Abstract
A 23-y-old gelding was presented to a veterinary teaching hospital with a history of chronic, refractory diarrhea. Clinically, the horse was in poor body condition, with a thickened and corrugated large intestine identified by transcutaneous abdominal ultrasonography. At postmortem examination following euthanasia, the large colon and cecum had segmental thickening of the intestinal wall with innumerable mucosal ulcers and prominent polypoid mucosal masses. Many mesenteric and hepatic lymph nodes were enlarged. Histology revealed granulomatous and ulcerative typhlocolitis and granulomatous lymphadenitis with myriad acid-fast, variably gram-positive, intrahistiocytic bacilli that stained by immunohistochemistry for mycobacteria. Molecular testing by PCR and sequencing identified the causative agent as Mycobacterium genavense, which is an unusual presentation of infection in a horse.
Mycobacterium genavense is a ubiquitous, nontuberculous, acid-fast mycobacterium with zoonotic potential. The earliest reported infections in animals emerged in the 1990s when cultured from several pet birds. 11 At that time, M. genavense was considered a novel pathogen; it has been increasingly detected in immunosuppressed humans. 10 Infection in a variety of other species has been reported since, including a so-litary case in a horse with a disseminated, granulomatous presentation. 3 Horses are considered naturally resistant to infection with mycobacterial disease, and diagnosis of Mycobacterium tuberculosis complex or atypical infection occurs infrequently. 14 Herein we report a case of M. genavense in an adult horse with a primary gastrointestinal manifestation.
A 23-y-old Quarter Horse gelding was presented to a veterinary teaching hospital with a 1-mo history of diarrhea and inappetence, as well as episodic weight loss during the winter months over a 3-y period. Prior treatment and management by the referring veterinarian included administration of ranitidine, omeprazole paste (Gastrogard; Boehringer Ingelheim), and di-tri-octahedral smectite (Bio-Sponge; Platinum Performance), with minimal improvement reported. Upon admission, the horse was in thin body condition and had liquid diarrhea.
Transcutaneous abdominal ultrasound examination reve-aled diffuse thickening and corrugation of the large intestine up to 2 cm thick, with appropriate motility and no other significant findings. Transrectal palpation of the caudal abdomen was unremarkable. Abdominal radiographic examination revealed minor sand accumulation in the large colon. Peritoneal fluid collected by abdominocentesis was cytologically normal. The fluid was straw-colored with a nucleated cell concentration of 1,490 cells/µL (RI: < 2,000 cells/µL) and a total solids concentration of 7 g/L (RI: < 25 g/L) consistent with a protein-poor transudate. Antemortem fecal testing detected no intestinal parasites by either McMaster fecal egg count or Sheather fecal flotation. The CBC revealed mild leukopenia (5.4 × 109/L; RI: 6.0–12.0 × 109/L) characterized by lymphopenia (0.6 × 109/L; RI: 1.5–5.0 × 109/L) and pseudothrombocytopenia (40 × 109/L; RI: 100–230 × 109/L) with platelet clumping. Increased plasma fibrinogen concentration (6 g/L; RI: 1–4 g/L) and hypoferremia (9.1 µmol/L; RI: 23.1–31.0 µmol/L) were consistent with systemic inflammation. Abnormalities on serum biochemistry analysis were presumptively attributed to enteropathy of unknown etiology and included hypoproteinemia (59.8 g/L; RI: 60.0–86.0 g/L) characterized by hypoalbuminemia (17.2 g/L; RI: 27.0–41.0 g/L), hyponatremia (130 mmol/L; RI: 134–150 mmol/L), hypomagnesemia (0.66 mmol/L; RI: 0.70–0.86 mmol/L), mildly decreased bicarbonate concentration (20.5 mmol/L; RI: 21.0–31.0 mmol/L), and decreased calculated osmolality (262 mmol/kg; RI: 276–288 mmol/kg). Given the chronic nature of the clinical signs and the abnormal findings noted on transcutaneous abdominal ultrasonography, the owners elected to stop further clinical assessment and not to pursue therapeutic management. The gelding was euthanized and submitted for postmortem examination.
