Abstract
An immunochromatographic test strip for Rabies virus was evaluated with dog saliva samples. The test was initially validated against 237 dogs of known infection status, and then evaluated in the field with 1,290 live dogs. By validation of paired saliva–brain specimens obtained from dogs at necropsy, the saliva strip test was 94.4% specific and 93.0% sensitive when compared to the gold standard fluorescent antibody test (FAT) on brain smears. The sensitivity and specificity of a nested polymerase chain reaction (nPCR) assay using saliva were 100% compared to the FAT results. The performance of strip test with field saliva samples from street dogs had a specificity of 98.7% in comparison to nPCR as the reference method. As the strip test kit can potentially be used outside the laboratory and be applicable as an on-site testing assay, it represents a powerful screening tool for epidemiological surveys and disease control. The test could be useful for the surveillance of rabies in dogs and, in particular, be used to monitor the success of rabies control programs.
Rabies, the most remarkable infectious disease of the central nervous system with the highest known case-fatality rate, remains a threat in Asia and Africa. 24 In Thailand, where dogs are the animals most frequently reported as rabid, the disease is endemic and wide spread. Cases of rabies in domestic animals other than dogs have been traceable to, or believed to have been at the origin of exposure to, rabid dogs. 11 In a sampling survey conducted by the Bureau of National Statistics and the Bangkok Metropolitan Administration (BMA) in 1999, it was estimated that 633,814 dogs inhabit Bangkok. Additionally, an estimation of the total pet dog population was reported to be 823,504 in 2006. This number is thought to have risen steadily, and the management of these dogs has become a serious rabies control problem in the capital.
According to a World Health Organization recommendation, at least 70% of the dog population must be continuously vaccinated to control rabies transmission. 25 Despite an intensive citywide rabies vaccination campaign, only 62% of the dogs investigated had received adequate immunization, 12 which indicates that at least 38% of the dogs were not vaccinated for rabies. Past attempts to conduct a mass dog vaccination have been limited to Bangkok City and have not been successful. Current rabies control programs include the elimination of the infected dog, but the risk of human rabies still exists. Dog bite–related rabies cases in human beings have accounted for 23 (96%) of the 24 deaths reported in 2009 of which 8 cases occurred in Bangkok (Ministry of Public Health annual report). Low vaccination coverage and ineffective management of stray dogs are the most likely reasons for the program’s lack of success.
Reports of apparently healthy dogs that intermittently shed Rabies virus in saliva have been reported. 5 Experimentally infected dogs were reported to have virus in the saliva up to 13 days before overt signs of rabies were observed. 6 Field observations suggest that a dog may remain healthy and shed Rabies virus for longer than the expected 10-day observation period prior to clinical development of rabies.21-23 Moreover, a dog may recover from rabies with the potential for intermittent virus shedding. 14 It cannot be predicted in which dogs rabies infection will persist, and it may lead to infection of an unexpected, untreated bite victim. The offending dogs usually escape, which can result in a potential spread of infection. Effective dog rabies control strategies not only serve to reduce human deaths but also can reduce the overall costs associated with rabies prevention. The surveillance of Rabies virus infection in dogs would help to prevent its spread to other animals.
In areas where dog rabies is endemic, all dog-bite victims should seek medical attention immediately, and administration of rabies post-exposure prophylaxis after being bitten should not be delayed. A biting dog should be euthanized whenever possible and its fresh brain tissue tested for rabies by the fluorescent antibody test (FAT). 4 Although sophisticated and expensive, nucleic acid amplification methods can be more sensitive than FAT with brain tissue samples that are too decomposed for direct examination 7 and can detect viral RNA in samples, such as saliva and cerebrospinal fluid (CSF), which are unsuitable for FAT testing. 3 Serological testing of serum is not usually helpful in dogs with previous rabies vaccination history. Recently developed molecular techniques permit the diagnosis of rabies in live animals by looking for evidence of Rabies virus infection in saliva and CSF. However, Rabies virus can be detected in a much higher frequency antemortem in saliva than in CSF. 20 Molecular methods have detected evidence of Rabies virus successfully in human cases where conventional methods could not be applied. 2 Reverse transcription polymerase chain reaction (RT-PCR) of RNA extracted from saliva was the most reliable diagnostic test.13,16 However, nested (n)PCR was required in almost all cases to compensate for the often extremely limited amount of RNA in antemortem samples. A more convenient and affordable diagnostic test should be developed that can be used outside the laboratory and be applicable as an on-site test. Recently, a rapid immunochromatographic test strip was developed and evaluated using clinical samples, particularly brain tissue.8,15 In the present study, a test strip capable of detecting Rabies virus was conducted on large numbers of dog saliva samples, and its performance evaluated by comparing with standard methods.
