Abstract
A rapid immune colloidal gold test strip for detecting 1 lineage of the H5 subtype of highly pathogenic avian influenza was developed based on membrane chromatography with monoclonal antibodies (mAbs) against hemagglutinin (HA) of the H5 subtype of Avian influenza virus (AIV). Bagg albino C mice that were immunized with AIV A/Goose/Guangdong/3/96(H5N1) yielded 4 hybridomas secreting mAbs (1A6, 1B8, 3C4, and 8F6) specific for HA. Monoclonal antibody 3C4 was conjugated with colloidal gold as the detector antibody. On the test strip, 1A6, in combination with rabbit anti-H5N1 polyclonal antibody, was used as the capture complex at the “test line,” and goat antimouse immunoglobulin G antibody was used as the capture antibody at the “control line.” The strip test correctly detected the H5 subtype of AIV in known positive samples and did not react with the standard antigens of AIV H1-H4 or H6-H14, or the antigens of Newcastle disease virus, Infectious bursal disease virus, Infectious bronchitis virus, or Avian infectious laryngotracheitis virus. When 483 clinical specimens suspected of infection with H5N1 were tested, results from the strip test were nearly identical with those from the HA/hemagglutination inhibition test, reverse transcription polymerase chain reaction, and a commercial enzyme-linked immunosorbent assay kit. Because the newly developed strip was specific, rapid, and sensitive, it should be useful for diagnosing the H5 subtype of AIV and for investigating its epidemiology. The test strip is only reliable for the lineage of H5N1. As for the other strains of AIV, further experiments will be needed.
Introduction
Influenza A virus (FLUAV; family Orthomyxoviridae, genus Influenzavirus A) infects humans, chickens, pigs, horses, and other animals, but the H5 subtype of Avian influenza virus (AIV-H5) causes the most damage. 1,2,4,8 In 1997, highly pathogenic avian influenza (HPAI) subtype H5N1 emerged in Hong Kong and now threatens the poultry industry and human health worldwide. 5,12 In the spring of 2004, HPAI H5N1 spread throughout the poultry populations in Asia and caused enormous economic losses. 16 In recent years, outbreaks of AIV-H5 have occurred in China and in many other regions of the world, again causing fatal disease and economic loss in the poultry industry. 15 Most importantly, this isolate of virus acquired the ability to infect humans, and by December 2007, HPAI H5N1 had infected 337 humans worldwide, among whom 207 died. For emergency diagnosis and monitoring of suspected influenza outbreaks, a method for rapidly detecting AIV-H5 is clearly needed.
The most common methods for detecting AIV-H5 include virus isolation, hemagglutination (HA)/hemagglutination inhibition (HI) test, reverse transcription polymerase chain reaction (RT-PCR), fluorescent quantitative PCR, and digoxin-labeled probe detection. 3,11 These methods, however, require relatively sophisticated laboratories, which are sometimes unavailable or inconvenient for clinical application. 6 In contrast, the colloidal gold test strip method provides rapid results but does not require sophisticated tools or special skills. 10 The colloidal gold test strip is a new immunochromatographic technique in which a cellulose membrane is used as the carrier, and a colloidal gold-labeled antigen or antibody is used as the tracer. This technology has several advantages over traditional immunoassays, such as simplicity of procedure, rapid operation, immediate results, low cost, and no requirements for skilled technicians or expensive equipment. Because of these characteristics, the colloidal gold test strip is suitable for the on-site detection of antibodies or antigens. The technology has been adapted for the surveillance and diagnosis of poultry infectious diseases, such as Newcastle disease and infectious bursal disease. Although a chromato-graphic strip test for rapid detection of nucleocapsid protein of AIV has also been developed based on this method, 9 a strip test specific for the H5 subtype of AIV has not been reported.
In the present study, 4 strains of anti-AIV-H5 antibodies were prepared, and an immune strip test for detecting AIV-H5 was developed. This strip test, which uses chromatographic technique and colloidal gold, provides a fast and sensitive detection tool for the diagnosis and epidemiological investigation of AIV-H5 and also provides a good foundation for the development of a final product. Because multiple lineages of H5 viruses (including Chicken Scotland, low pathogenicity strains, and other currently endemic strains) were unavailable, the reliability of the strip has only been demonstrated for the lineage of H5N1 investigated in the current study. The reliability of the strip test for other strains remains to be determined.
