Abstract
A rapid (<5 min) immunochromatographic strip using a colloidal gold–labeled antigen probe was successfully developed and applied for the detection of Porcine circovirus-2 (PCV-2) antibodies in swine. Recombinant Cap protein truncated nuclear localization signal of PCV-2, was expressed and labeled with colloidal gold. This conjugate was dispensed on a conjugate pad as the detector. Staphylococcal protein A and purified porcine anti–PCV-2 antibodies were blotted on a nitrocellulose membrane for the test and control lines, respectively. Sensitivity and specificity of this strip test was evaluated using PCV-2 antisera as well as other sera from pigs infected with a variety of swine viruses. For the validation of this strip test, 500 clinical swine serum samples were assessed both by the strip and a commercial enzyme-linked immunosorbent assay (ELISA) kit. The agreement between the immunochromatographic strip and ELISA kit was 94.00%. This strip possesses high sensitivity and specificity and may be useful as a candidate for rapid diagnosis of PCV-2 antibodies in the field.
Introduction
Porcine circovirus, a nonenveloped, single-stranded, circular DNA virus, is the smallest virus that replicates autonomously in mammalian cells.16,23 The virus has 2 genotypes: Porcine circovirus-1 (PCV-1) and Porcine circovirus-2 (PCV-2). Porcine circovirus-1 was originally identified as a contaminant of porcine kidney epithelial (PK15) cells, and does not induce disease in swine. 21 Porcine circovirus-2 is the crucial etiological agent of postweaning multisystemic wasting syndrome,1,17 a disease responsible for significant economic losses in the swine industry. Three major open reading frames (ORFs) have been recognized for PCV-2: ORF1 encodes a replication-associated protein (Rep), 15 ORF2 encodes a major structural capsid protein (Cap), 18 which is the main antigenic determinant of the virus, and ORF3 is involved in PCV-2 pathogenesis.12,13
To date, several commercial PCV-2 vaccines are in use and, therefore, a means of evaluating anti–PCV-2 antibody response that is convenient and rapid for use in the field is essential. Immunoperoxidase monolayer assay (IPMA), 8 indirect fluorescent antibody test (IFAT), 2 and enzyme-linked immunosorbent assay (ELISA) are the most common diagnostic methods for detecting PCV-2 antibodies. These methods require specialized equipment and technical expertise, and are suitable for laboratory use only. Furthermore, IPMA and IFAT require the cultivation of PCV-2 on PK15 cells, which is time-consuming, labor-intensive, and expensive. 22
The membrane-based immunochromatographic lateral flow strip test, which was initially developed as a convenient test for pregnancy, 26 represents a well-established and appropriate technique for a variety of point-of-care and field-use applications. The strip test has several advantages over traditional immunoassays, such as simplicity of procedure, rapid operation and immediate results, low cost, and no requirements for technical expertise or specialized equipment. 7 In the current study, a simple and rapid immunochromatographic strip was developed for the detection of PCV-2 antibodies in the field.
Materials and methods
Serum samples
The PCV-2–positive (5) and –negative (5) control sera a were obtained and then stored in Henan Provincial Key Laboratory of Animal Immunology, Henan Academy of Agricultural Sciences, Zhengzhou, People’s Republic of China. The PCV-2–positive (423) and –negative (77) clinical sera were collected in Henan Province, China from 10 swine herds (the age of the sampled pigs was more than 2 weeks). Some samples were collected from vaccination herds, others from infection herds. All serum samples were from different pigs. The PCV-2 status of the field samples was initially determined with a commercial PCV-2 ELISA kit. Positive serum samples for PCV-1, Classical swine fever virus (CSFV), Porcine reproductive and respiratory syndrome virus (PRRSV), Porcine parvovirus (PPV), and Pseudorabies virus (PRV) were stored in Henan Provincial Key Laboratory of Animal Immunology, Henan Academy of Agricultural Sciences (Zhengzhou, People’s Republic of China). Additional positive control sera and all PCV-2–positive and –negative samples were obtained from the authority department of the state, were certified by that department to have no cross-reactivity with other swine pathogens, and then were retested with ELISA kits and found to have no cross-reactivity with other swine pathogens. Following the protocol of their corresponding ELISA kits, all other control sera were determined to be strong positive.
