Abstract
We characterized the immunohistochemical expression profiles of dysgerminomas from a 16-y-old maned wolf and 13 domestic dogs using the following biomarkers: Sal-like protein 4 (SALL4), octamer-binding transcription factor 3/4 (OCT3/4), placental alkaline phosphatase (PLAP), c-kit, and vimentin. The maned wolf had nonspecific and long-standing clinical signs of lethargy, anorexia, and weight loss, and was euthanized because of poor prognosis. At autopsy, the left ovary was effaced by a 12 × 8 × 6 cm mass, comprised of anaplastic cells with a mitotic count of 20 mitoses in 10 high power fields. Dysgerminomas from 7 of 13 domestic dogs had nuclear expression of SALL4. Dysgerminomas from the maned wolf and 2 domestic dogs had both nuclear and cytoplasmic expression of SALL4. Cytoplasmic expression of PLAP and OCT3/4 was present in dysgerminomas from the maned wolf and 3 (PLAP) or 4 (OCT3/4) domestic dogs. All dysgerminomas expressed vimentin. Membranous c-kit expression was rare in the dysgerminoma from the maned wolf, and variable in dysgerminomas from 4 domestic dogs. A dysgerminoma from a domestic dog had cytoplasmic expression of c-kit. SALL4 is a useful marker to confirm germ cell origin of dysgerminoma in canids.
The maned wolf (Chrysocyon brachyurus) is the largest wild canid distributed throughout South America, most commonly in Brazil,11,12 from Rio Grande do Sul state in the South Region to the Southeast, Midwest, and some states of the Northeast Region. 12 The species is listed as near-threatened by the IUCN. 11 Information regarding neoplastic diseases in maned wolves is scarce and mainly restricted to a few case reports, such as mammary tumors, 6 pancreatic gastrinoma, 5 bladder teratoma, 8 and ovarian tumors.2,15 Dysgerminoma is among the most common ovarian tumor in this species, and this high prevalence is suspected to be the result of a hereditary predisposition. 15 These tumors are clinically silent, similar to the situation in domestic dogs. Nonetheless, metastases in the omentum and iliac lymph nodes have been reported in a maned wolf. 15 Immunohistochemical profiling of ovarian dysgerminoma has not been performed in maned wolves, to our knowledge. We characterized the pathologic and immunohistochemical features of a unilateral dysgerminoma in a senile captive maned wolf, with a special focus on germ cell biomarkers, and compared the immunolabeling for these markers with that of dysgerminomas from 13 domestic dogs.
A 16-y-old, intact female maned wolf was housed alone in a zoo located in Sapucaia do Sul, Rio Grande do Sul state, Brazil. The wolf was lethargic and inappetent for 2 mo and had a 1-mo history of constant pruritus from suspected flea allergy dermatitis. Although the skin lesions improved with treatment, the maned wolf was euthanized because of long-standing anorexia, weight loss, lethargy, and poor clinical prognosis. Physical examination findings included a palpable space-occupying abdominal mass, mild generalized gingival hyperplasia around the mandibular incisors, absence of lower canine teeth, and upper canine dental pulp exposure. Ultrasonography revealed a 6.5-cm round mass within the left mesogastric region of the abdomen. This mass had variable echogenicity with heterogeneous echotexture.
Cytologic examination of the ovarian mass revealed a highly cellular sample composed of large, individual, round-to-polygonal cells, with scant and poorly limited cytoplasm and round-to-oval nuclei displaying coarse and aggregated chromatin. Nucleoli were prominent, variably shaped, and frequently multiple. The cells had marked anisocytosis, anisokaryosis, and anisonucleoliosis. Additional criteria of malignancy included marked macrokaryosis and cytomegaly. Mitoses were noted, including abnormal mitotic figures. Small lymphocytes were intermixed occasionally with neoplastic cells.
