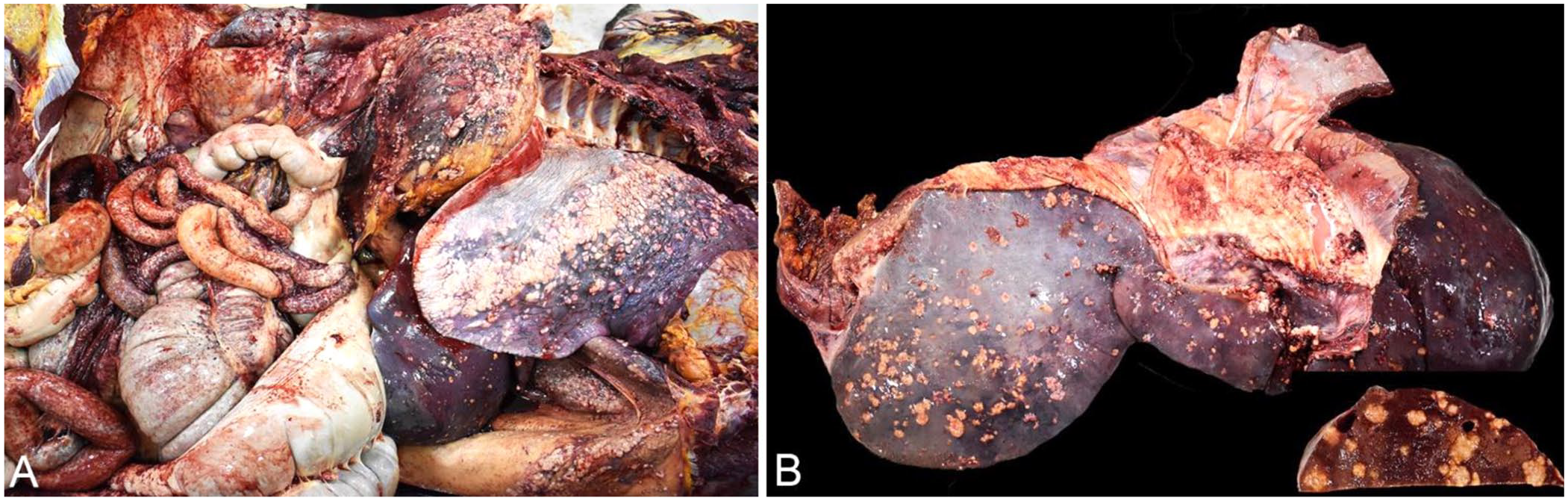
Adenocarcinoma of unknown primary site in a mare.
Histologically, a multicentric, non-encapsulated, densely cellular neoplasm of cuboidal-to-columnar cells forming tortuous anastomosing tubules and acini replaced the hepatic (Fig. 3A) and pulmonary parenchyma (Fig. 3B). Neoplastic cells had a moderate amount of eosinophilic cytoplasm with variably defined cell borders. Nuclei were mostly basilar, round-to-ovoid with finely stippled chromatin and an indistinct nucleolus. Anisocytosis and anisokaryosis were moderate, and there were 69 mitotic figures in 10 consecutive 400× (2.37 mm2) fields. No intraluminal secretory product was identifiable within neoplastic tubules, and desmoplasia was not a prominent feature. Coalescent serpiginous areas of leukocytic and karyorrhectic cellular debris comprised 20–30% of the central areas of the neoplasm.