Preparation of recombinant Cap protein of PCV-2
The ORF2 gene, truncated nuclear localization signal (NLS) sequence, was amplified by polymerase chain reaction (PCR) from the recombinant plasmid pMD18T-ORF2 (stored in Henan Provincial Key Laboratory of Animal Immunology, Henan Academy of Agricultural Sciences). Nuclear localization signal–deleted ORF2 gene (587 bp) was expressed in Escherichia coli BL21. The following primer sequences were used: forward primer 5′-AATGGATCCGGCATC TTCAACACCCGCCTCTC-3′ and reverse primer 5′-AAT AAGCTTAAGGGTTAAGTGGGGGGTCT-3′.
The purified PCR product was digested with BamHI and HindIII, b and cloned into the pET28a vector. c The recombinant plasmid was transformed to E. coli BL21-competent cells. A positive clone was grown at 37°C in lysogeny broth supplemented with 100 μg/ml kanamycin to an optical density of 0.6 at 600 nm; isopropyl-β-D-thiogalactopyranoside d was then added to a final concentration of 1 mM. After 15 hr of induction at 30°C, cells were harvested by centrifugation (8,000 rpm for 30 min) and resuspended in 20 ml of 100 mM Tris–HCl (pH 8.0). The cells on ice were disrupted by sonication and centrifuged at 12,000 rpm for 25 min. The supernatant was collected, and the protein was purified using nickel–nitrilotriacetic acid (Ni-NTA) resin e following the manufacturer’s protocol. The final protein product was examined by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE; Fig. 1) before storing at −80°C.
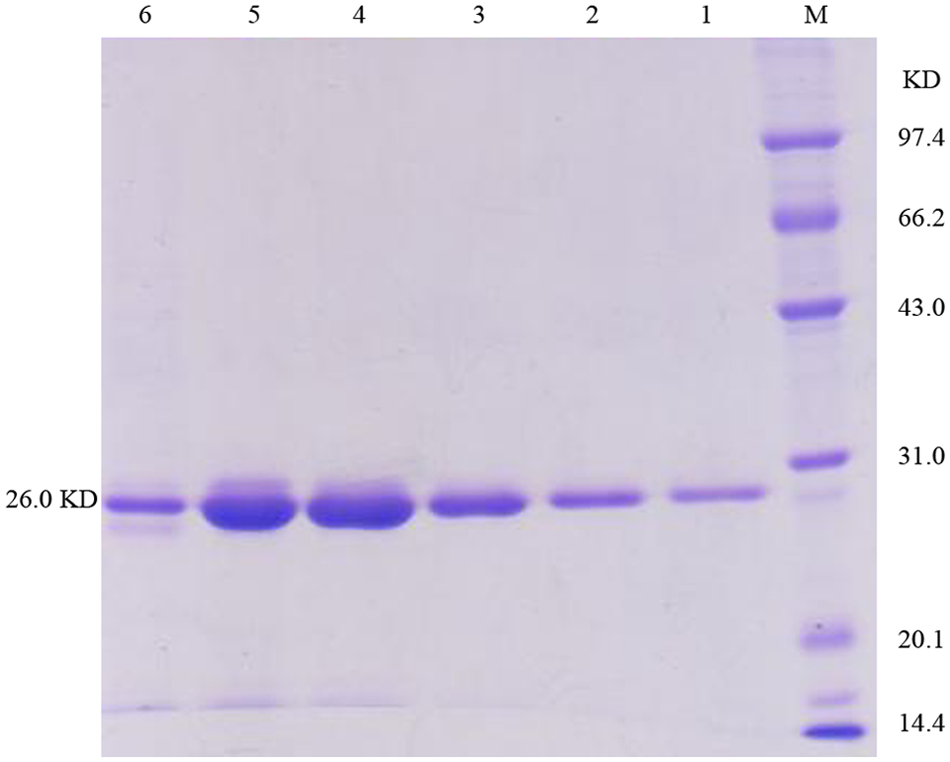
Analysis of the purified recombinant Cap subunit. Lane M: protein molecule marker; lanes 1–6: purified recombinant Cap subunit.