A total of 237 dog saliva samples were collected from dog carcasses submitted to the rabies diagnostic unit at Queen Saovabha Memorial Institute, the Thai Red Cross Society. Most of these carcasses were suspected rabid dogs. Saliva collection was performed as described elsewhere. 10 From the same dogs, brain tissue specimens (medulla and hippocampus) were collected and submitted for routine FAT. For FAT staining, fluorescein isothiocyanate–conjugated anti-rabies antibody prepared in rabbits a was used. An additional 1,290 saliva samples were obtained from apparently healthy dogs seen for surgical sterilization at the Rabies Control Subdivision of the Veterinary Public Health Division, BMA. All of the dogs were randomly captured from streets and public places of Bangkok. Sample collection was done with the dog under deep barbiturate anesthesia. All saliva samples were stored at −20°C until used.
Total RNA was extracted from saliva specimens using a chemical solution. b The RT-PCR was performed in 2 µg of extracted RNA following a one-step process using a commercial kit. c Subsequently, the primary amplified product was subjected to a second-round PCR. All amplifications were carried out on a thermocycler, d using a denaturation temperature of 94°C, an annealing temperature of 60°C, and an extension temperature of 72°C. Two sets of primers were used to amplify 1,473–base pair (bp; 5’-GTAACACCCCTACAATGGATGC-3’, at position 57-78, and 5’-CAAAGATCTTGCTCATGTTTGG-3’, at position 508-1529) and 524-bp (5’-GACATGTCCGGAAGACTGG-3’, at position 319-337, and 5’-GTATTGCCTCTCTAGCGGTG-3’ at position 823-842) fragments of Rabies virus nucleoprotein gene, for the first- and second-round PCR, respectively. The nPCR products were electrophoresed in 1.2% agarose gels and stained by ethidium bromide.
The test strip was constructed on the principles of immunochromatography using a combination of purified polyclonal antibody (pAb) and monoclonal antibody (mAb). The pAb was raised in horses, which were regular donors for equine rabies immunoglobulin (Ig)G production at the Thai Red Cross Horse Farm. 19 Such horses received a series of purified Vero (African green monkey kidney epithelial) cell rabies vaccine. e The IgG was fractionated out from the whole serum by the salting-out method with saturated ammonium sulfate. 17 Horse anti-rabies IgG was conjugated to colloidal gold particles (diameter: 20 nm) f and sprayed on a glass fiber pad g at 10 µl/cm using a dispensing system. h Mouse mAb was produced according to a standard method. 1 Briefly, somatic cell hybrids between mouse myeloma cells and spleen cells derived from BALB/c mice immunized with a purified chick embryo cell rabies vaccine i were cloned. One hybridoma clone producing mAb RM25 specific to Rabies virus glycoprotein was selected to grow in protein-free medium. j Purified anti-rabies mAb RM25 was micro-sprayed with an automated dispenser k onto a nitrocellulose membrane l at 1.5 µl/cm in the test zone. Rabbit anti-horse IgG m was micro-sprayed on the same nitrocellulose membrane at 1.5 µl/cm in the control zone. The membrane was dried at 40°C overnight. The test strip was assembled and housed in plastic cassettes n and stored in a desiccated plastic bag.
One hundred microliters of saliva was added to the sample hole of the test strip. The results were available within 10 min. Positive saliva sample was signified by the appearance of 2 red–purple bands, one at the test line and one at the control line, respectively. The sample was considered negative if only one band appeared in the control zone of the strip. The test was not valid if the control line did not appear.
The strip test was initially performed with the fixed CVS-11 strain of Rabies virus from the culture supernatant of infected BHK (baby hamster kidney)-21 cells and saliva samples from dogs that were previously known to be positive or negative for Rabies virus infection. The detection limit of the strip test was determined using CVS-11 strain culture supernatant possessing an infectious titer of 104.67 LD50/0.03 ml. The test detected approximately 10−3.31-fold diluted CVS-11 (101.36 LD50/0.03 ml). The PCR detection limit in the same assay was 100.46 LD50/0.03 ml, which was lower than that of the strip test (data not shown).
Saliva samples from 237 dog carcasses, which were previously known to be positive or negative for Rabies virus infection by FAT at necropsy, were examined for the presence of Rabies virus by PCR and test strips. Each specimen was coded and tested blindly. The results obtained by FAT using brain and strip test using saliva specimens from the same dog for Rabies virus antigen testing were as follows. Of 237 specimens tested, 53 were concordantly positive and 170 were concordantly negative. The remaining 14 samples showed discordant results. Four were positive by FAT but negative by strip test. The other 10 samples were negative by FAT but positive by strip test (Table 1). The sensitivity and specificity of the strip test relative to the standard FAT were 93.0% and 94.4%, respectively, whereas the sensitivity and specificity of the PCR using saliva were 100% compared to the FAT on brain smears (Table 1).