The most significant postmortem findings were in the large colon and cecum, which comprised 15–20, semi-firm, tan, polypoid proliferations extending into the lumen from the mucosal surface. Circumferentially, affected segments of the intestinal wall, predominantly involving the mucosa and submucosa, were severely thickened by edema and contained innumerable crateriform ulcers (Fig. 1A). Covering the serosa of the large colon and cecum were 0.3–2-cm, irregular and anastomosing, slightly raised, black plaques grossly resembling hemomelasma ilei. The small intestine and small colon were relatively unaffected. Hepatic and mesenteric lymph nodes (LNs) were subjectively enlarged. Tissue samples from the lesions were fixed in 10% neutral-buffered formalin, processed routinely, and stained with H&E for histologic examination.
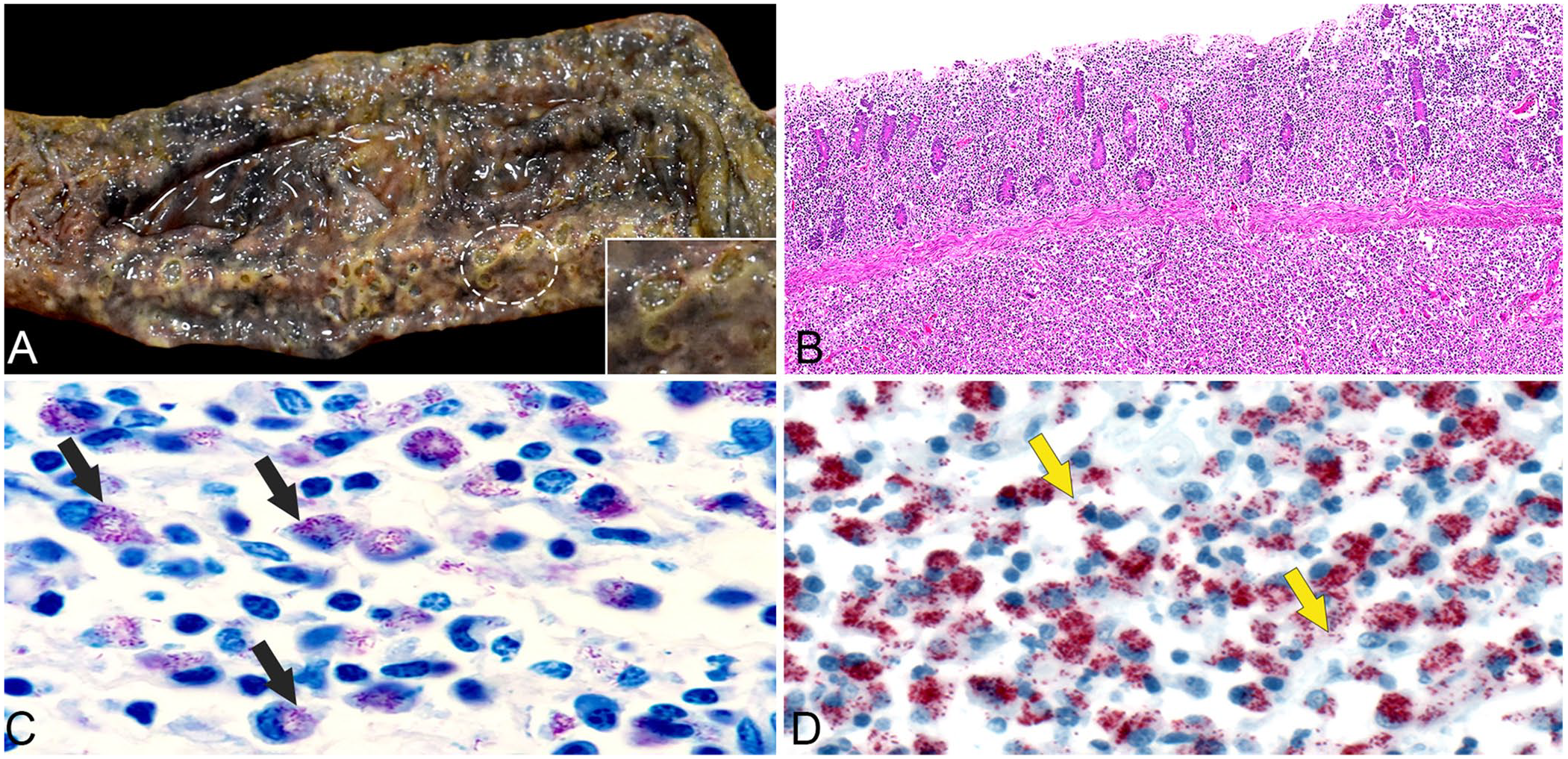
Ulcerative and granulomatous typhlocolitis caused by Mycobacterium genavense in an adult Quarter Horse gelding.