Conjugation of antigen with colloidal gold
Colloidal gold with an average particle diameter of approximately 20–25 nm was obtained by deoxidizing 1% chloroauric acid f with 1% trisodium citrate. In brief, 3 ml of 1% trisodium citrate (w/v) was added rapidly to 100 ml of 0.01% chloroauric acid solution (w/v) preheated to the boiling point, and then boiled for 5–10 min with constant stirring. The colloidal gold solution was then cooled to room temperature, and stored at 4°C.
The colloidal gold–labeled antigen was prepared according to a previously described protocol.27,28 Briefly, 0.5 ml of purified protein (0.5 mg/ml) was incubated with 20 ml of colloidal gold solution (pH 9.0) with rapid stirring. After the mixture was stabilized for 30 min at room temperature, 1 ml of 10% bovine serum albumin (BSA) f solution was added to the stirred solution, and stabilized for another 10 min at room temperature. The resulting suspension was centrifuged (15,000 rpm) at 4°C for 30 min. Finally, the colloidal gold–labeled antigen was resuspended with 2 ml of 0.02 M sodium borate buffer (containing 1% BSA, 1% sucrose, 0.1% NaN3), and stored at 4°C.
Preparation of the conjugate pad and sample pad
Ten percent BSA was dispensed on the glass fiber g conjugate pad using a dispensing platform. h The pad was dried at 42°C for 45 min. Subsequently, conjugate solution was dispensed on the modified glass fiber conjugate pad using the dispensing platform to produce the conjugated pad. The conjugate pad g was dried at 42°C for 1 hr and stored in a vacuum drying oven. The sample pad g was saturated with a buffer (pH 8.0) containing 0.02 M sodium borate, 0.1% Tween-20, and 0.1% NaN3. The pad was dried at 42°C for 2 hr and stored in a desiccator at room temperature.
Purification of PCV-2 antibody
Clinical serum samples were collected from several swine herds, and evaluated using an ELISA kit. i Sera with the highest titer of PCV-2 antibody were first purified by sequential precipitation with 50%, 40%, and 33% saturated ammonium sulfate (w/v), and further purified using a protein A/G spin kit, j following the manufacturer’s protocol. The PCV-2–specific immunoglobulin G (IgG) was isolated from other virus antibodies using an immobilization kit j (coupling cap). In this method, PCV-2 protein was cross linked to a solid matrix. The titer of the purified anti–PCV-2-specific IgG up to 1:1,600,000 was determined by an ELISA kit. The purified pig anti–PCV-2 IgG was then stored at −80°C. For the preparation of the control line, sera with highest titer of PCV-2 antibodies were selected to be purified. Sera with highest titer of PCV-2 antibodies were screened from a number of clinical serum samples using a commercial ELISA kit. As PCV-2–specific antibodies are only used on the control line as an indication of the effectiveness of the test, antibody purity was not exactly quantified.
Immobilization of capture reagents
Staphylococcal protein A f (1.2 mg/ml) and the pig anti–PCV-2 IgG (3.5 mg/ml, prepared above) were dispensed on the nitrocellulose (NC) membrane g using a dispensing platform h as the test (T line) and control lines (C line), respectively. The 2 lines were applied approximately 0.5 cm apart. The membrane was dried for 7 days at room temperature and stored under dry conditions.