Evaluation of a rapid immunochromatographic test strip for detection of Rabies virus in dog saliva samples: performance of strip test, fluorescent antibody test, and nested polymerase chain reaction assay in detecting Rabies virus in dog carcasses.*
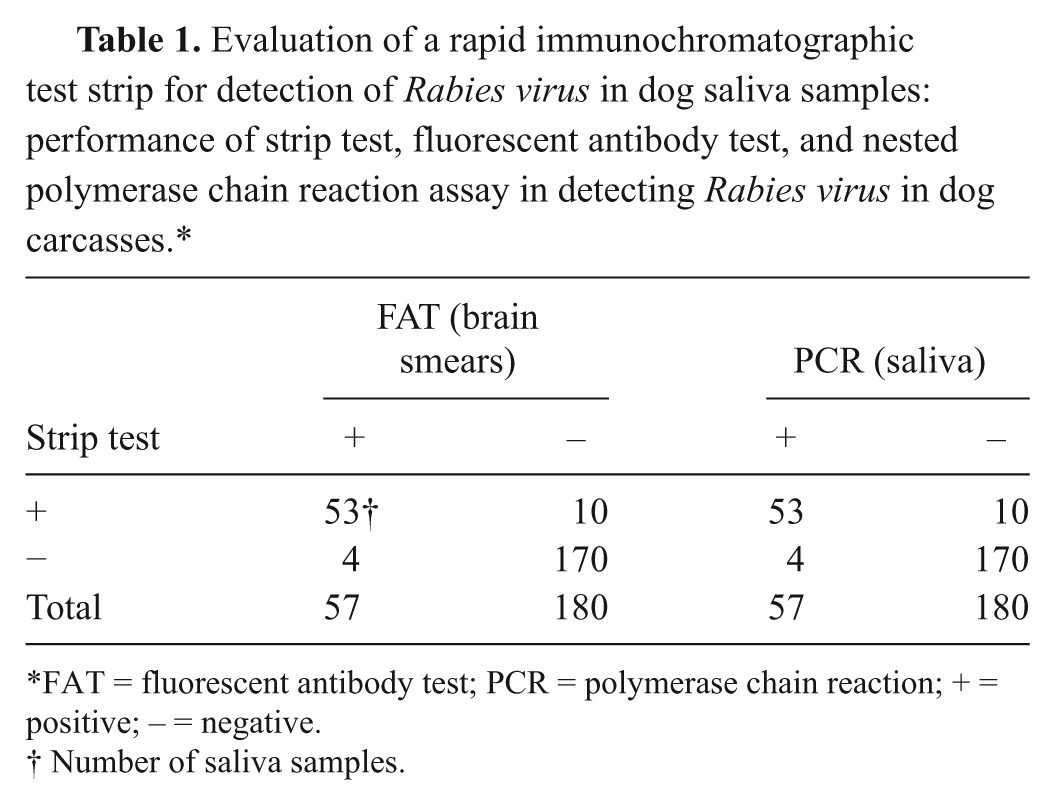
FAT = fluorescent antibody test; PCR = polymerase chain reaction; + = positive; – = negative.
Number of saliva samples.
The performance of strip test was further evaluated with field saliva samples from stray dogs by using PCR as the reference method. Most samples were largely expected to be negative. Among 1,290 samples, 17 produced a positive result on the strips but all were negative on analysis with the PCR. The occurrence rate of false-positive reactions was 1.3%, which equates to a specificity of 98.7% (Table 2).
Evaluation of a rapid immunochromatographic test strip for detection of Rabies virus in dog saliva samples: comparison of strip test to nested polymerase chain reaction assay for detecting Rabies virus in field saliva samples.*

PCR = polymerase chain reaction; + = positive; – = negative.
Number of saliva samples.
It is a fact that the load of infective virus in saliva is lower than that in brain. Virus may be absent or present at very low levels in saliva. The findings in the current study showed that saliva samples could be used as an alternative to brain specimens for Rabies virus testing, but results might depend on the stage of disease at the time of sample collection. Saliva collection during the late stage yields more positive results. 18 The present work would suggest that at least the PCR and FAT in the case of 237 established cases performed equally well. The reason for such a result might be that the dogs were probably euthanized late in the course of the disease. Virus is well distributed in many parts of the body of dogs (including salivary glands) during the final stage of infection. However, in this condition, the strip test presented lower sensitivity compared to the PCR. The detection limit of PCR (100.46 LD50/0.03 ml) is approximately 8-fold more sensitive than the strip test (101.36LD50/0.03 ml).