At autopsy, the wolf was in poor nutritional condition with mildly pale mucous membranes and severe dehydration. A white-to-red well-circumscribed soft tissue mass of 12 × 8 × 6 cm effaced the left ovary. On cut surface, the mass was predominantly solid and intermixed with multiple cystic pinpoint areas (Fig. 1). The contralateral ovary was unaffected and exhibited only a few well-differentiated corpora lutea. During postmortem examination, the left ovarian mass was evaluated through imprint cytology and a fine-needle aspiration biopsy stained with Romanowsky stain. Multiple tissue samples were collected, fixed in 10% neutral-buffered formalin for 24–48 h, processed routinely for histology, and stained with hematoxylin and eosin (H&E). A presumptive diagnosis of ovarian dysgerminoma was made.
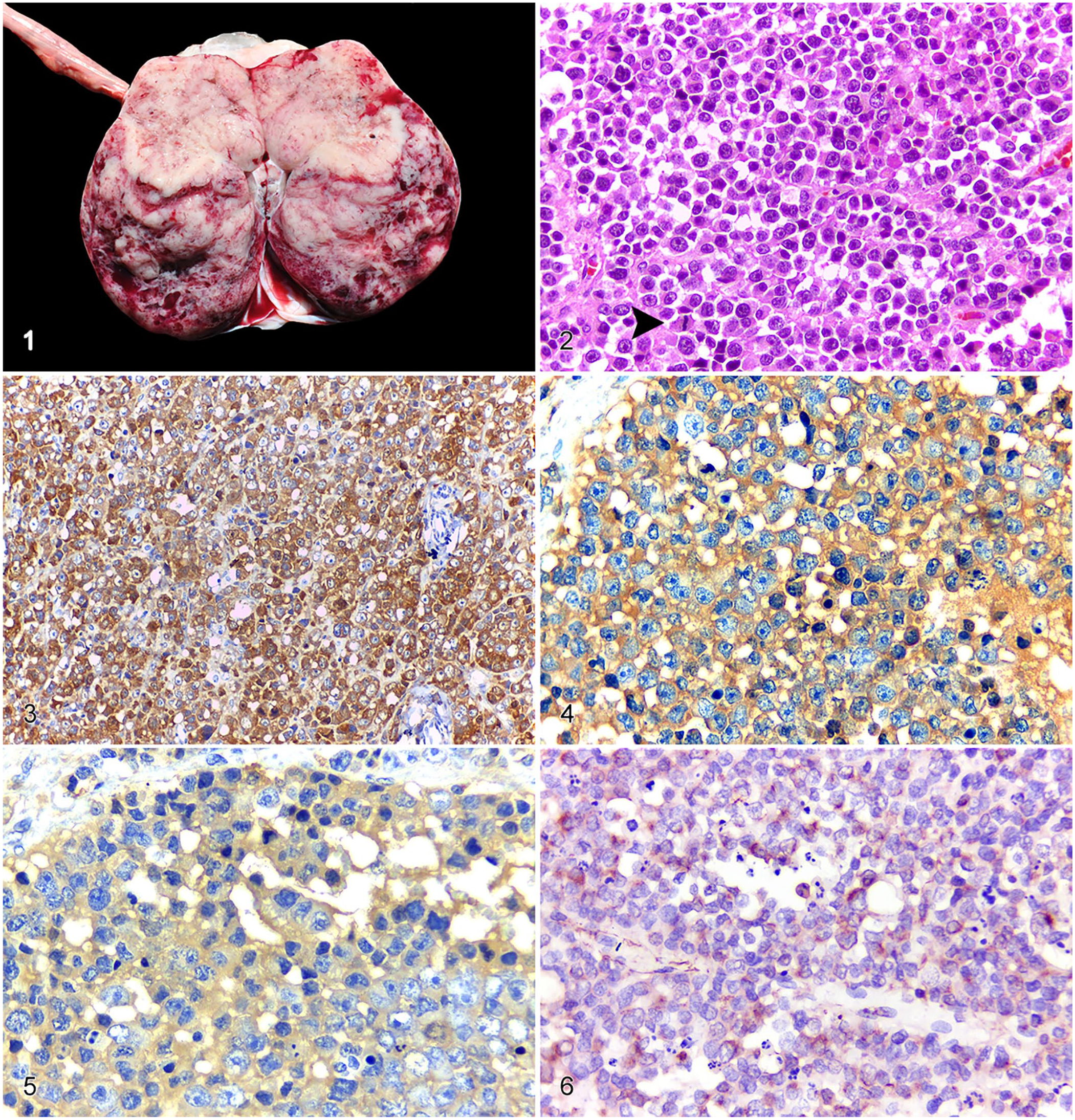
Pathologic and immunohistochemical features of unilateral ovarian dysgerminoma in a captive maned wolf.