Assembly of the test strip
The assembly procedure for the strip was as previously described. 10 The strip consisted of the NC membrane, antigen-gold conjugate pad, sample pad, and absorbent pad. The NC membrane was pasted at the center of an adhesive plastic backing. The absorbent pad and conjugate pad were then pasted by overlaying 1 mm on the upper and bottom of the NC membrane, respectively. The sample pad was then pasted by overlaying 2 mm on the bottom of the conjugated pad (Fig. 2). The master card was cut into 4.08 mm wide strips using a guillotine cutter. h
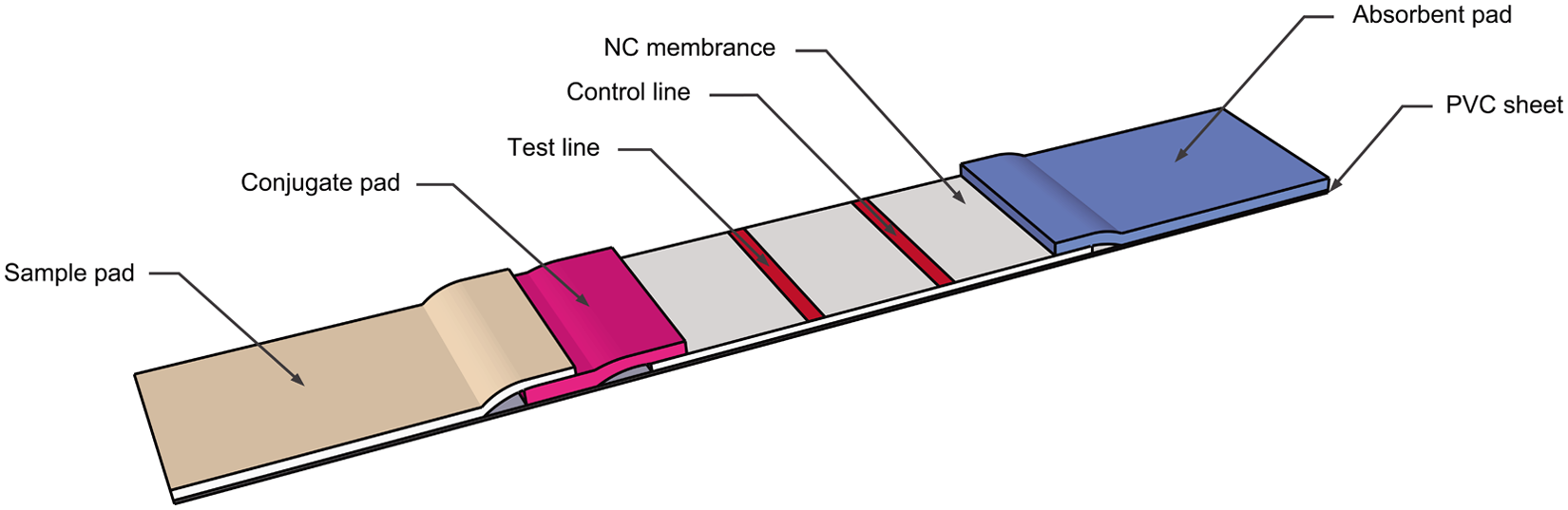
Schematic of the immunochromatographic strip device.
Principle of the test strip
The sample, applied to the sample pad, migrates toward the absorbent pad. If the serum sample tested contains PCV-2–specific antibody, the antibody and gold-labeled antigen will form a gold-antigen–antibody complex on the conjugate pad. The complex migrates into the NC membrane, and reacts with immobilized SPA giving a red-colored band at the test line. Unreacted gold-labeled antigen and any excess gold-antigen–antibody complex migrates further and reacts with the immobilized PCV-2 antibody giving another red-colored band at the control line. Absence of the red-colored band at the test line region suggests a negative result. The color phenomenon of the control line is regarded as a standard to evaluate if the strip is valid (color) or invalid (colorless; Fig. 3).