Materials and methods
Cells, virus standard antigens, experimental animals, and reagents
Myeloma cell line SP2/0 and the antigens of chicken Newcastle disease virus (NDV), Infectious bursal disease virus (IBDV), Infectious bronchitis virus (IBV), and Avian infectious laryngotracheitis virus (AILV) were maintained in the authors' laboratory; the A/Goose/Guangdong/1/96 (H5N1) strain of AIV-H5 was isolated and maintained in the authors' laboratory. 20 The standard antigens of AIV H1-H14 were provided by the Central Veterinary Laboratory (CVL) and World Health Organization (WHO). Specific pathogen-free (SPF) chicken embryos (age 9–11 days), New Zealand white rabbits (age 6 months), and Bagg albino C mice (age 6–8 weeks and 10 weeks), which were used for immunization and ascites preparation, were provided by the Experimental Animal Center in the Harbin Veterinary Research Institute (Harbin, China). Cloacal swabs from 483 birds that were suspected of being infected with avian influenza and had been submitted to the National Avian Influenza Reference Laboratory were subjected to the new test strip, HA/HI test, RT-PCR, and a commercial enzyme-linked immunosorbent assay (ELISA) kit (Anigen® H5 AIV Ag ELISA Kit a ). The following reagents were used: HAuCl4-4H2O, b nitrocellulose membrane and glass-fiber paper, c histone acetyltrans-ferase (HAT), d hypoxanthine thymine (HT) selective medium, d polyethylene glycol (PEG), d RPMI (Roswell Park Memorial Institute)-1640 medium, e Staphylococcal Protein A-conjugated goat antimouse immunoglobulin G (SPA-IgG), d a protein G perfusion affinity chromatographic column, f and SPF chicken anti-AIV-H5 serum (prepared and maintained in the authors' laboratory).
Monoclonal antibody preparation
Subtype H5N1 of AIV, which was cultured in chicken embryo, was purified with a sucrose density gradient as previously described. 14 For the subcutaneous multiple injections into the backs of mice, the virus was emulsified with an equal volume of Freund's complete adjuvant, and each mouse received approximately 80 μg of virus. The splenocytes from immunized mice were fused with SP2/0 cells at the logarithmic phase by treatment with PEG (MW6000). The hybridomas secreting monoclonal antibodies (mAbs) were selected by indirect ELISA. Large quantities of mAbs were prepared with the mouse ascites method. Monoclonal antibodies were purified using a protein G perfusion affinity chromatographic column. The purified mAbs were dialyzed in 10 mmol/l phosphate buffered saline (PBS). After dialysis, the protein concentration was determined with an ultraviolet spectrophotometer at 280 nm, and the preparation was cryopreserved in aliquots of 0.5 ml.
Determination of mAb titer
The titer of the 4 mAbs was determined by indirect ELISA. 7,18 The supernatant culture of hybridomas and crude ascites was diluted in a gradient from 1:10 to 1:100. The same diluted supernatant of SP2/0 cell culture and normal ascites was used as the negative control. Enzyme label plates coated with one kind of antigen (H5N1 antigen, AIV H1-H14 standard antigen, or antigens of NDV, IBDV, IBV, or AILV) were used to determine the specificity of the 4 mAbs.
Preparation of polyclonal antibody
Three New Zealand white rabbits were intramuscularly injected with purified H5N1 virus (0.5 mg/rabbit) mixed with Freund's complete adjuvant in a 1:1 ratio. Rabbits were subsequently injected 3 more times with the same volume every 3 weeks. One week after the fourth injection, rabbit antisera were collected.
Colloidal gold preparation, ratio of colloidal gold to coated antibodies, and labeling procedure
The colloidal gold (diameter of 20 nm) was prepared with a sodium citrate reduction method as previously described. 13 Colloidal gold fluid was adjusted to pH 8.2 with 0.2 mol/l of K2CO3. The high-titer and high-affinity mAb 3C4 (7 μg/ml) was added to 20 μl of colloidal gold, followed by 5% bovine serum albumin (BSA) to provide a final concentration of 1%. The precipitate was discarded after centrifugation at 1,000 X g for 20 min; the supernatant was discarded after centrifugation at 11,000 X g for 40 min. The precipitate was resuspended with colloidal gold fluid to a volume of 1 ml (optical density at 520 nm (OD520) = ∼0.25), and 0.5 mg/ml sodium azide was then added. The preparation was stored at 4°C.
The mAb 3C4 with conjugated colloidal gold was sprayed onto a glass fiber pad at 1.0 and 1.5 μl/cm, then dried at 40°C overnight. The combination of mAb 1A6 and rabbit anti-H5N1 antibody (1:1, 1 mg/ml) was micro-sprayed onto a nitrocellulose membrane (1 μl/cm) at a position that would become the capture test line (T) of the completed strip. Goat antimouse IgG antibody in PBS (1 mg/ml) was microsprayed onto the same nitrocellulose membrane (1 μl/cm) at a position that would become the capture control line (C). The membrane was dried at 40°C overnight.
Detection of the H5 subtype of Avian influenza virus (AIV) in allantoic fluids of clinical samples by the immune colloidal gold strip, virus isolation, hemagglutinin (HA)/hemagglutination inhibition (HI) test, reverse transcription polymerase chain reaction (RT-PCR) amplification, and the Anigen® H5 AIV Ag ELISA Kit.