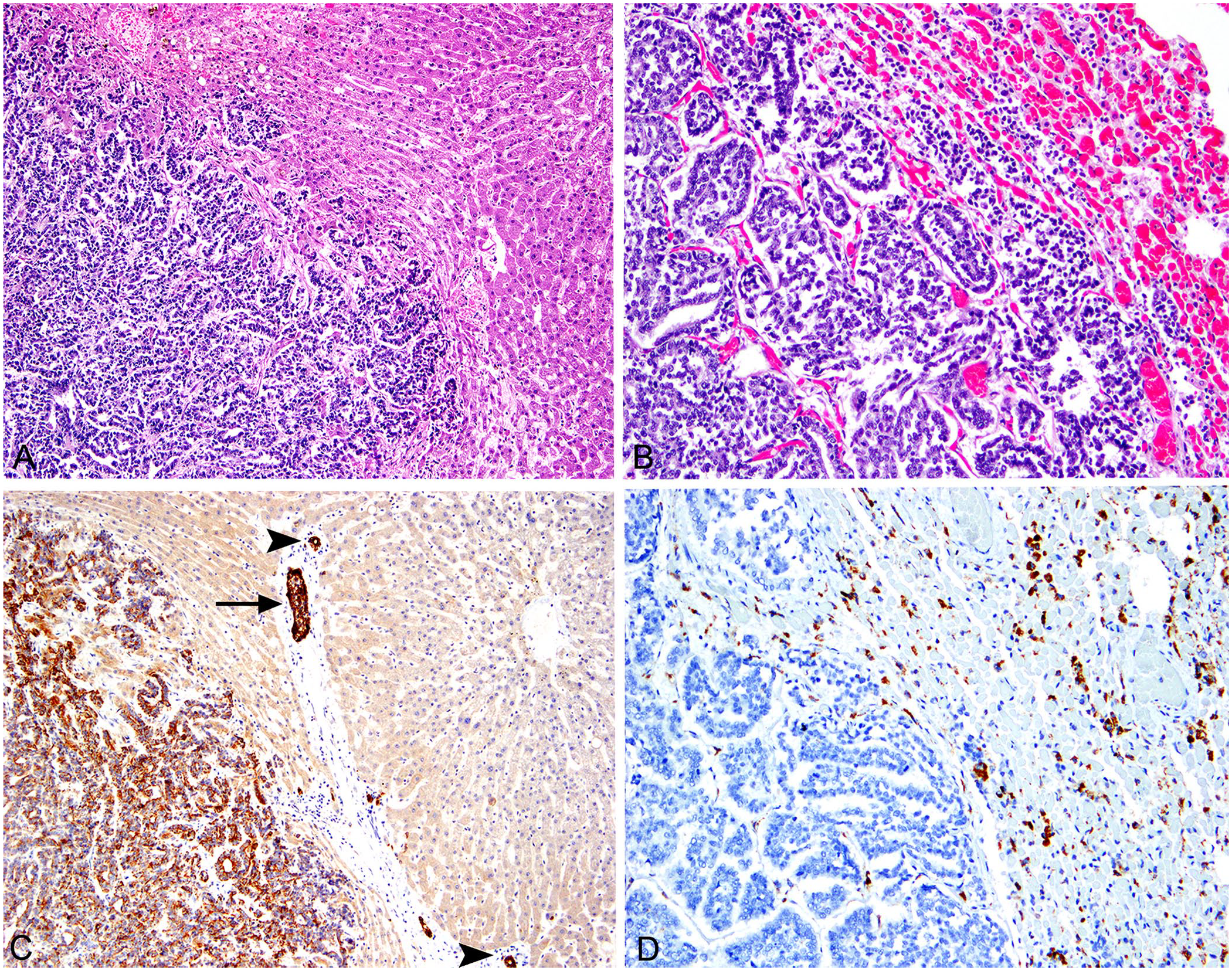
Adenocarcinoma of unknown primary site in a mare.
The assorted immunohistochemical stains applied to our case had variable results (Table 1). Neoplastic cells had strong and diffuse cytoplasmic immunoreactivity to pancytokeratin (AE1/AE3; mouse monoclonal antibody, Biocare Medical; Fig. 3C) and lacked immunoreactivity to vimentin (mouse monoclonal antibody; Biocare Medical), napsin A (rabbit monoclonal antibody, Biocare Medical; Fig. 3D), and Pax8 (mouse monoclonal antibody; Biocare Medical). The lack of reactivity in normal biliary duct epithelium cells with cytokeratin 7 (CK7; mouse monoclonal antibody, Dako), as well as faint nuclear labeling of a few bronchiolar epithelial cells, and not in alveolar cells with thyroid transcription factor-1 (TTF-1; mouse monoclonal antibody; Biocare Medical), were likely the result of abortive cross-reactivity in equine tissue and, thus, non-contributory in our case.
Labeling results of immunomarkers applied to the adenocarcinoma of unknown primary in a mare and other equine control tissues.