Relative to FAT, the sensitivity of the strip test was 93.0%, and indicates that approximately 7% false negatives should be expected. False-negative results identified by the strip test may be explained by the fact that the load of virus in saliva samples was below the detection limit of the strip. False negatives may be a consequence of technical limitations when saliva is the preferred clinical sample. Given the fact that misdiagnosis of rabies-shedding dogs by strip test may occur, careful interpretation is required in cases where Rabies virus infection is suspected. The test can be limited in terms of the amount of antigen that can be detected. The testing of serial saliva taken at different time intervals may be necessary. The quantum of virus and the duration of virus shedding in saliva are of paramount importance for rabies diagnosis. Any doubtful or negative results need to be confirmed by animal history or other laboratory techniques. Samples taken for antemortem diagnosis cannot definitively rule out rabies. If a suspicion of rabies persists despite negative findings, repeated sampling may be necessary. Additionally, performance validation carried out on saliva samples demonstrated that false-positive results were generated with the test strip. It is possible that some components in saliva, such as a small number of immune active and epithelial cells, small amounts of IgG, digestive enzymes, and a broad spectrum of bacteria of the normal flora 9 can affect the specificity of the test. The results of the field evaluation with stray dogs might suggest that the diagnostic specificity of strip test is possibly a little better than the initial validation suggested (98.7% vs. 94.4%) when compared to PCR.
Although PCR demonstrated a better performance when compared to strip test in the diagnosis of infected dogs, both the technological expertise necessary to perform saliva strip tests and the requirement for specialized laboratory equipment are minimal compared to PCR. From an epidemiological point of view, a rapid test is the fundamental basis for surveillance and control of every major infectious disease that allows interventions to be implemented in real time. Nested PCR takes at least 7 hr to perform, which can not be done practically in the field, whereas strip test could be carried out at the sites where the specimen is collected and the results can be read directly by the naked eye within 10 min. Such a test can help veterinarians, public health professionals, and others concerned with rabies prevention and control in the area to decide whether prompt euthanasia of the dogs or having them caged and observed for clinical signs is indicated. Alternatively, samples that are classified as positive by strip test can be forwarded to the local laboratory, where confirmatory diagnosis could be performed. The strip test has a significant potential use for diagnosing rabies under field conditions where rabies diagnosis is unavailable for the moment and when brain material is unavailable for testing. However, even strip test has sensitivity limits, and neither conventional postmortem FAT nor PCR can rule out a diagnosis of rabies unless samples are taken appropriately, forwarded to the examining laboratory in good condition, and tests are controlled rigorously. If used appropriately, the saliva strip test can be a valuable adjunct to traditional methods for rabies antemortem diagnosis. As cited above, Rabies virus may be present in the saliva of infected dogs for some considerable period of time before the onset of clinical signs. The diagnosis of rabies in a living dog by strip test may provide a positive diagnosis. This may be beneficial for dog rabies control. However, a negative result should not be interpreted as indicating a rabies-negative animal. The results of the current study demonstrate that the test strip works for Rabies virus detection in dog saliva. The strip test can offer speed, simplicity, and reasonable sensitivity and has a great potential for field use.
Footnotes
Acknowledgements
The authors thank Pacific Biotech Co., Ltd. for technical assistance.
a.
TRC®, Thai Red Cross Society, Thailand.
b.
TRIzol-LS™, Gibco-BRL, Gaithersburg, MD.
c.
SuperScript III with Platinum Taq®, Invitrogen, Carlsbad, CA.
d.
MWG-Biotech, Germany.
e.
Verorab®, Sanofi Pasteur, France.
f.
Heron Diagnostics, Thailand.
g.
Rapid 24®, Whatman, Fairfield, NJ.
h.
Imagene, Hanover, NH.
i.
Rabipur®, International AG, Basel, Switzerland.
j.
CD Hybridoma Medium AGT, Gibco-BRL, Gaithersburg, MD.
k.
BioDot, Irvine, CA.
l.
Prima60®, Whatman, Fairfield, NJ.
m.
Sigma-Aldrich, St. Louis, MO.
n.
Mcmould Entico, Thailand.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This work was supported by the Vejdusit Foundation under the Royal Patronage of Her Royal Highness Princess Galyani Vadhana Krom Luang Naradhiwas Rajanagarindra.