Histologic evaluation of the ovarian mass revealed a well-circumscribed, encapsulated, and densely cellular neoplasm composed of germ cells arranged in solid sheets and supported by a scant fibrovascular connective tissue (Fig. 2). Neoplastic cells were round-to-polygonal, with distinct cell borders, and contained small amounts of faint eosinophilic cytoplasm. Nuclei were large and rounded with a high nuclear-cytoplasmic ratio, finely stippled chromatin, and multiple nucleoli. The cells had moderate anisocytosis and marked anisokaryosis, and the mitotic count was 20 mitotic figures per 10 high-power fields (2.37 mm2). Additionally, rare multinucleate neoplastic cells and abundant single cell necrosis were noted. Multifocal areas of necrosis were observed within the neoplasm, mixed with fibrin deposits, scattered hemorrhage, and mild infiltrates of lymphocytes.
We searched the records of the Veterinary Pathology Laboratory from the Universidade Federal do Rio Grande do Sul (UFRGS) from 2007 to 2020 for ovarian dysgerminomas diagnosed in domestic dogs. We included in our study only excisional biopsies received fixed in neutral-buffered formalin from dogs with a history of an abdominal space-occupying mass in the ovarian region. The fixation time for these samples was unknown, given that samples received for analysis originated from both the Veterinary Teaching Hospital of the UFRGS and from distant veterinary clinic facilities. We retrieved 13 paraffin-embedded tissues from these cases, cut sections at 3 µm, and stained with H&E for initial review. Histologically, characteristic features of ovarian dysgerminomas were observed in all cases (i.e., highly cellular tumors, large polyhedral cells arranged in broad sheets supported by scant stroma, high nuclear-cytoplasmic ratio, and variable-to-numerous mitotic figures). 1
We compared the immunolabeling of the left ovarian mass from the maned wolf with biopsies from similar cases in 13 domestic dogs. Amplification signal was obtained (MACH 4 universal HRP-polymer kit; Biocare Medical) and revealed with 3-amino-9-ethylcarbazole for the following antibodies (Biocare Medical): vimentin (V9; 1:200), pancytokeratin (AE1/AE3; ready-to-use), CD3 (F7.2.38; 1:250), CD79 αcy (HM47/A9; 1:10), and c-kit (CD117; 1:300). Positive tissue controls consisted of canine peripheral nerve sheath tumor (vimentin), canine skin (cytokeratin), porcine palatine tonsils (CD3, CD79 αcy), and canine mast cell tumor (c-kit). Broad lineage markers (pancytokeratin [AE1/AE3], vimentin, CD3, CD79αcy) were used to determine cell line differentiation. Additionally, a complete panel was performed to characterize neoplastic cell expression of specific germ cell lineage biomarkers: octamer-binding transcription factor 3/4 (OCT3/4; MRQ-10, ready-to-use; Dako), Sal-like protein 4 (SALL4; 6E3, ready-to-use; Cell Marque), and placental alkaline phosphatase (PLAP; NB10, ready to use; Dako). Immunohistochemistry for OCT3/4, PLAP, and SALL4 was performed (Ultra View universal 3,3′-diaminobenzidine detection kit; Ventana), and positive controls consisted of tissue samples of human seminoma (OCT3/4, SALL4) and human placental tissue (PLAP). All negative controls for both broad and specific germ cell lineage markers consisted of replacing the respective primary antibody with phosphate-buffered saline.
