Abstract
Avian influenza A(H5) viruses (avian IAVs) pose a major threat to the economy and public health. We developed an antigen-ELISA (ag-ELISA) and a colloidal gold–based immunochromatographic strip for the rapid detection of avian A(H5) viruses. Both detection methods displayed no cross-reactivity with other viruses (e.g., other avian IAVs, infectious bursal disease virus, Newcastle disease virus, infectious bronchitis virus, avian paramyxovirus). The ag-ELISA was sensitive down to 0.5 hemagglutinin (HA) units/100 µL of avian A(H5) viruses and 7.5 ng/mL of purified H5 HA proteins. The immunochromatographic strip was sensitive down to 1 HA unit/100 µL of avian A(H5) viruses. Both detection methods exhibited good reproducibility with CVs < 10%. For 200 random poultry samples, the sensitivity and specificity of the ag-ELISA were 92.6% and 98.8%, respectively, and for test strips were 88.9% and 98.3%, respectively. Both detection methods displayed high specificity, sensitivity, and stability, making them suitable for rapid detection and field investigation of avian A(H5) viruses.
Avian influenza A(H5) viruses (avian IAVs; Orthomyxoviridae, Alphainfluenzavirus) have caused worldwide alarm given their high pathogenic potential. 6 In 1997, the highly pathogenic avian influenza A(H5N1) virus (HPAIV) that infected humans from birds was first detected in Hong Kong. 1 The A(H5N1) subtype appeared again in East and Southeast Asia in 2003 and 2004. 14 Since September 2019, the World Health Organization (WHO) has reported 861 human cases of A(H5N1) infection and 455 deaths from 16 countries. 5
Since being found in a domestic duck in Guangdong Province in 1996, the HPAIV H5N1 subtype has evolved into 10 different gene groups (0−9) and multiple subclasses. 11 Clade 2.3.4.4 appeared in China in 2010–2011. 17 HPAIV H5N2, H5N5, and H5N8 subtypes have been identified in duck and live poultry markets in China. 11 The virus is widespread in wild birds, is spread around the world through their migration, and has been detected in Asia, Europe, and North America. 11 The H5N6 subtype has infected at least 19 people and caused 13 deaths in China and Southeast Asia. 3 From 2014 to 2015, the United States experienced the worst HPAI incident, with nearly 50 million birds slaughtered. 2 In addition, since 2014, clade 2.3.4.4 H5 avian IAVs has been found in Korea, Germany, the United Kingdom, Egypt, and the Netherlands, resulting in economic losses to local poultry industries. 11 The HPAIV H5 subtype is still circulating in avian species, and represents a major threat to humans. Therefore, it is important to develop methods for rapid detection and continuous surveillance of this virus subtype.
Classic IAV detection relies on identifying hemagglutinin (HA) and neuraminidase (NA) proteins by antigen detection after virus isolation. 10 Although molecular detection methods and real-time PCR (rtPCR) have been used widely in laboratory detection of IAV infection, these methods are technically demanding. 20 Thus, less expensive and more convenient detection methods for detection of IAVs are needed, such as antigen-ELISA (ag-ELISA) and colloidal gold–based immunochromatographic strip approaches. Ag-ELISA can be used to evaluate A(H1N1) vaccine efficacy and detect infections; an immunochromatographic assay has been developed to detect A(H7N9) viruses with good specificity and sensitivity for clinical samples from patients, poultry, and the environment.9,16,18 We developed ag-ELISA and immunochromatographic strip methods based on monoclonal antibodies (mAbs) for the detection of avian A(H5) viruses to assist disease prevention and help control epidemics.
