Abstract
Atypical porcine pestivirus (APPV) is a cause of congenital tremors (CTs) in piglets and has been found in swine populations around the globe. Although systemic distribution of the virus has been reported, there is limited information regarding viral localization at the cellular level and distribution at the tissue level. We collected multiple tissues from 2-d-old piglets (n = 36) born to sows inoculated at 45 or 62 d of gestation with APPV via 3 simultaneous routes: intravenous, intranasal, and directly in amniotic vesicles. In addition, 2 boars from APPV-inoculated sows with CT were raised and euthanized when 11 mo old. In situ hybridization performed on tissue samples from piglets demonstrated a broad and systemic distribution of viral RNA including endothelial cells, fibroblasts, and smooth muscle. Labeling in tissues was more pronounced in piglet tissues compared to boars, with the notable exception of diffuse labeling of the cerebellum in boars. Presence of APPV in boar tissues well after resolution of clinical signs suggests persistence of APPV similar to other pestiviruses.
Pestivirus K is more commonly known as atypical porcine pestivirus (APPV; Flaviviridae, Pestivirus). 15 Pestiviruses that cause economically important diseases in livestock include bovine viral diarrhea virus (BVDV), border disease virus, and classical swine fever virus (CSFV), and are known to establish persistent infections in host species. 16 APPV is an enveloped, single-stranded, and positive-sense RNA virus with a genome of 10.8–11.5 kb.7,17 Since it was first sequenced in 2015, APPV has been found in swine samples worldwide including North and South America, Europe, and Asia.2,5,7,18
Although APPV has been found in swine samples from apparent healthy animals, a number of investigations into piglets with congenital tremors (CTs) found samples positive for APPV.1,4,13 CTs are characterized by action-related clonic contractions of skeletal muscle, and have been observed in swine for nearly a century. 9 Multiple etiologies have been proposed for the types of CTs in neonates, including CSFV (type A-I), an unknown infectious agent (type A-II), genetic defects (type A-III, -IV), and toxicosis (type A-V). 4 It was hypothesized that APPV could be a cause of type A-II CTs in piglets, which was experimentally supported by the inoculation of APPV-positive serum into pregnant animals that gave birth to litters exhibiting CTs.1,4
In piglets with APPV-associated CTs, gross findings have been absent, but reported histologic lesions have included vacuolation of the cerebellar white matter and hypomyelination of the white matter in the spinal cord.13,14 Research groups have performed PCR and IHC on tissues, showing systemic distribution of the virus.1,11,13,18 In addition, others have performed prolonged longitudinal sampling of CT-affected litters in the field and reported samples such as serum, feces, and saliva positive for APPV by PCR for months.4,14 However, cellular tropism, as determined by RNA in situ hybridization (ISH) and persistence of virus in tissues, has not been evaluated extensively.
Our aims were 3-fold: 1) analyze tissue samples from experimentally inoculated piglets to evaluate distribution of APPV; 2) analyze tissue samples from ~1-y-old pigs in which APPV-associated CTs were present at birth and had resolved, to determine persistence of the virus in select tissues; and 3) compare viral distribution patterns from piglets and older pigs using RNA ISH and quantitative tissue image analysis. Fetal amniotic vesicles were directly inoculated as well as intranasal and intravenous inoculation of the sows with APPV-positive serum or phosphate-buffered saline sham at 45 or 62 d of gestation as reported previously (Institutional Animal Care and Use Committee of Iowa State University, Log 1-14-7907-S 2). 1 Most piglets in each virus-inoculated litter (57–100%) developed CTs. All 36 piglets were euthanized 2 d after birth, and tissues were collected. As part of another experimental inoculation study, 2 male piglets with CTs were euthanized when 11 mo old to analyze tissue distribution of the virus.
Tissue sections were placed in 10% neutral-buffered formalin for 24 h, processed routinely, paraffin-embedded, and sectioned at 4-μm thickness. An ISH assay (RNAscope; ACD) was used for detection of APPV viral RNA in piglet and boar tissues. RNAscope probes were designed specifically for the Npro-Erns coding region of the APPV strain used in the experimental inoculation. Slides were converted to digitized images (Aperio digital pathology system; Leica), and image analysis software was used for visualization and interpretation of the digitized images (HALO; Indica). Images were analyzed to quantify the amount of positive viral labeling for each tissue and specific anatomic location.