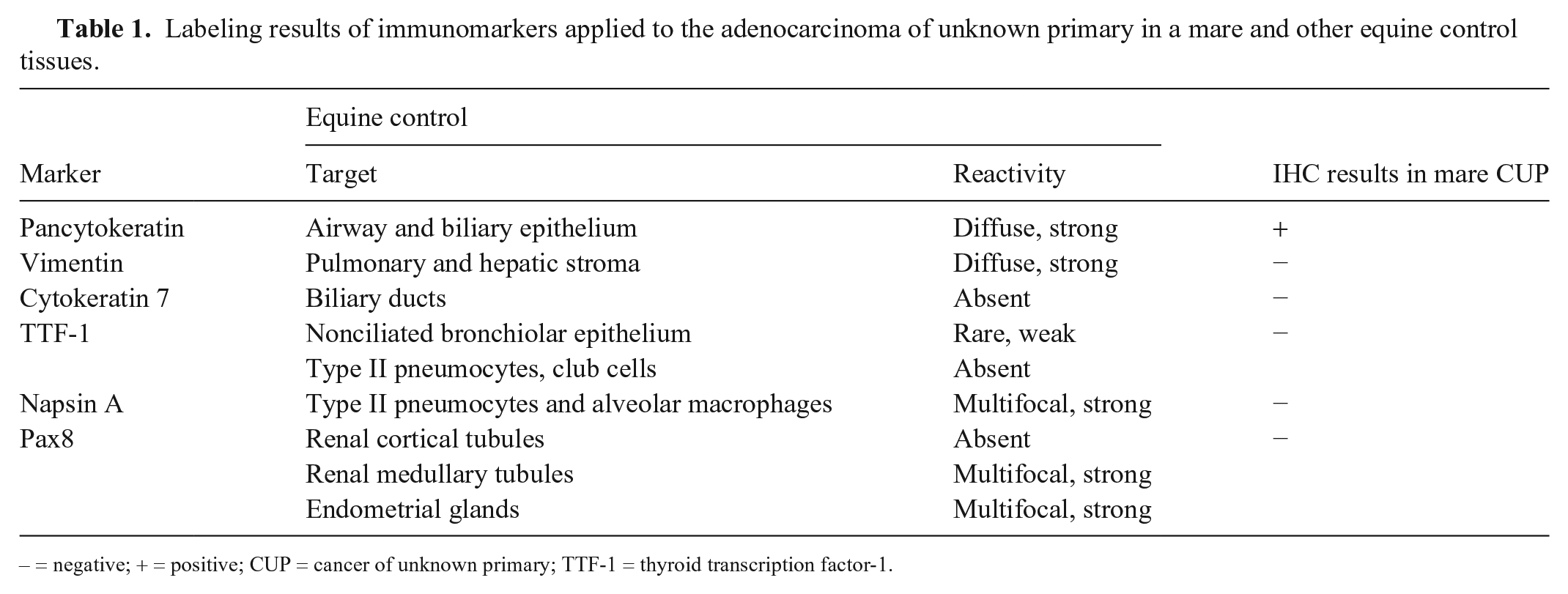
– = negative; + = positive; CUP = cancer of unknown primary; TTF-1 = thyroid transcription factor-1.
Based on clinicopathologic findings followed by IHC panel results, a diagnosis of disseminated adenocarcinoma of unknown primary site with peritoneal and pleural carcinomatosis was established for this mare. Such a presentation is uncommon in horses; cancers originating from the kidney,18,19 stomach, 17 and bile ducts5,16 are the predominant reported sites of origin.
Although uncommon in domestic animals, RCCs are the most common primary renal tumors in horses. By the time RCC incites clinical disease, the neoplasm is already in advanced stages, with disseminated metastases expected in ~70% of patients. 9 Animals often have hematuria and, on autopsy, have a kidney locally effaced by an aggressive neoplasm with perirenal infiltration.18,19 Lung and liver are the most consistent metastatic sites, but involvement of other organs is not uncommon, including but not limited to spleen, lymph nodes, contralateral kidney, myocardium, stomach, bones, pancreas, diaphragm, and omentum. 19 In horses, immunoreactivity for pancytokeratin, and not CK7, has been shown. 19 Pax8 is a transcription factor expressed in epithelia of renal tubules, thyroid follicles, and Müllerian tract. This immunomarker, used to identify neoplasms arising from these organs, is already validated in humans and, to a limited extent, in small companion animals. 10 In our case, the involvement of the retroperitoneal fat and associated lymph nodes, but not the renal parenchyma, makes this diagnosis unlikely. Pax8 IHC was partially contributory given that only medullary renal tubules had nuclear expression and, consequently, RCC arising from this segment could be ruled out. Absent Pax8 expression by neoplastic cells but strong reactivity in the normal equine endometrium, which can rarely give rise to carcinoma with widespread serosal implantation in mares, 4 makes endometrium an improbable primary site.