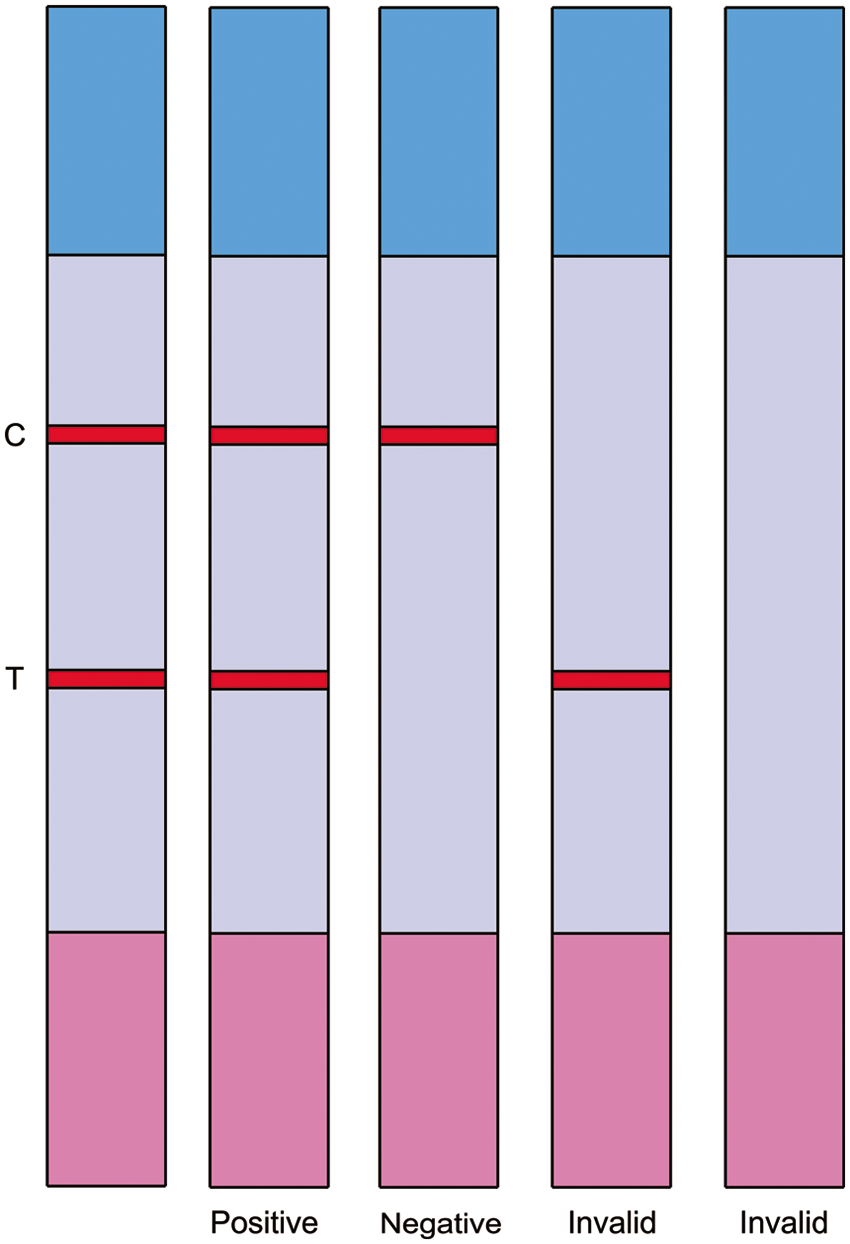
Principle of result judgment on the immuno-chromatographic strip.
The strip would indicate a red-colored test line when a sample was positive or strong positive. If a sample contained no PCV-2 antibodies, only a red-colored control line was seen. A weak positive sample could result in a poor test line with weak intensity. An ambiguous result occurred if the test line was almost invisible or if practitioners have different judgments about the result.
The results provided by the strip were clear positives or negatives. Samples with ambiguous results were retested. If the immunochromatographic strip provided another ambiguous result, the sample was scored negative.