Initially, cerebrum and cerebellum were analyzed for viral labeling from all piglets. HALO scores of the labeling in the cerebellum were utilized to provide objective quantification. Tissue area for quantification of labeling was determined manually to contain all cerebellum tissue present on the slide for each piglet. Scores were provided as a percentage of tissue area that contained viral labeling. One animal had such extensive and intense labeling that the score provided by the software was >100%. Scores varied widely within those piglets born with CTs (0–118%, mean 15%) and those born without CTs (0–87%, mean 12%). In addition, evaluation of viral labeling patterns differed between piglets, with some being more localized and others diffuse. Four piglets, with CTs, were selected for further tissue analysis based on HALO scores that represented a range of diffuse-to-localized viral labeling patterns. Tissues selected included liver, kidney, spleen, heart, testis, lymph nodes, tonsil, thyroid, small and large intestine, lung, adrenal gland, and thymus. These tissues were evaluated for viral labeling to determine if the distribution or degree of viral labeling in the cerebellum was associated with the degree and distribution of viral labeling in other tissues. Based upon results from the piglet tissue analysis, cerebellum, large intestine, and lymph nodes were the only tissues analyzed from the boars. Testicular tissue was also evaluated from the boars, although there were difficulties with tissue adherence to the slide for RNA ISH evaluation.
Areas where viral RNA was observed were similar between boars and piglets; however, in general, tissues from piglets had more intense labeling. Viral RNA was commonly observed within endothelial cells (Fig. 1A), vascular tunics (intima, media, adventitia), fibroblasts, and the fibromuscular stroma in tissues such as the liver, kidney, colon, lung, lymph nodes, tonsil, thymus, and thyroid (i.e., capsule, trabeculae, septa). In the heart, viral RNA was detected in the epicardial mesothelium and endocardial endothelium but less so in the myocardium. In the testes, abundant labeling was observed in the tunica albuginea, with moderate labeling noted in the lumen of seminiferous tubules. In the spleen, abundant labeling was observed in the fibrous capsule and trabeculae; viral RNA was less commonly associated with cells of the red or white pulp, with the exception of the arterioles of the periarteriolar lymphoid sheaths. Positive labeling was observed in the smooth muscle layers of the small and large intestines (Fig. 1B) with occasional labeling in the serosal connective tissue and epithelium, lamina propria, and goblet cells.
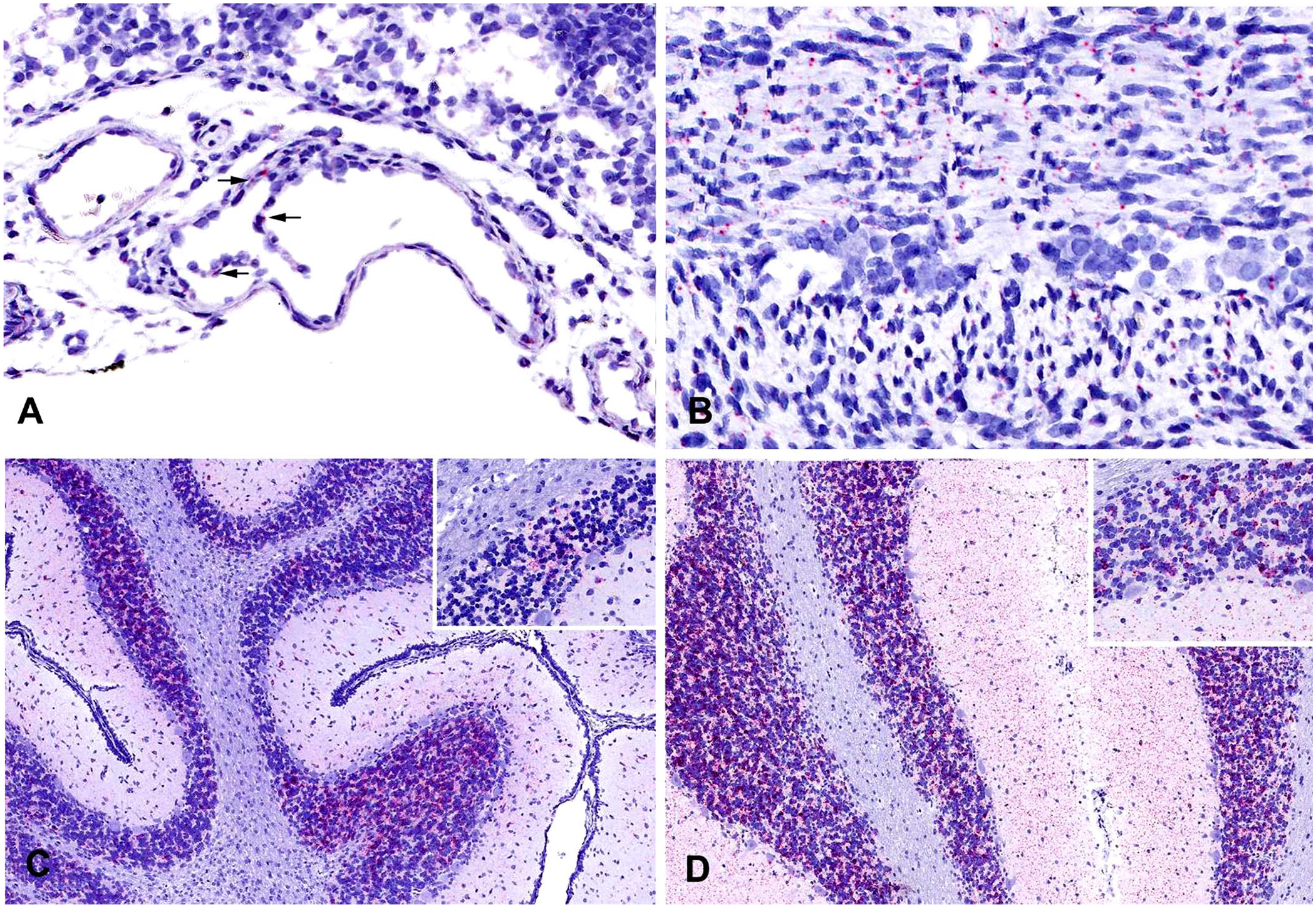
Piglets and boars experimentally infected with atypical porcine pestivirus (APPV). RNAscope in situ hybridization.