Initial postmortem ancillary tests included fecal culture from a large colon sample that yielded Salmonella species group b1 from growth medium broth only; there was no significant bacterial growth from the small intestine. An additional postmortem Sheather fecal flotation test did not recover any parasites.
Histologically, affected large colon and cecum had ulceration of the mucosa, interspersed with hyperplastic epithelial foci corresponding to the gross polypoid lesions. In segmental regions of the circumferentially thickened large intestine, the lamina propria and submucosa were expanded up to 1-cm thick by granulomatous inflammation comprised of dense sheets of macrophages, mixed with a few lymphocytes, plasma cells, neutrophils, and rare scattered necrotic cells (Fig. 1B). Similar granulomatous inflammation expanded the serosa, forming multifocal plaque-like areas, and filling the sinuses of the hepatic and mesenteric LNs and ectatic subserosal lymphatic vessels. The liver had porto-portal bridging fibrosis and foci of lymphohistiocytic inflammation mixed with a few neutrophils in the parenchyma and periportal areas. Abundant 1–2-μm, acid-fast (Fig. 1C), variably gram-positive, non-argyrophilic (by Steiner stain) bacilli were observed in the cytoplasm of macrophages within the granulomatous inflammation in the colon, cecum, ectatic subserosal lymphatic vessels, and hepatic and mesenteric LNs. A few bacilli were seen in inflammatory cells in the liver.
Given the acid-fast bacilli in macrophages of the intestinal tract, differential diagnoses of Mycobacterium spp. and Rhodococcus equi were considered. Formalin-fixed tissues from the autopsy were submitted to the Infectious Diseases Pathology Branch (IDPB) of the Centers for Disease Control and Prevention (CDC; Atlanta, GA, USA) for further evaluation and testing. An immunohistochemical assay for Mycobacterium spp. was performed using a protocol described previously. 2 Abundant bacilliform and granular mycobacterial antigens were detected within macrophages in the large colon and cecum (Fig. 1D), ectatic subserosal lymphatic vessels, and mesenteric and hepatic LNs. Rare staining was also seen within inflammatory cells in the liver.
DNA was extracted from formalin-fixed, paraffin-embedded (FFPE) tissue samples (QIAamp ultraclean production pathogen mini kit; Qiagen). A Mycobacterium genus–specific nested PCR assay targeting the 16S rRNA gene 1 (primer sequences unpublished) was performed. Positive amplicons of 214 bp and 178 bp were identified by gel electrophoresis, extracted from the gel, and directly sequenced by Sanger sequencing. BLAST sequence analysis (https://blast.ncbi.nlm.nih.gov/Blast.cgi) showed 100% identity to M. genavense.
Based on the pathologic and molecular findings, diagnoses of mycobacterial typhlocolitis, lymphadenitis, and hepatitis were established for this gelding. To our knowledge, this is a unique case of M. genavense typhlocolitis in an adult horse. M. genavense is an atypical mycobacterium and is considered a predominant mycobacterial etiologic agent in psittacine pet birds. 15 Natural infections have been documented in diverse species, including humans, dogs, cats, snakes, ferrets, guinea pigs, birds, and a horse.3,6,7,10–13 Most of these cases had disseminated granulomatous inflammation with systemic infection.
M. genavense rarely causes disease in immunocompetent humans and is generally an opportunistic pathogen associated with an immunocompromised status, such as those with human immunodeficiency viral infection, organ transplant, chronic steroid usage, and other immunodeficiency syndromes. 10 A case of disseminated infection with M. genavense was reported in a cat with feline immunodeficiency virus infection, 12 further illustrating the importance of immunodeficiency as a predisposing factor for disseminated disease.
In both humans and birds, the organism has been isolated from the intestinal tract of healthy individuals. Information regarding which species act as a reservoir source is lacking. 6 This is partially because the gold standard for detection often requires weeks of mycobacterial culture given its slow growth, which is a challenge, especially antemortem. 4
Detection of M. genavense has been detailed in a 16-mo-old Thoroughbred filly, with granulomatous dissemination to the spleen, lung and pleura, adventitia of the aorta, and the aortic, inguinal, and mandibular LNs. 3 Notably, granulomatous lesions were not observed in the intestinal tract. Compared to other typically documented systemic infections, the filly had a paucity of bacilli histologically. 3 Analogous to our case, the animal failed to thrive, resulting in elective euthanasia. Molecular testing was equally required to reach a final diagnosis; however, unlike that case, 3 our case had an abundance of bacilli noted predominantly in the gastrointestinal tract. Before PCR testing, differential diagnoses in our case included paratuberculosis caused by M. avium subsp. paratuberculosis, Rhodococcus equi, and Salmonella infection. A clinically relevant Salmonella infection was considered least likely, given that disease progression is generally acute in salmonellosis, and no gram-negative rods were observed microscopically.