Sensitivity and specificity
The sensitivity of the immunochromatographic strip was evaluated with PCV-2–positive serum. The titer of the serum samples was obtained using a commercial ELISA kit. i Following the protocol, twofold serial dilutions of the sample were made to detect the coating antigen. Using the cut-off value provided by the kit, the highest dilution factors of positive samples were considered to be their corresponding titers. Sera with titers of 1:25,600 tested by ELISA kit were diluted from 1:200 to 1:51,200 and tested 3 times with an immunochromatographic strip by different operators. The specificity of the immunochromatographic strip was tested with 30 PCV-2–positive serum samples, 10 PCV-2–negative serum samples, 1 PCV-1–positive serum sample, and 22 serum samples positive for CSFV, PRRSV, PRV, and PPV. The performance of the strip was evaluated by testing different categories of serum samples including strong positive, positive, weak positive, and negative. The anti–PCV-2 antibody levels in these samples were available after they were tested with the commercial ELISA kit. Each category of antibody levels contained many samples.
Comparison with ELISA
The immunochromatographic test strip was applied to the testing of 500 field serum samples collected from 10 swine herds in Henan province. The sera were also evaluated for anti–PCV-2 antibodies using the commercial ELISA kit following the manufacturer’s instructions. All the 500 sera were removed from −20°C storage, thawed swiftly, and then tested.
Results
Expression and purification of the Cap protein in E. coli
The recombinant Cap protein, lacking the N-terminal 40 residues, was expressed and purified as a monomeric subunit from E. coli (BL21). The protein of approximately 26 kDa contained an inserted hexahistidine (His6) tag. After sonication treatment, the target protein existed completely as soluble protein. Following purification, SDS-PAGE showed the purity of the protein was over 90%. In an ELISA protocol, this purified protein specifically reacted with PCV-2–positive serum, indicating it possessed high antigenicity (data not shown).
Sensitivity and specificity of the immunochromatographic strip
The PCV-2–positive serum samples were tested using immunochromatographic strips, 3 times, by different operators. A dilution of 1:25,600 was used. The PCV-2–positive serum samples provided a red–purple band at both test and control line, while other sera showed only one visible band at control line. The results indicated that the strip had a high sensitivity for detecting PCV-2 antibodies.
The specificity of the immunochromatographic strip was demonstrated by testing 30 PCV-2–positive serum samples, 10 PCV-2–negative serum samples, 1 PCV-1–positive serum samples, and 22 serum samples positive for CSFV, PRRSV, PRV, and PPV. As shown in Figure 4, PCV-2–positive serum samples provided a red–purple band at both test and control line, while other sera showed only 1 visible band at control line. The strip gave 30 PCV-2–positive results from 30 PCV-2–positive samples, while all PCV-2–negative samples and the samples positive for other virus antibodies tested negative by the strip. The specificity of the immunochromatographic strip was 100.00% (63/63).