Regardless of age, the cerebrum and cerebellum contained the most abundant viral RNA. In piglets, viral RNA had a multifocal distribution within the internal granular and molecular layers; less abundant labeling was seen in the external granular layer (Fig. 1C). The degree of labeling varied between piglets, ranging from one to several small foci to larger focally extensive areas. In contrast to the multifocal distribution of labeling seen in piglets, cerebellum from infected boars had widespread, intense positive labeling of granular and molecular layers (Fig. 1D). Minimal-to-no viral RNA was noted in the white matter of either piglets or boars. Interestingly, whether piglets had more localized or diffuse labeling, viral localization in all other tissues in the piglets was similar in labeling intensity and location.
These data from a controlled experimental inoculation support previous fieldwork detailing the systemic distribution of APPV in tissues of piglets in CT-affected litters.13,17 RNA ISH results also support previous IHC analysis detailing APPV-positive staining in the molecular layer of the cerebellum, reticular cells in the lymph nodes and spleen, and endothelial cells in the lymph nodes, spleen, liver, and kidney. 11 Virus was found in neonatal piglets with CTs, but virus was also detected in piglets without CTs and asymptomatic boars ~11 mo old. Systemic distribution of viral antigen has been reported in animals infected with other pestiviruses. For example, BVDV can also be found in the vascular walls, lymphoid tissues, and in the intestines, although virus was localized to the epithelium rather than the smooth muscle layer as seen with APPV. 10 CSFV antigen can been found in endothelial cells of the tonsil and spleen, lymphoid tissue, and occasionally in the smooth muscle cells of blood vessels. 12 In addition, CSFV can be found in other tissues including kidneys and lungs, similar to APPV. Finally, Phocoena pestivirus, a pestivirus that infects cetaceans, was also found in smooth muscle cells in muscular layers of tissues throughout the body. 8
The boar tissue findings suggest persistence of the virus, which has been reported for other pestiviral infections. 16 This persistence could also explain report from the field of the virus in apparently healthy older swine.6,13,18 Further research will be needed to determine if persistently infected animals continue to shed infectious virus. APPV has been demonstrated in semen from boar studs by PCR testing, but infectivity is unknown. 6 Timing of infection, such as during insemination, may also play a role in the pathogenesis of APPV infection and recognition by the fetal immune system. For example, BVDV infection of a naïve cow during gestation can result in abortion or a calf that is immunotolerant and persistently infected, depending on the timing of infection. 3
Although evidence of viral RNA was observed in most tissues, the amount of labeling and location of APPV in the cerebellum and central nervous system of piglets could provide insights into the pathogenesis of CTs in piglets infected with APPV. The multifocal distribution of APPV in piglets could also provide insight into the process by which APPV in the cerebellum transitions from abnormal signals that may be related to control of the virus, to localized regions, to complete dissemination of the persisting virus, as observed in the boars. It is unknown if resolution of CTs is correlated with complete dissemination of the virus in the cerebellum. Despite the extensive viral distribution, the boars did not show clinical signs of neurologic disease. Perhaps different neural connections are formed to compensate, or tremors are still present but not observable to the naked eye. Localization of APPV in immune-privileged sites such as the cerebellum may provide an area for the virus to evade the host immune response.
Understanding the cascade of events occurring in the cerebellum in CT, as well as the physiologic effects of secondary remodeling or rewiring that may be occurring in piglets with CTs that result in resolution of clinical disease despite persistent infection of cells in the cerebellum, may allow for translational avenues in tremor disorders. In addition, further research into the persistence of virus in animals without clinical signs should be pursued to evaluate the effect on the immune system, reproduction, and the ability of those animals to transmit APPV to naïve animals.
Footnotes
Acknowledgements
We thank Patricia Federico, Judith Stasko, and Adrienne Shircliff for their technical assistance. Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. USDA is an equal opportunity provider and employer.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