We acquired avian IAVs and other viruses, including infectious bursal disease virus (IBDV), Newcastle disease virus (NDV; Avian orthoavulavirus 1), infectious bronchitis virus (IBV; Avian coronavirus), and avian paramyxovirus (APMV) from our laboratory repositories (Suppl. Table 1). All virus concentrations were determined using HA titration and 50% tissue culture infectious dose (TCID50) assays according to the Reed–Muench method. 15
Mouse myeloma cell line SP2/0 and Madin–Darby canine kidney (MDCK) cells were preserved in our laboratory. Purified HA proteins from A(H5N6) virus (A/Duck/Guangdong/GD01/2014) were purchased from Sino Biological (China). The animal experiment was approved by the First Affiliated Hospital, School of Medicine, Zhejiang University (2017-015). BALB/c mice were immunized twice intramuscularly at intervals of 2 wk with purified HA protein and Freund adjuvant (Millipore Sigma). Mice were boosted with the same HA protein 3 d before the fusion. 19 Hybridoma culture supernatants were screened by ELISA. Positive monoclonal hybridoma cells were obtained after 3 limiting dilutions and then injected intraperitoneally into mice. Mouse ascites fluids were collected and purified using a protein G column (GE Healthcare) to obtain mAbs.
The isotypes of mAbs were determined (mouse monoclonal antibody isotyping kit; Bio-Rad) according to the manufacturer’s instructions. The affinities of mAbs were evaluated by ELISA. 19 Briefly, a 96-well plate was coated with purified HA protein (20 ng per well). Monoclonal antibodies at a starting concentration of 1 mg/mL were 2-fold serially diluted and incubated for 1 h. Goat anti-mouse IgG (Novus) diluted to 100 ng/mL was added to the plate and incubated for 30 min. The color was developed with 3,3′,5,5′-tetramethylbenzidine (TMB; KPL) substrate solution and stopped with TMB stop solution. Between steps, the plate was washed with phosphate-buffered saline containing 0.1% Tween 20 (PBST, pH 7.2). The optical density (OD) was measured at 450 nm (iMARK microplate reader; Bio-Rad), and the affinity was estimated as the minimum concentration of mAb required for a positive reaction. The heavy- and light-chain genes of the hybridomas cells were sequenced by Sino Biological (China).
Six murine mAbs (1A2, 4E2, 2C8, 3B7, 1B1, 2F11) were screened for efficient detection of different A(H5) strains. Monoclonal antibodies 4E2, 2C8, 3B7, 1B1, and 2F11 belong to the IgG2a subclass, and mAb 1A2 belongs to the IgG1 subclass. Monoclonal antibodies 4E2 and 2F11 were chosen for further studies given their high affinity (Table 1); mAbs 4E2 and 2F11 reacted with all H5 subtype viruses from different clades (Suppl. Table 1). Therefore, mAbs 4E2 and 2F11 were complementary and likely to be suitable for rapid detection of avian A(H5) virus.
Characterization of the affinity of 5 monoclonal antibodies (mAbs) against avian influenza A(H5) viruses.
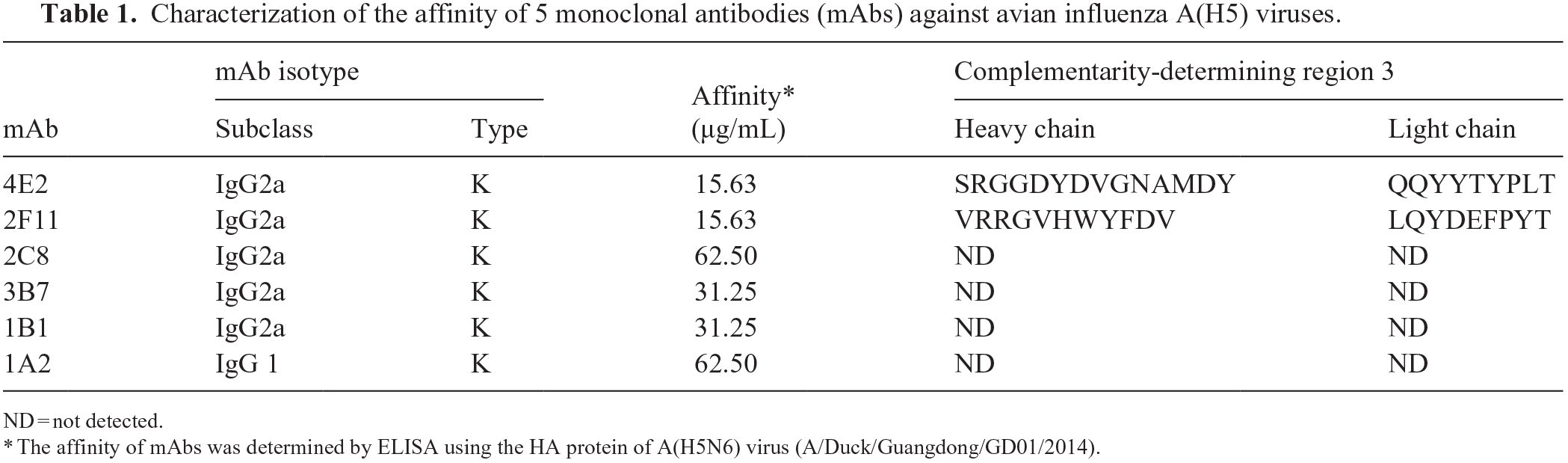
ND = not detected.