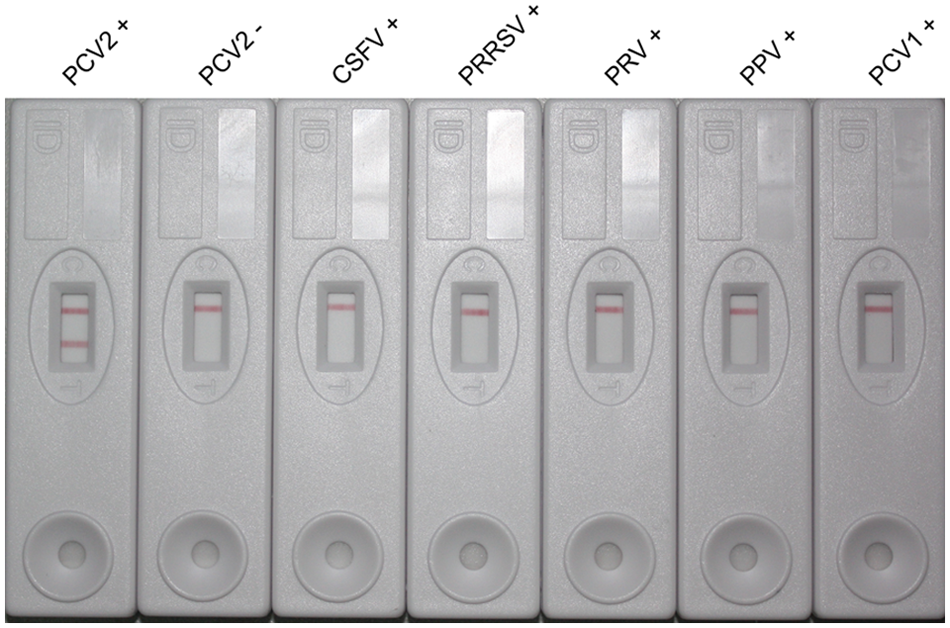
Specificity of the immunochromatographic strip test. The positive sera of Porcine circovirus-2 (PCV-2), Porcine circovirus-1 (PCV-1), Classical swine fever virus (CSFV), Porcine reproductive and respiratory syndrome virus (PRRSV), Pseudorabies virus (PRV), Porcine parvovirus (PPV), and PCV-2–negative sera were simultaneously tested by using the immunochromatographic strip. The result can be determined by visual examination.
Comparison with ELISA
Five hundred serum samples collected from farms were tested in parallel with the immunochromatographic strip and the ELISA kit (Table 1). There was an excellent agreement between immunochromatographic strip and ELISA kit (94.00%, 470/500). The ELISA kit determinations were established as the true value and were used as the standard for comparison. Sensitivity and specificity of the test were calculated as TP/(TP + FN) and TN/(TN + FP), respectively. The corresponding values were 93.14% and 98.70%, respectively.
Comparison of the newly developed immunochromatographic strip test with the enzyme-linked immunosorbent assay (ELISA) kit.*

ELISA kit obtained from JENO Biotech Inc., Chuncheon, South Korea.
Discussion
With the development of immunogold conjugates3,6,9 and the lateral-flow technique, 5 the immunochromatographic strip has been widely applied to the detection of allergies, infectious diseases, environmental contaminants, drugs, cardiac markers, and fertility. Several immunochromatographic strips for the detection of virus antibodies, such as PRRSV 7 and FMDV, 25 have been developed and applied to the evaluation of antibody titers.
The Cap protein, as the major structural protein of PCV-2, is highly immunogenic and strongly reactive with PCV-2–positive serum samples. 18 PEPSCAN analysis revealed the presence of type-specific immunodominant epitopes, as potential probes for the specific diagnosis of PCV-2. 14 Thus, a peptide ELISA using immunodominant B-133 epitope derived from the Cap protein was established. 24 This method is not suitable for PCV-2 sera diagnosis, as it gives high specificity but low sensitivity. Previous studies have shown that the PCV-2–ORF2 protein was recognized by a serum from pigs following PCV-2 infection, but no reactivity was observed with the PCV-1–ORF2 protein. 14 Therefore, a Cap protein–based ELISA for PCV-2 specific diagnosis was developed using glutathione-S-transferase (GST)-fused antigen expressed in recombinant baculovirus-infected insect cells. 4 Recombinant GST-fused protein lacking NLS expressed in E. coli cells was also used in an ELISA test. 19 However, these assays only provide accurate results in the laboratory using specialized equipment. In the current study, a soluble His-fused Cap protein was expressed in E. coli cells and purified using Ni-NTA resin. An immunochromatographic strip using His-fused recombinant protein was developed and applied to the detection of anti–PCV-2 antibodies in swine.