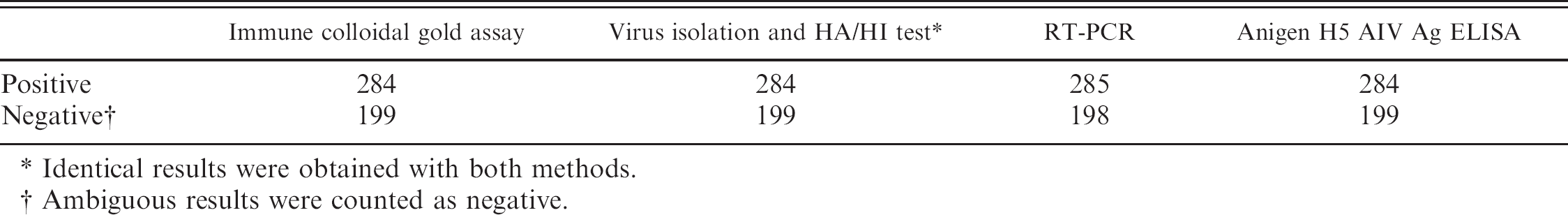
Identical results were obtained with both methods.
Ambiguous results were counted as negative.
Test strip package
To assemble a package or kit, the nitrocellulose membrane was arranged with mAb 3C4 conjugated with colloidal gold at one end, upstream of the test line, and an absorbing pad at the opposite end, downstream from the control line. Then, the assembly was cut into 3 × 60 mm strips. Each strip was placed in a plastic case, which was individually stored in a desiccated plastic bag.
Specificity, sensitivity, stability, and tests with clinical specimens
Specificity of the colloidal gold strip test was determined with H5N1 chicken embryonic culture, normal chicken allantoises fluid, standard antigens of AIV H1-H14, and antigens of NDV, IBDV, IBV, and AILV (for results, see Fig. 1). A pipet was used to add the sample or allantoic fluid to the sample pad, which was then placed on a horizontal surface for 5–20 min before the strip was examined for bands. The results were determined by band number and position. Positive specimens yielded 1 band at the control line and 1 band at the test line, whereas negative specimens yielded 1 band at the control line and no band at the test line (Fig. 2). The test results were discarded if there was no band at the control line. 17
To determine sensitivity, the H5N1 embryonated egg culture with known HA titer was 10-fold serially diluted, and the dilutions were tested with the colloidal gold strip, HA/HI test, and RT-PCR. To determine stability, strips that had been stored for 3, 6, 9, 12, 15, and 18 months at 4°C were tested with H5N1-positive and -negative specimens. At each sample time, 30 strips were tested. To determine how the strip test performed with clinical samples, 483 clinical specimens (cloacal swabs) suspected of having H5N1 infection were simultaneously subjected to the colloidal gold strip test, HA/HI test, RT-PCR, and Anigen H5 AIV Ag ELISA.
Results
Hybridoma preparation and titer of supernatants of positive hybridomas and ascites
The hybridoma cells were screened in 96-well cell culture plates. After selection by ELISA, 4 hybridomas that stably secreted mAbs were isolated, and the strains and their respective mAbs were labeled 1A6, 1B8, 3C4, and 8F6. Based on indirect ELISA, the titer of the 4 mAbs in ascites was greater than 1:8 × 104.
Specificity of mAbs
Specificity of the 4 mAbs was determined by ELISA. The mAbs 1A6 and 3C4 were highly specific; they reacted with H5N1 virus and AIV-H5, but did not cross react with other subtypes of AIV or the antigens of NDV, IBDV, IBV, or AILV.
Specificity of the immune colloid gold test strip
H5N1 allantoic fluid, standard antigen of AIV H1-H14, and virulent and normal allantoic fluid of NDV, IBDV, IBV, AILV, Marek's disease virus, and AIV-H9 were tested on the strip. The results were positive for allantoic fluid of H5N1 and standard antigen of AIV-H5 but were negative for the other antigens, indicating that the test strip was highly specific.
Sensitivity of the immune colloid gold test strip
One milliliter of the allantoic fluid of H5N1 with HA titer of 1:27 was serially diluted, and 50 μl of each diluted sample was added to test strips. The band appeared at the test line with all dilutions less than 1:1,000, but no band appeared when the dilution was 1:1,000 or greater. This sensitivity was similar to that with the HA/HI test, but lower compared with RT-PCR, which detected the virus at a minimal dilution of 1:10,000.
Stability of immune colloid gold strips
Strips that were stored for 3, 6, 9, 12, 15, 18, and 24 months at 4°C were tested with known H5N1-positive and -negative specimens to determine strip stability. All test results were the same from 3 to 15 months, with all known positives testing positive and all known negatives testing negative. After 18 months, however, 15% of the known positives tested negative, and after 21 months, 30% of the known positives tested negative. False positives were never detected.