The affinity of mAbs was determined by ELISA using the HA protein of A(H5N6) virus (A/Duck/Guangdong/GD01/2014).
The immunofluorescence assay (IFA) was performed to measure binding of mAbs to A(H5) virus–infected cells. 19 Confluent monolayers of MDCK cells were infected with IAV at a multiplicity of infection of 0.5. After 24 h, cells were fixed with 4% paraformaldehyde at room temperature and permeabilized with 0.5% Triton X-100. Cells were incubated with mAb 4E2 or 2F11, then a goat anti-mouse IgG heavy plus light-chain (H + L)-Alexa Fluor (Abcam) was added. The results were photographed by fluorescence microscopy (Olympus).
The ag-ELISA (Suppl. Fig. 1) was performed as described previously.4,16 Monoclonal antibody 4E2 was selected and used to coat a 96-well plate at 160 ng/well in 100 µL of coating buffer. Monoclonal antibody 2F11 was picked for detection and labeled with horseradish peroxidase (HRP; Innoreagents). Plates were washed 5 times with PBST, antibody-coated plates were blocked with 5% bovine serum albumin (BSA; 200 µL/well) for 2 h, plates were washed another 5 times, and samples were added and incubated for 1 h at 37°C. After washing, 2F11-mAb-HRP (4 µg/mL) was incubated for 30 min. After washing 5 times, activated TMB substrate solution was added at 100 µL/well. The reaction was stopped by adding 100 mL of TMB stop solution to each well, and the OD450 nm was read (iMARK microplate reader). An OD > 2.1-fold the negative control was considered positive.
Colloidal gold solution was prepared as described previously.9,13 Briefly, 100 mL of 0.01% gold chloride was heated to boiling, and 3.6 mL of 1% trisodium citrate solution was added with continuous gentle stirring. The colloidal gold solution was continuously boiled until the color of the mixture changed to a brilliant wine red. The pH was adjusted to 7.2 using 1% potassium carbonate. Monoclonal antibody 4E2 (100 µL, 1 mg/mL) was added to 10 mL of colloidal gold solution and incubated for 20 min. After blocking with BSA, the solution was centrifuged for 30 min, the colorless supernatant was removed, and the loose sediment was resuspended with 1 mL of PBST (containing 1% BSA and 0.1% Tween 20).
The immunochromatographic strip was composed of a sample pad, a mAb-gold conjugated pad, nitrocellulose membranes, and an absorbent pad (Suppl. Fig. 1). Nitrocellulose membranes were coated with 1.0 mg/mL mAb 2F11 on the test line and 1.0 mg/mL goat anti-mouse IgG (Solarbio) on the control line.
The specificity of the assays was tested with different subtype avian IAVs (including 45 A(H5) viruses, 21 other IAVs), and other viruses. Two-fold serial dilutions of A(H5) virus allantoic fluids and purified H5 protein were used to determine test sensitivity. Avian A(H7N3) allantoic fluid was used as the negative control. To evaluate reproducibility, each sample in a single assay was tested in triplicate, and each assay was repeated at least 3 times.