Following purification by Ni-chelating chromatography, a target protein concentration of at least 90% was obtained. Real purity may be somewhat higher as shown by Figure 1. Only nanogram amounts of target protein were required for conjugation with colloidal gold. The working conditions for this strip test were maximally optimized to reduce nonspecific reactions caused by E. coli. The high agreement between the strip test and the ELISA is an indication of the high purity of target protein.
The immunochromatographic strip is composed of a sample pad, conjugate pad, NC membrane, and absorbent pad. The sample pad, dispensed with Tween-20, and the conjugate pad, treated with BSA solution, allowed for fast release of the gold-conjugated probes. Glass fiber paper 33 was found most suitable for the conjugate and the sample pad. The pore size of the NC membrane was a crucial factor to control diffusion speed and protein-binding capacity on the immunochromatographic strip. Membrane with a large pore size gives high rates of diffusion but low protein-binding capacity and vice versa. It was found that the 90 s/4 cm NC membrane was most suitable for the strip. The gold-labeled probes dispensed on the conjugate pad, and the SPA dried on the NC membrane, will strongly react as the sample migrates toward the absorbent pad. In the current study, the purified protein was labeled with colloidal gold, and the 1.2 mg/ml SPA was dispensed on the T line, giving suitable sensitivity and specificity for the detection of any immune complex. Otherwise, the 3.5 mg/ml high titer PCV-2 antibodies, purified with serial methods, were dispensed on the C line, showing a good capture capacity.
The strip test was validated by testing clinical swine sera and comparing the results to those using ELISA kit results. The strip had a high agreement with the ELISA kit. The strip was found to possess high sensitivity for the detection of PCV-2 antibody and no cross-reactivity with other pathogenic antibodies, such as PCV-1, CSFV, PRRSV, PPV, and PRV. Thus, it has the potential for field practice to monitor antibodies induced by PCV-2 vaccine. However, the strip cannot discern vaccine-induced antibodies from antibodies derived from natural exposure. This is a problem with all detection methods. Monitoring the overall increase and decline of antibody levels in swine herds may to some extent help evaluate vaccine response. Testing before and after vaccination may be helpful in identifying vaccine-related antibody induction. The kit can also be used as a serological diagnostic tool for PCV-2 in nonvaccinated swine herds.
Porcine circovirus-2 can be divided into 2 genetic groups, PCV-2a and PCV-2b. In the current study, the ORF2 gene fragment was derived from PCV-2b. Theoretically, this assay could distinguish between anti–PCV-2 sera and anti–PCV-2a. However, as there is cross reactivity in serotype between PCV-2a and PCV-2b, the strip test can also detect both of those sera.
Further investigations are needed to develop a portable strip reader 11 to provide the end user with options for on/off screening and quantitative results. Past studies have shown that PCV-2 antibodies were ubiquitous in swine, mouse, cattle, and human beings. 20 As SPA used in the test line of this strip has the capacity to bind most Fc fragments of mammalian IgG, this strip should be suitable for testing PCV-2 antibodies of other species. Further investigations will focus on the detection of PCV-2 in other mammalian sera.
Footnotes
Acknowledgements
The authors are grateful to Professor Chuanqing Wang, College of Veterinary Medicine, Henan Agricultural University, for providing clinical serum samples. Qianyue Jin, Jifei Yang, and Qingxia Lu contributed equally to this study.
a.
Veterinary Medicine Research Development Inc., Washington, DC.
b.
TAKARA Bio Inc., Tianjin, China.
c.
Novagen Inc., Darmstadt, Germany.
d.
Solarbio Science & Technology Co., Beijing, China.
e.
Qiagen China Co., Shanghai, China.
f.
Sigma Chemical Co., St. Louis, MO.
g.
HiFlow 90, Millipore Co., Billerica, MA.
h.
XYZ-3000, BioDot Inc., Irvine, CA.
i.
JENO Biotech Inc., Chuncheon, South Korea.
j.
AminoLink Plus, Pierce Biotechnology, Rockford, IL.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by China Agriculture Research System (no. CARS-36).