The neoplastic cells of the maned wolf ovarian neoplasm expressed marked and diffuse cytoplasmic immunolabeling for SALL4; nuclear labeling was mild and multifocal (Fig. 3). In addition, there was marked diffuse cytoplasmic immunolabeling for PLAP (Fig. 4); OCT3/4 immunolabeling was moderate and cytoplasmic (Fig. 5). Expression of c-kit was rarely seen within neoplastic cells; most cells exhibited a membranous pattern. Vimentin expression was moderate, multifocal, and perinuclear (Fig. 6). The remaining assessed markers were negative, with the exception of CD3, which labeled the background lymphocytic infiltrate, confirming the T-cell origin of these cells. Based on these findings, the neoplasm was diagnosed as a unilateral ovarian dysgerminoma.
Other pathology findings included cystic endometrial hyperplasia–pyometra syndrome, incidental cholangiocarcinoma, splenic nodular lymphoid hyperplasia, left adrenal cortical adenoma, follicular adenoma of the thyroid, and moderate mitral valve endocardiosis.
Most (9 of 13) dysgerminomas from domestic dogs were immunolabeled by SALL4 antibodies, with a predominantly nuclear pattern (7 of 9), but variable degrees of labeling were observed (Suppl. Figs. 1–3). Nonetheless, both nuclear and cytoplasmic immunolabeling were observed in 2 cases, similar to the maned wolf. The other germ cell markers, PLAP and OCT3/4, were expressed irregularly, and cytoplasmic labeling was detected in 3 and 4 cases, respectively (Suppl. Figs. 4, 5). In addition, all dysgerminoma cases in dogs consistently expressed vimentin, similar to the maned wolf dysgerminoma, with a scattered tissue distribution and perinuclear to membranous-restricted immunolabeling (Suppl. Fig. 6). Moreover, c-kit was expressed in 5 of 13 cases, with variable patterns of labeling, but mainly membranous (4 of 5), with the exception of 1 case in which stippled cytoplasmic immunolabeling was noted. The background lymphocytic infiltrate was immunolabeled for CD3 in 4 of 13 cases, confirming the T-cell origin of the cells. The remaining broad lineage markers (i.e., pancytokeratin and CD79 αcy) were negative, excluding epithelial origin of the neoplasm and ovarian lymphoma, respectively. Based on these immunohistochemical findings (Table 1) and on the histologic features of the cells, these tumors were diagnosed as ovarian dysgerminomas.
Summary of distribution and immunolabeling degree of Sal-like protein (SALL4), vimentin, placental alkaline phosphatase (PLAP), octamer-binding transcription factor 3/4 (OCT3/4), and c-kit in dysgerminomas in a maned wolf and domestic dogs.