To further evaluate the clinical application of our assays, oropharyngeal and cloacal swabs were collected from 100 chickens in Zhejiang Province and were tested with the ag-ELISA, immunochromatographic strip, and multiplex reverse-transcription rtPCR (RT-rtPCR) methods. The clinical samples were collected in tubes containing 1 mL of PBS containing 0.5% BSA, penicillin G, and streptomycin sulfate. Then the samples were briefly vortexed, centrifuged at 1,800 × g for 30 min, and the supernatant collected for testing. The RT-rtPCR assay (Influenza A virus real time RT-PCR kit, avian influenza virus H5 subtype nucleic acid detection kit, fluorescence PCR method; Liferiver) was performed.
Neither mAb 4E2 nor 2F11 exhibited nonspecific binding to MDCK cells infected with avian IAV H9N2 or H7N3 subtypes; however, both mAbs showed strong reactivity toward A(H5) virus–infected cells in an IFA (Suppl. Fig. 2). For both the ag-ELISA and the immunochromatographic strip methods, no cross-reactivity was observed for any of the non-A(H5) viruses consisting of H1, H2, H3, H4, H6, H7, H9, H10, and H11 subtypes; IBDV, NDV, IBV, and APMV were also negative (Suppl. Table 1; Fig. 1A). The ag-ELISA limit of detection (LOD) was 100 µL of sample containing 0.5 HA units of IAV (A/duck/Zhejiang/6D2/2013 [H5N6] and A/duck/Zhejiang/W24/2013 [H5N8]); the LOD was 1 HA unit for A/goose/Zhejiang/97/2014 (H5N1) and 2 HA units for A/duck/Zhejiang/6DK19/2013 (H5N2). Our ag-ELISA could detect as little as 7.5 ng/mL of purified H5 HA protein (Suppl. Table 1; Fig. 1B, 1C). The coefficients of variation (CVs) for intra- and inter-batch reproducibility of our ag-ELISA were < 10% (Suppl. Tables 2, 3), which indicates that the method had good reproducibility.
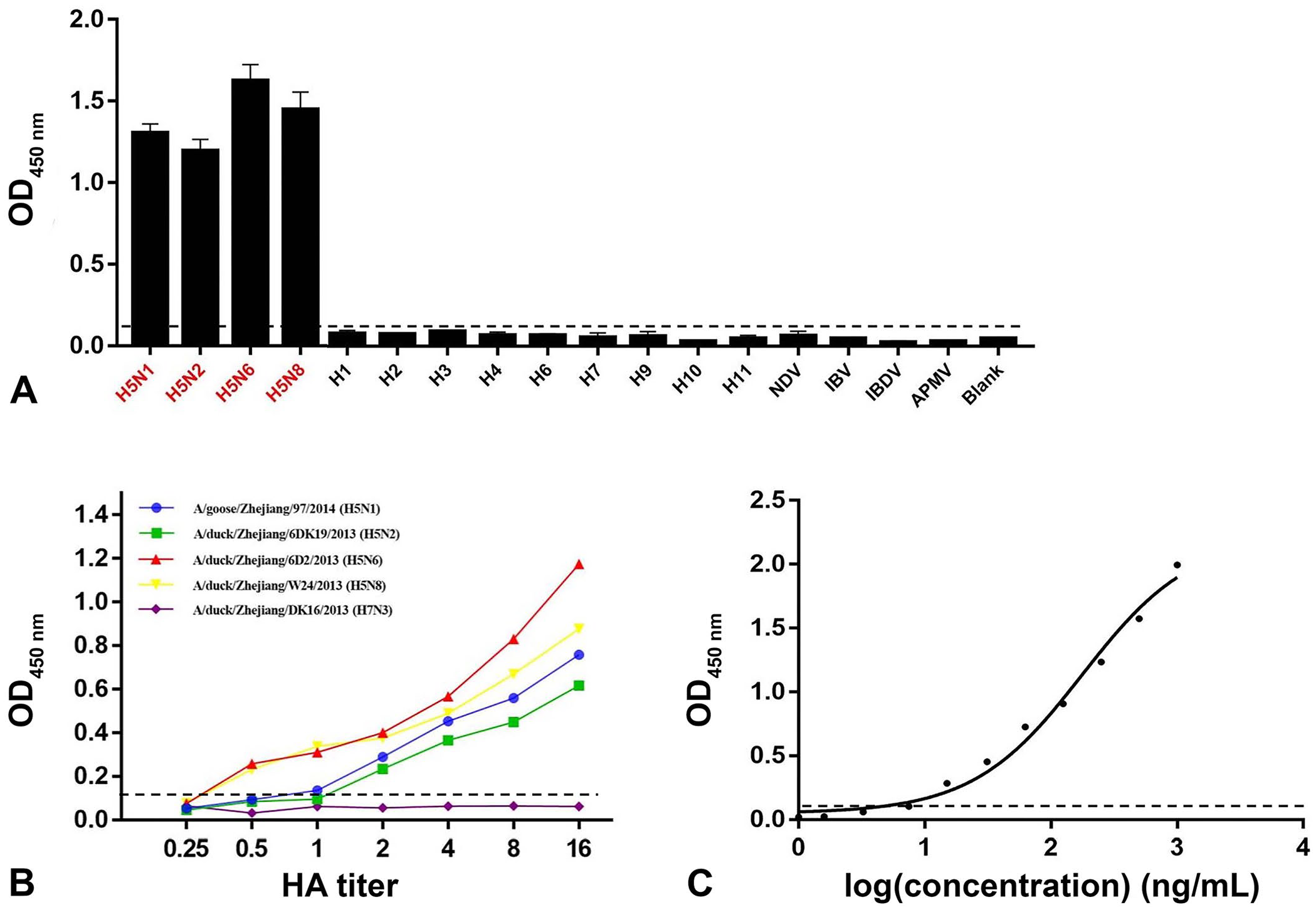
Specificity and sensitivity of the antigen-ELISA (ag-ELISA) for the detection of avian influenza A(H5) virus (avian IAV).
Positive results were observed for immunochromatographic strips tested with avian A(H5) viruses but no other viruses (Fig. 2A). This result suggests that strips could be used to specifically detect avian A(H5) viruses (Suppl. Table 1). Two-fold serially diluted allantoic fluid from avian A(H5N6) virus (A/duck/Zhejiang/6D2/2013) was used to test the sensitivity of the immunochromatographic strips. The detection limit of the culture method was 100 µL of allantoic fluid containing 1 HA unit of virus or a TCID50 of 101.79 (Fig. 2B).

Specificity of the immunochromatographic strip method for the detection of avian influenza A(H5) virus.