The precise timing of onset of disease in our case is difficult to pinpoint, considering the reported episodic 3-y history of weight loss during the winter months. Additionally, the immunocompetence of the gelding was not assessed during the short time from examination to euthanasia; however, some degree of immune system debilitation was clinically considered due to advancing age and the possibility of other comorbidities, such as pituitary pars intermedia dysfunction (PPID). The pituitary gland was not evaluated histologically; however, its size and parenchymal cross-sections were grossly unremarkable on postmortem examination. In addition, other signs of PPID, such as hypertrichosis, muscle wasting, and laminitis, were absent. Similarly, in the equine case report of disseminated infection, 3 evidence of immune suppression was not identified.
A definitive environmental source could not be determined, which is common with nontuberculous mycobacterial infections, given their ubiquity in the environment. Possible epidemiologic origins considered included: other animals on the property, feces of wild birds in the pasture, and water sources. 9 It is important to note that exposure to clinically affected animals is not a prerequisite for disease, further confounding identification of the source of infection.
The likely route of infection was ingestion of the bacteria, with proliferation in the intestine. The organisms then spread through infiltration of the lymphatic vessels to the mesenteric LNs within macrophages. The mechanisms of localization to the hepatic LNs are hypothesized to result from ascending infiltration of bacteria from the intestinal tract, through the portal tract, and then to the hepatic LNs. This method of dissemination is similar to the spread of M. avium in pigs with hepatic lesions 8 and M. avium subsp. paratuberculosis in which the liver and lymph nodes are common secondary sites with advancing disease.5,16
Many features in our case paralleled M. avium subsp. paratuberculosis infection in cattle, including the chronic history of weight loss and diarrhea, failure to thrive, age, and gross thickening of the intestine that histologically contained sheets of macrophages with intracytoplasmic acid-fast bacilli.5,16 The polypoid masses within the intestine that were formed from mucosal epithelial hyperplasia and were rather devoid of inflammation are not a described feature of mycobacterial infection and the significance of this finding is unknown. Typically, M. avium complex species members are responsible for most of the reported mycobacterial infections in horses. Granulomatous tuberculoid lesions predominate and less prevalent lesions comprise, but are not limited to, the LNs and rarely the intestine. 10 PCR assay, followed by sequencing of amplicons, is requisite in differentiation of M. avium from M. genavense, although prognostic significance between species has yet to be elucidated in horses.
Ours is the second report of M. genavense in horses, and the first with primarily intestinal manifestations, to our knowledge. We retrieved no cases of gastrointestinal lesions subsequent to M. genavense infection in horses in a search of Google, PubMed, CAB Direct, Web of Science, and Scopus using the search terms “M. genavense, gastrointestinal, veterinary pathology, and horse.” This suggests that a primary intestinal manifestation has not been reported before in horses.
Many factors contribute to the difficulty in detection of this causative agent. It is possible that the disease is more prevalent in horses, but not commonly recognized because of lack of specialized testing (e.g., mycobacterial culture). M. genavense should be considered as a differential diagnosis in horses with chronic diarrhea and gross findings of ulcerative typhlocolitis. Furthermore, our case highlights a potential zoonotic One Health concern for immunocompromised animals and humans. However, current data suggest that environmental contamination poses a higher threat for exposure because horizontal transmission has not been proven experimentally. 3
Footnotes
Acknowledgements
We thank Dr. Sherif Zaki; the immunohistochemical team at IDPD for supporting immunohistochemistry; Dr. Julu Bhatnagar for supporting PCR testing; Dr. Jana Ritter for input provided in the manuscript; and the histopathology laboratory at Auburn University for slide preparation.
Previous presentation or publication disclosure: Alana J. Kramer, Shune Kimura, Rachel Neto. Mycobacterium genavense typhlocolitis in a horse. Case presented at: Northeastern Veterinary Pathology Conference. 2021 Sept 17–18. Virtual meeting.
Our findings and conclusions are those of the authors and do not necessarily represent the official position of the U.S. Centers for Disease Control and Prevention (CDC)
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