Given the multicentric hepatic nodules in this horse, cholangiocellular carcinoma was suspected, but differentiation from metastatic adenocarcinomas can be a diagnostic challenge. Identifying larger masses in other organs with epithelial tissue should favor a pattern of metastatic spread to the liver, which was not apparent in this mare. In horses, cholangiocellular carcinomas are rare, and the liver is most commonly affected by spread from other cancers. 5 In a single report of cholangiocarcinoma with extensive peritoneal carcinomatosis, 20 recognizing a solitary principal mass effacing the hepatic parenchyma was crucial for the diagnosis, dismissing ancillary IHCs. Ductal epithelium should express CK7, which, in conjunction with other markers, has been used to diagnose cholangiocellular carcinomas in veterinary medicine; however, labeling is not exclusive to this neoplasm given that other metastatic tumors can express it. 13 Even though this marker’s successful use in horses was reported elsewhere, 1 the antibody and protocol applied in our case (optimized and validated for dogs) did not have interspecies cross-reactivity. Other institutions that offer CK7 IHC for domestic animals were contacted in hopes of new insights; however, lack of immunoreactivity by equine bile ducts was confirmed (Miller AD, Uaje M, pers. comm., 2020 Nov 30), ruling out potential issues with prolonged formalin fixation. Variations in the amino acid sequence of certain antigens are thought to play a role in discrepancies in interspecies reactivity. 13
The most noteworthy pulmonary neoplasm in horses is the granular cell tumor, given its greater frequency than pulmonary carcinomas. However, the latter was an important differential in our case, given the respiratory distress and microscopic pattern. Few equine cases of pulmonary carcinoma are described in the literature,2,6,7 and none report widespread peritoneal spread beyond renal involvement. 6 In one instance, 7 neoplastic cells lacked expression for TTF-1, similar to our case, which warranted IHC for napsin A, a proteinase expressed by type II pneumocytes and some canine lung epithelial neoplasms. Even though TTF-1 results were unsatisfactory in our patient’s lung samples, the negative expression of napsin A by neoplastic cells makes a primary pulmonary carcinoma unlikely.
The similar-appearing multicentric nodules, both macro- and microscopically, in multiple viscera, and lack of a salient large primary mass in this mare recapitulated CUP. In human oncology, CUP corresponds to a distinctive subset of widespread metastatic disease without a discernible site of origin following comprehensive clinical and diagnostic investigation, covered in standardized guidelines shared by medical communities. 3 Even after postmortem examination, 30–70% of cases remain undefined in humans. When found, most are diminutive and of pulmonary or pancreatic origin. 11 In addition, peculiar seeding to targeted and unusual organs, such as lung, brain, and bone, and unique biological properties also warrant CUP to be classified as a specific, yet enigmatic, cancer entity. CUP is an obscure process that fosters multiple theories because the traditional linear, stepwise cancer progression is not met; genetic and protein profiling is of great research interest. 8 Proposed explanations, such as regression of the primary tumor with an expansion of metastases with a different biologic profile, or disseminated cancer cells (DCC) with stemness properties circulating in the bloodstream during early tumorigenesis, are still elusive and under validation.
Systematic IHC algorithms are pivotal in human CUP because some subsets of cancers, depending on their tissue of origin, are eligible for targeted therapeutic choices and may have a more favorable outcome. 15 However, diagnostic guidance often has significant time and budgetary impact. Diagnostic workflows for undetermined carcinomas begin with the CK7/CK20 expression profile by neoplastic cells followed by various complementary organ-specific antibodies to better define probable primary sites.13,15 However, even with such established validated protocols in human tissues, pitfalls still exist, such as intra-tumor heterogeneity and anomalous or unexpected antigen expression in undifferentiated phenotypes or at metastatic sites. 15
Veterinary literature on CUP is still rudimentary and limited to small companion animals.14,16 In a review of canine cases, 14 the IHC algorithm was very limited compared to human studies, and undifferentiated carcinomas predominated. In canine patients with metastatic CUP, the median survival time was 30 d, which also restrains antemortem testing, and no primary site was determined in the only animal submitted for postmortem examination. 14
To our knowledge, CUP studies in horses are still elusive to nonexistent. Performing any stepwise exclusion approach in equine species is even more challenging given the patient’s size, which greatly limits the use of diagnostic imaging. Also, clinical signs generally become evident at already advanced stages of the disease. Apart from logistic and financial constraints, validation of interspecies antibody cross-reactivity, as witnessed in our case, remains an important obstacle to the promotion of CUP study in domestic animals. Our attempts to extrapolate some of the existing IHC protocols to equine tissue had informative, although somewhat disappointing, results as summarized. For instance, CK7 did not cross-react with normal equine biliary duct epithelium, which hinders the first step of the carcinomatous CUP diagnostic algorithm. Of all the markers used in our case, pancytokeratin, vimentin, napsin A, and Pax8 were useful for identifying equine epithelial, mesenchymal, alveolar, renal medullary tubules, and endometrial glandular cells, respectively. Our results indicate that TTF-1 IHC has some potential on horse lung but still needs technical refinement given that rare bronchiolar epithelial cells had faint equivocal nuclear expression. Thus, although the veterinary diagnostic community has come a long way, there is still room for improvement. Postmortem evaluation of veterinary patients with provisional CUP followed by an IHC panel, with continuous work on interspecies protocol validation whenever possible, is encouraged in hopes of investigating whether this entity can be applied to other domestic animals and how it parallels the human counterpart.
Footnotes
Acknowledgements
We thank Dr. Ramos-Vara (Purdue University) and Dr. Miller (Cornell University) for providing consultation and assistance with cytokeratin 7 on equine tissues, and Lisa Parsons for technical assistance and slide preparation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