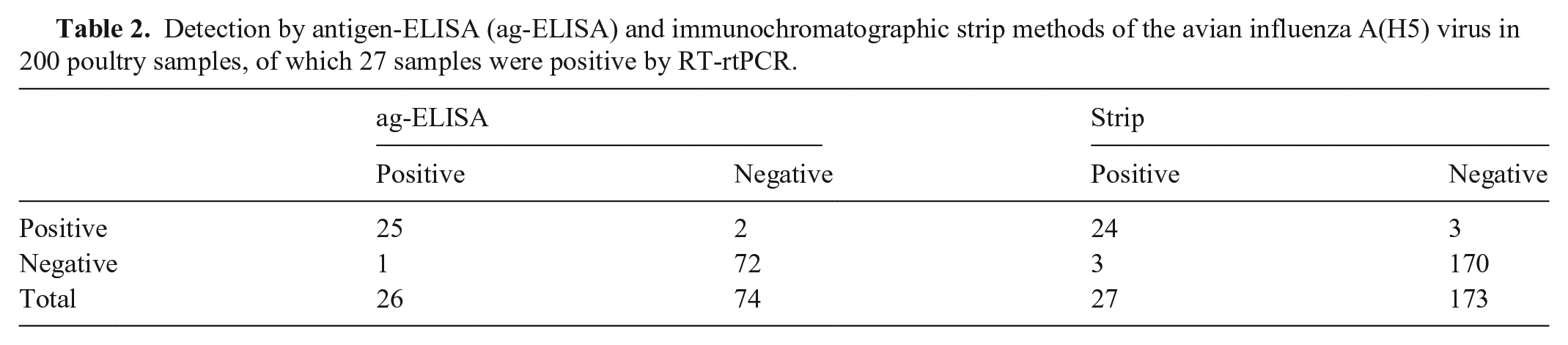
To verify stability, immunochromatographic strips were tested after being assembled for 30 and 60 d; specificity and sensitivity were comparable to newly assembled test strips. We used the results of a RT-rtPCR as our standard, and 27 of the 200 field samples were determined to be positive by using RT-rtPCR; in the ag-ELISA, 25 of the 200 poultry samples were positive for avian A(H5) viruses, and in the strip assay, 24 of 200 poultry samples were positive (Table 2). Furthermore, the positive samples identified by RT-rtPCR and ag-ELISA were the same specimens. The sensitivity and specificity of the ag-ELISA was 92.6% (25 of 27) and 98.8% (172 of 174), respectively. The sensitivity and specificity of the test strips was 88.9% (24 of 27) and 98.3% (170 of 173), respectively.
Detection by antigen-ELISA (ag-ELISA) and immunochromatographic strip methods of the avian influenza A(H5) virus in 200 poultry samples, of which 27 samples were positive by RT-rtPCR.
Monoclonal antibodies 4E2 and 2F11 did not show HI and virus neutralization activities, they also reacted specifically with HA antigens. This indicates that they might bind to a linear epitope on the HA antigen, which also meets the detection requirements. 8 Therefore, we used these mAbs to establish the H5 subtype detection method employed in the ag-ELISA and immunochromatographic strips. In addition, it is expected that these 2 mAbs may also be used to develop other immunologic methods to detect A(H5) virus, such as dot blot and in situ detection.
In our testing of poultry samples, the ag-ELISA achieved slightly higher specificity and sensitivity than the immunochromatographic strips. The immunochromatographic strip is read by the naked eye, which may result in different results for different observers. A mAb-based dot ELISA for universal detection of avian A(H5) virus, which was aimed mainly at clade 2.1 and a small number of other clades, has been reported. 7 An immunochromatographic strip has been developed with a minimal detection limit (EID50) of 104.2 to 106.5 per test. 12 Our methods were mainly used to detect clade 2.3.4.4, and both methods achieved acceptable specificity and sensitivity. Although PCR is still the best method for detecting viral infections, the ag-ELISA method and immunochromatographic test strip method are both suitable for large-scale screening in resource-limited environments.
Supplemental Material
sj-pdf-1-jvd-10.1177_10406387211027538 – Supplemental material for Development of an antigen-ELISA and a colloidal gold–based immunochromatographic strip based on monoclonal antibodies for detection of avian influenza A(H5) viruses
Supplemental material, sj-pdf-1-jvd-10.1177_10406387211027538 for Development of an antigen-ELISA and a colloidal gold–based immunochromatographic strip based on monoclonal antibodies for detection of avian influenza A(H5) viruses by Yixin Xiao, Fan Yang, Fumin Liu, Linfang Cheng, Hangping Yao, Nanping Wu and Haibo Wu in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our study was funded by grants from the National Science and Technology Major Project for the Control and Prevention of Major Infectious Diseases in China (2018ZX10711001, 2018ZX10102001, 2020ZX10101016-004-002), Zhejiang Provincial Natural Science Foundation of China (LY19H260006), and the Independent Task of State Key Laboratory for Diagnosis and Treatment of Infectious Diseases (2020ZZ17).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