Tests with clinical specimens
Cloacal swabs from 483 birds suspected of having AIV-H5 infection were handled according to the Manual of Standards for Diagnostic Tests and Vaccines. 19 The swabs were treated with 3 μl of PBS (10,000 U/ml of penicillin, 10 mg/ml of streptomycin, 5,000 U/ml of streptomycin) for 1 hr at room temperature. After centrifugation at 1,000 X g for 10 min, the supernatant was inoculated into the allantoic cavities of 9–11-day-old SPF embryonic eggs. After incubation at 37°C for 7 days, the allantoic fluid of each egg was subjected to the immune colloidal gold strip, HA/HI test, RT-PCR, and the Anigen H5 AIV Ag ELISA. The results from the immune colloidal gold strip agreed with those from the HA/HI test and the Anigen H5 AIV Ag ELISA. Results of the RT-PCR, which used the primers of the HA gene of AIV-H5, also agreed with those of the strip test, with 1 exception (Table 1).
Specificity of the immune colloidal gold test strip. The positive specimens suspected of being infected with Newcastle disease virus, Infectious bursal disease virus, Infectious bronchitis virus, Avian infectious laryngotracheitis virus, Marek's disease virus, AIV-H9, AIV-H5N1, and negative sample from specific pathogen-free chickens were simultaneously tested by this strip test.
Discussion
Prevention and control of AIV requires rapid and accurate detection methods. At present, the common methods for detecting AIV-H5 include virus isolation and identification, HA/HI test, and RT-PCR. Less common methods include fluorescent quantitative PCR and digoxin-labeled probes. Virus isolation has the disadvantage of requiring a long test cycle that is too long for the quick and useful diagnosis of influenza. Even though RT-PCR, fluorescent quantitative PCR, and digoxin-labeled probe detection are sensitive, there are practical limitations for their widespread commercial and clinical application. These limitations include the need for special equipment and highly trained personnel. The HA/HI test is a recommended method in AIV detection, but it is inconvenient and unsuitable for rapid, on-site identification of AIV-H5 and cannot distinguish between AIV-H5 and many other serotypes. In addition, results of the test may vary with laboratory personnel and the quality of erythrocyte and must be corroborated with other kinds of evidence. Therefore, a rapid method for detecting influenza virus is urgently needed. In the current study, mAbs against HA of AIV-H5 were coated with colloidal gold nanoparticles to prepare the “test strip.” A foundation for fast and specific diagnosis of AIV-H5 and epidemiological investigation, as well as for the development of a final end product, was developed.

Comparison between positive (
The key to differentiating subtypes of AIV with the immune colloidal gold test strip method is obtaining H5 subtype-specific mAb. In the current study, purified chicken embryonic culture containing H5N1 influenza virus was injected into mice, and 4 H5 subtype-specific mAbs were obtained by screening with ELISA plates that had been coated with different subtypes of AIV antigens. The specificity of the test strip was verified during the experiment in that the results were positive only for H5N1 virus and AIV-H5 standard antigens. This indicates that the test strip can be used to differentiate AIV-H5 from other subtypes of AIV and the antigens of NDV, IBDV, IBV, and AILV.
The results obtained with 483 clinical specimens suspected of having H5N1 AIV infection showed that detection of H5N1 was similar with the HA/HI test and the colloidal gold test strip. In addition, total RNA was extracted from the 483 specimens, and RT-PCR amplification was conducted using the primers of the AIV-H5 HA gene. The amplification results were again consistent with those of the colloidal gold test strip in all but 1 case. All of these results suggest that the test strip prepared in the current study could be used in the specific diagnosis and epidemiological investigation of AIV-H5. Owing to a limited availability of specimens, further research is needed to verify that the AIV-H5 test strip does not produce positive results with other subtypes of AIV. Determining whether the strip test produces reliable results with multiple lineages of H5 viruses (only 1 lineage was used in the current study) will require additional research.
Acknowledgements
This work was supported by grants from the Chinese National Program of R&D in Highly Pathogenic Avian Influenza Prevention and Treatment (2004BA519A01) and the Chinese National 10th Five-Year Science and Technology Plan, the Foundation for Rapid Detection Technology of H5 and H7 Subtypes of Avian Influenza Virus (2004BA519A07).
Footnotes
a.
Animal Genetics Inc., Gyeonggi-do, Korea.
b.
Shanghai Sangon Biological Engineering Technology & Services Co., Shanghai, China.
c.
Millipore Corp., Billerica, MA.
d.
Sigma-Aldrich China Inc., Shanghai, China.
e.
Invitrogen Corp., Carlsbad, CA.
f.
Amersham Health Inc., Princeton, NJ.