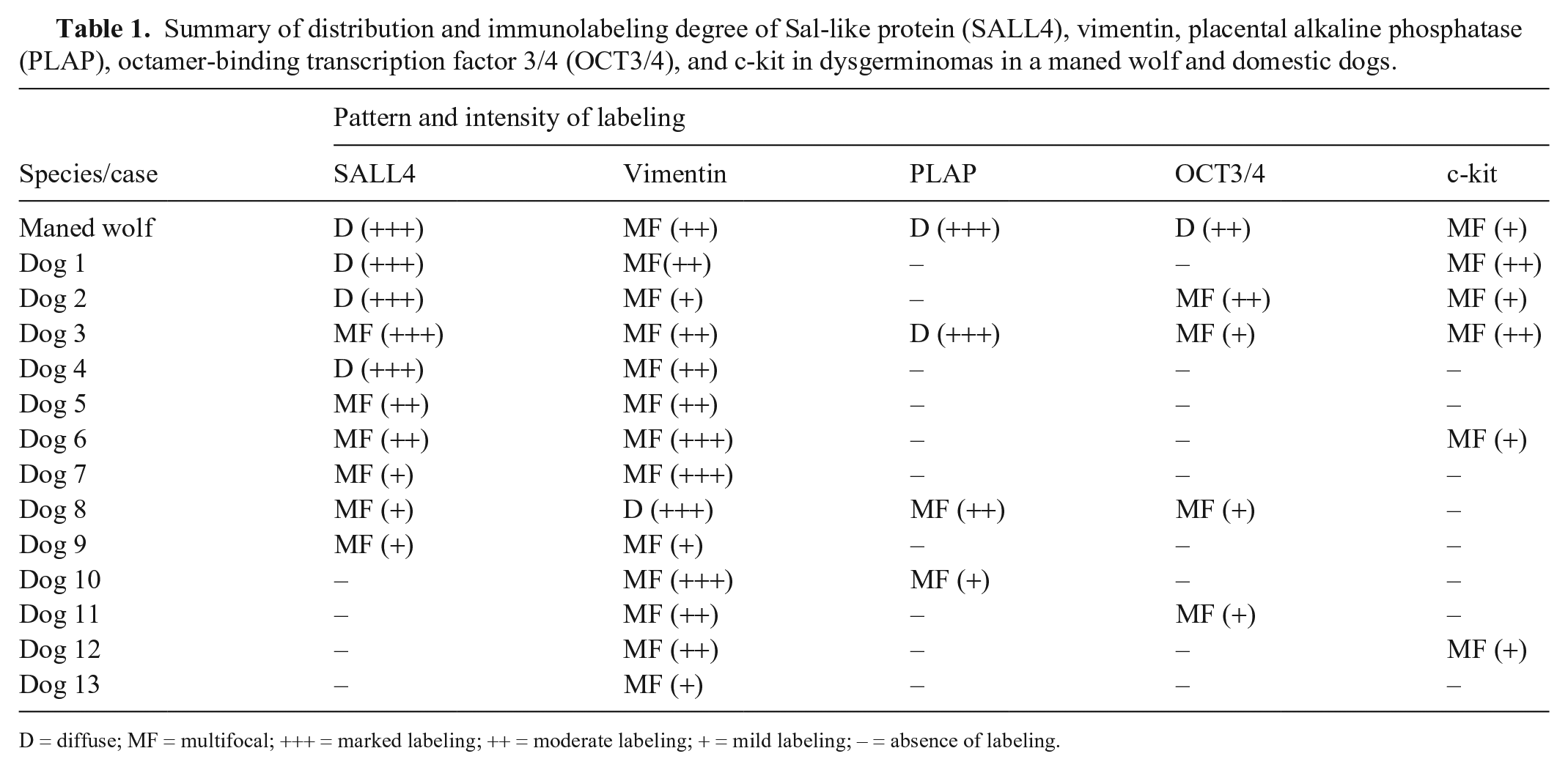
D = diffuse; MF = multifocal; +++ = marked labeling; ++ = moderate labeling; + = mild labeling; – = absence of labeling.
Dysgerminoma is an uncommon ovarian tumor in domestic animals, 1 but it has been described in most species, including maned wolves.2,15 Clinical signs associated with dysgerminomas are usually nonspecific and reflect, in part, the effects of a space-occupying mass within the abdominal cavity, 9 as observed in our case. Concurrent pyometra has been described in domestic dogs with ovarian dysgerminoma but their relationship, if any, is unknown.7,9 A post-estrus luteal phase was the most likely source of hormone production implicated in endometrial hyperplasia–pyometra syndrome in our case, as evidenced by the presence of a few well-differentiated corpora lutea in the contralateral ovary; therefore, pyometra and dysgerminoma were concurrent conditions of uncertain relationship to one another.
Dysgerminomas in canids can be either uni- or bilateral,2,9,15 with gross features characterized by total replacement of the affected ovary by a spherical mass, 1 as in our case. The cut surface of the tumor is usually white-gray, firm, and homogeneous, and may have cystic pinpoint areas of necrosis and/or hemorrhage. 1
Cytologically, dysgerminomas may have appearances distinct from other primary ovarian tumors, 4 and, therefore, a high level of agreement between cytologic and histologic diagnoses is observed.3,4 Ultrasound-guided aspiration of large masses that fill the cranial portion of the abdomen may be an inexpensive and minimally invasive tool to obtain a diagnosis. However, the gold standard for dysgerminoma diagnosis remains histologic evaluation. Dysgerminomas have a distinctive profile with large, round-to-polygonal cells arranged in sheets and, with their prominent nucleoli and high mitotic count, can be confidently identified and distinguished from other primary ovarian tumors. 1
Human dysgerminomas are usually c-kit positive with membranous immunolabeling. 17 In contrast, 8 of 14 cases in our study did not express c-kit, consistent with findings in 3 dysgerminomas in domestic dogs reported elsewhere.4,10 Although c-kit may not be useful in distinguishing dysgerminoma from other ovarian neoplasms in veterinary medicine, c-kit must be investigated for its prognostic and predictive value, given that c-kit was expressed in 6 of 14 dysgerminomas in wild and domestic canids in our investigation.
SALL4 is a transcription factor required for the development of embryonic stem cell pluripotency, 16 and is expressed consistently in malignant germ cell tumors. In addition, it has been reported to be expressed in human acute myeloid leukemia, 14 diffuse large B-cell lymphoma, 19 and mammary carcinoma. 20 Some researchers have employed this antibody in investigations in veterinary medicine, but the specificity of this marker is still unknown. 10 We observed SALL4 expression with a predominantly nuclear and/or cytoplasmic pattern both in wild and domestic canids in 10 of the 14 cases that we examined, which reinforces that this antibody may be employed to differentiate germ cell tumors in canid ovarian masses. Although exclusive nuclear labeling has been reported frequently in humans, 16 cytoplasmic labeling was also observed in our investigation, both in the maned wolf and in 2 dogs. Despite antigen detection in a cellular location not consistent with its function, like most proteins, transcription factors are translated in the cell’s cytoplasm, explaining why cytoplasmic labeling was observed occasionally. Furthermore, cytoplasmic expression of SALL4 seems to be related to poorer prognoses and malignant tumor phenotype in certain types of human cancers. 20
PLAP is another sensitive marker for germ cell tumors in humans, 16 as observed in a case series that evaluated 15 dysgerminomas, of which 14 were labeled for PLAP, exhibiting diffuse cytoplasmic staining. 13 This marker has been used only rarely in veterinary medicine, and negative results are reported for dysgerminomas in domestic dogs. 10 Our finding of mild cytoplasmic immunolabeling is similar to previous observations in human seminomas and dysgerminomas. 16 PLAP and OCT3/4 markers were irregularly expressed in the cases of dysgerminoma in dogs, whereas they were expressed markedly and moderately in the maned wolf, respectively. A possible reason is the unknown formalin fixation time of the archival ovarian samples examined in our investigation, which may have resulted in decreased antigenicity and immunolabeling for these markers. 18
Although we did not detect metastases in the maned wolf, germ cell tumors should be treated as malignant neoplasms; metastases may occur to regional, mesenteric, and mediastinal lymph nodes, adjacent organs, and by transcoelomic spread through the peritoneal cavity.1,15 Dysgerminoma is usually unilateral, but bilateral presentation has been also reported.2,15 In our case, the contralateral ovary was examined carefully, but no neoplastic cells were noted.
Supplemental Material
sj-pdf-1-jvd-10.1177_10406387211019959 – Supplemental material for Comparison of immunohistochemical profiles of ovarian germ cells in dysgerminomas of a captive maned wolf and domestic dogs
Supplemental material, sj-pdf-1-jvd-10.1177_10406387211019959 for Comparison of immunohistochemical profiles of ovarian germ cells in dysgerminomas of a captive maned wolf and domestic dogs by Rafael B. Rosa, Matheus V. Bianchi, Paula R. Ribeiro, Fernando F. Argenta, Andréia Vielmo, Felipe A. B. de Sousa, Manoela M. Piva, Camila B. Pohl, Yasmin Daoualibi, Fernanda G. Cony, Carolina B. Brunner, Raquel Von Hohendorff and Luciana Sonne in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the staff veterinarians at the Sapucaia do Sul Zoological Park for their assistance, and Gabriela Remonatto (pharmaceutical biochemistry) and Francine Hehn de Oliveira (adjunct professor) of the Pathology Department of the Medical School of UFRGS (FAMED-UFRGS) for consulting service and immunohistochemistry slide preparation for SALL4, OCT3/4, and PLAP.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received scholarship grants from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
